Adherent Cell Detachment: Techniques, Challenges, and Future Directions for Biomedical Research
This article provides a comprehensive overview of adherent cell detachment, a critical yet challenging step in cell culture for researchers, scientists, and drug development professionals.
Adherent Cell Detachment: Techniques, Challenges, and Future Directions for Biomedical Research
Abstract
This article provides a comprehensive overview of adherent cell detachment, a critical yet challenging step in cell culture for researchers, scientists, and drug development professionals. It covers the foundational science of cell adhesion, details established and emerging detachment methodologies, offers practical troubleshooting and optimization strategies, and presents a comparative analysis of techniques to validate their impact on cell viability, surface markers, and downstream applications. The scope extends from routine subculturing to the demands of large-scale biomanufacturing for cell therapies and regenerative medicine.
The Science of Cell Adhesion: Why Detaching Cells is a Fundamental Challenge
Defining Adherent Cells and Anchorage Dependence in Cell Culture
Adherent cell cultures represent a fundamental methodology in biological research and biomanufacturing, defined by the requirement for cells to attach to a solid, growth-promoting surface in order to proliferate—a biological imperative known as "anchorage dependence" [1] [2]. This cellular behavior contrasts sharply with suspension cultures, where cells proliferate freely in liquid medium without surface attachment requirements. Most vertebrate-derived cells (with the notable exception of hematopoietic cells) are anchorage-dependent, requiring a two-dimensional monolayer to facilitate the essential processes of cell adhesion, spreading, and replication [2]. These morphological characteristics form the basis of cellular classification in culture, with fibroblast-like cells exhibiting a linear, stretched shape and migratory behavior when attached, while epithelial-like cells display a wider, polygonal morphology and remain relatively stationary on the monolayer [2].
The biological significance of anchorage dependence extends beyond simple physical attachment. Normal, non-transformed tissue-derived cells (including most stem cells) absolutely require this culture support for self-renewal and differentiation, with its absence triggering growth arrest and induction of anoikis—a specific form of programmed cell death induced when anchorage-dependent cells detach from their surrounding extracellular matrix (ECM) [3]. This critical dependency positions adherent cell culture as an indispensable technology across virology, drug discovery, regenerative medicine, and basic biological research, with recent advancements in detachment methodologies addressing long-standing challenges in cell viability, phenotype preservation, and process scalability [3] [4].
Biological Principles of Anchorage Dependence
Molecular Mechanisms of Cell Adhesion
The anchorage dependence of cells is mediated through sophisticated molecular machinery that connects the extracellular environment to intracellular signaling pathways. Integrins—transmembrane receptor proteins—serve as the primary mediators of cell adhesion by binding to specific ligands in the extracellular matrix and to cytoskeletal components within the cell. This bidirectional signaling not only provides structural anchorage but also transmits critical survival and proliferation signals that prevent anoikis [3]. The adhesion process involves sequential engagement of adhesion receptors, clustering of integrins at focal adhesion sites, and activation of intracellular signaling cascades that regulate cell cycle progression and metabolic activity.
Calcium ions play a crucial role in maintaining integrin-mediated adhesion, which explains the effectiveness of calcium chelators like EDTA in cell detachment protocols [5]. The mechanical properties of the substrate, including its stiffness and topography, are actively sensed by cells through these adhesion complexes and influence diverse cellular behaviors including migration, differentiation, and gene expression. This mechanotransduction capability means that adherent cells not only respond to biochemical cues but also to the physical properties of their attachment surface, creating a complex regulatory network that governs cell fate decisions in both physiological and culture conditions [3].
Implications for Research and Therapy
The anchorage-dependent nature of most primary cells carries profound implications for biomedical research and therapeutic development. In contrast to transformed tumor cells that often proliferate independently of attachment, normal cells require appropriate adhesion signaling to maintain viability and function, making faithful recapitulation of these signals essential for physiologically relevant culture models [3]. This requirement becomes particularly critical in stem cell research and regenerative medicine, where the culture environment must mimic natural stem cell niches containing appropriate surface-bound signaling factors, cell-cell contacts, ECM components, and biomechanical microenvironments to maintain pluripotency or direct differentiation along specific lineages [3].
The dependency on adhesion signaling also creates significant challenges for large-scale biomanufacturing processes. Traditional two-dimensional culture systems face inherent limitations in surface-area-to-volume ratios, restricting cell yield and necessitating the development of specialized technologies like microcarriers and fixed-bed reactors for industrial-scale applications [3]. Furthermore, the sensitivity of adherent cells to shear stress in bioreactor environments requires careful engineering of culture conditions to maintain viability and functionality while achieving necessary production scales for vaccines, cell therapies, and other biological products [3].
Established Cell Detachment Methodologies
The requirement to periodically detach adherent cells for subculturing or analysis has led to the development of various methodological approaches, each with distinct advantages, limitations, and effects on cellular properties. These methods can be broadly categorized as enzymatic, non-enzymatic, and mechanical techniques.
Enzymatic Detachment Methods
Enzymatic detachment remains the most widely used approach for dissociating adherent cells, utilizing proteolytic enzymes to cleave the proteins mediating cell-surface attachment.
Trypsin: The historical gold standard for cell detachment, trypsin cleaves peptides after lysine or arginine residues, effectively degrading most cell surface proteins and extracellular matrix components. The procedure involves washing cells with a balanced salt solution without calcium and magnesium to remove serum inhibitors, followed by application of pre-warmed trypsin (approximately 0.5 mL per 10 cm²) and incubation at room temperature for 2-5 minutes until ≥90% of cells have detached [1]. The enzymatic action is then neutralized with complete growth medium containing serum, and cells are centrifuged (200 × g for 5-10 minutes) before resuspension and counting [1]. While efficient, trypsinization causes substantial damage to surface proteins and requires careful control of incubation time to maintain cell viability.
Accutase: Considered a milder enzymatic alternative, Accutase is formulated for gentle detachment while better preserving cell surface markers [5] [6]. It contains proteolytic activity that gently breaks down cellular adhesion molecules and offers the practical advantage of not requiring neutralization or wash steps [6]. However, recent research has revealed that Accutase can compromise specific surface proteins, notably causing significant decreases in surface Fas ligands and Fas receptors by cleaving them into fragments [5]. Surface expression of these proteins requires approximately 20 hours to recover after Accutase treatment, highlighting the importance of allowing adequate recovery time before experiments investigating these markers [5].
Table 1: Comparative Analysis of Enzymatic Cell Detachment Reagents
| Reagent | Mechanism of Action | Incubation Conditions | Advantages | Limitations |
|---|---|---|---|---|
| Trypsin | Cleaves after lysine/arginine residues | 2-5 minutes at room temperature [1] | Highly efficient; well-established protocol | Damages most surface proteins; requires serum neutralization |
| Accutase | Proteolytic blend with collagenolytic and DNase activities | Ready to use once thawed; do not pre-warm to 37°C [6] | Gentle on cells; no neutralization required | Compromises specific surface proteins (FasL, Fas); requires recovery time [5] |
| TrypLE | Recombinant fungal trypsin-like protease | Similar to trypsin [1] | Animal-origin free; consistent performance | Variable efficiency across cell types |
Non-enzymatic and Mechanical Methods
Non-enzymatic approaches offer alternatives that avoid proteolytic damage to cell surfaces, preserving important markers and functions.
EDTA-Based Solutions: Ethylenediaminetetraacetic acid (EDTA) facilitates cell detachment by chelating calcium ions essential for integrin-mediated adhesion [5]. This method is considered mild and better preserves surface protein integrity, as demonstrated by maintained expression of Fas ligands and Fas receptors compared to enzymatic treatments [5]. However, EDTA alone is often insufficient for strongly adherent cells and frequently requires mechanical assistance through scraping or pipetting, which can potentially damage cells through mechanical stress.
Cell Scraping: This purely mechanical approach involves physically dislodging cells using a sterile scraper tool. Research indicates that scraping preserves the highest levels of surface FasL expression compared to both enzymatic and chemical detachment methods [5]. The significant drawback is the potential for substantial cellular damage, reduced viability, and generation of heterogeneous cell populations due to variable application of mechanical force.
Novel Electrochemical Approach: Recent innovation from MIT introduces an enzyme-free strategy using alternating electrochemical current on a conductive biocompatible polymer nanocomposite surface [4]. By applying low-frequency alternating voltage, this platform disrupts adhesion within minutes while maintaining over 90% cell viability, addressing limitations of both enzymatic and mechanical methods [4]. The approach demonstrates 95% detachment efficiency for human cancer cells (osteosarcoma and ovarian cancer) while minimizing waste and compatibility concerns associated with animal-derived enzymes, showing particular promise for large-scale biomanufacturing and automated workflows [4].
Diagram 1: Cell Detachment Method Selection Workflow. This decision tree illustrates the systematic selection of detachment methods based on experimental requirements and cell type considerations.
Quantitative Comparison of Detachment Method Impacts
The choice of detachment method significantly influences multiple cellular parameters, including viability, surface marker integrity, and functional characteristics. Understanding these quantitative impacts is essential for selecting appropriate methodologies for specific applications.
Table 2: Quantitative Effects of Cell Detachment Methods on Cellular Parameters
| Detachment Method | Cell Viability | Detachment Efficiency | Surface Protein Preservation | Recovery Time Required | Typical Application |
|---|---|---|---|---|---|
| Trypsin | Variable (protocol-dependent) | High (≥90%) [1] | Low (damages most surface proteins) | Immediate use possible | Routine subculturing |
| Accutase | High (gentler than trypsin) [5] | High (comparable to trypsin) | Moderate (compromises specific proteins like FasL/Fas) [5] | ~20 hours for full recovery of affected markers [5] | Flow cytometry (with caution) |
| EDTA-Based | High (mild chemical action) | Low to moderate (may require scraping) [5] | High (best for surface markers) [5] | Minimal | Surface protein analysis |
| Scraping | Moderate (mechanical damage) [5] | High for adherent cells | Highest (preserves all surface proteins) [5] | Immediate use possible | Critical surface marker studies |
| Electrochemical | >90% [4] | 95% [4] | Expected high (non-proteolytic) | Under investigation | Large-scale biomanufacturing |
The data reveal significant trade-offs between detachment efficiency, viability, and phenotype preservation. While enzymatic methods offer efficiency and convenience, they compromise surface protein integrity—a critical consideration for immunophenotyping, receptor studies, and functional assays. EDTA-based approaches better preserve surface markers but may require mechanical assistance for strongly adherent cells. The novel electrochemical method demonstrates exceptional promise with high efficiency and viability while avoiding proteolytic damage, though its comprehensive effects on diverse surface markers require further characterization across cell types [4].
Advanced Research Applications and Implications
Impact on 3D Culture Models and Drug Discovery
The limitations of traditional two-dimensional adherent culture have driven the development of three-dimensional (3D) models that better recapitulate in vivo tissue architecture and functionality. These advanced culture systems present unique challenges and considerations for cell detachment methodologies. 3D cultures demonstrate enhanced physiological relevance compared to 2D monolayers by preserving tissue-specific architecture, supporting critical cell-matrix interactions, and maintaining appropriate expression levels of essential proteins [7] [8]. The transition from 2D to 3D models reveals important scaffold-dependent variability in culture outcomes, as demonstrated in prostate cancer research where different scaffolding materials (Matrigel, GelTrex, and GrowDex) produced significantly different spheroid formation and gene expression patterns, including variations in androgen receptor expression and neuroendocrine marker genes [9].
The detachment of cells from 3D culture systems introduces additional complexity compared to monolayer dissociation. Enzymatic treatments often require longer exposure times and higher concentrations to penetrate matrix materials, potentially increasing damage to cell surface markers and functionality. These challenges have stimulated the development of specialized dissociation protocols for 3D cultures, often incorporating combination approaches using collagenases, dispase, and other matrix-specific enzymes alongside traditional trypsin or Accutase. The preservation of cell viability and phenotype during dissociation from 3D environments remains a significant technical hurdle, particularly for sensitive primary cells and stem cells, driving ongoing research into gentler, more specific detachment strategies [9] [7].
Automation and Artificial Intelligence in Cell Culture
Recent technological advances have introduced automation and artificial intelligence to address issues of consistency and efficiency in adherent cell culture workflows. Traditional visual assessment of cell morphology and confluency introduces substantial inter-operator variability, leading to inconsistencies in subculturing timing and experimental outcomes [10]. Automated monitoring systems like the Olympus Provi CM20 incubation monitoring system utilize AI technology to quantitatively measure culture health parameters including confluency and cell counts using consistent analysis parameters, eliminating subjective assessment and improving reproducibility [10].
These automated systems provide significant advantages for detachment timing decisions by continuously monitoring multiple points in culture vessels and detecting subtle changes in cell morphology and density that might precede morphological deterioration. By establishing standardized parameters for subculturing based on quantitative metrics rather than subjective assessment, automated systems reduce variability in experimental results and improve the reliability of downstream applications including drug screening and functional assays [10]. The integration of such automated assessment with novel detachment technologies like the electrochemical approach represents the future direction of adherent cell culture, enabling closed-system, minimally variable workflows from culture expansion to harvest.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagents and Materials for Adherent Cell Culture and Detachment
| Reagent/Material | Composition/Type | Primary Function | Application Notes |
|---|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme + calcium chelator | Dissociates adherent cells by cleaving adhesion proteins | Requires serum neutralization; monitor incubation time carefully [1] |
| Accutase | Proteolytic, collagenolytic, and DNase activities | Gentle cell detachment and single-cell suspension preparation | Does not require neutralization; avoid pre-warming to 37°C [6] |
| TrypLE | Recombinant fungal protease | Animal-origin-free enzymatic dissociation | Consistent performance; suitable for therapeutic applications |
| EDTA Solution | Ethylenediaminetetraacetic acid | Calcium chelation disrupts integrin-mediated adhesion | Mild method; may require mechanical assistance for strongly adherent cells [5] |
| DPBS (without Ca²⁺/Mg²⁺) | Balanced salt solution | Washing step to remove serum and ions prior to dissociation | Essential pre-treatment to prevent enzyme inhibition [1] |
| Complete Growth Medium | Basal medium + serum/growth factors | Neutralizes enzymatic activity and provides nutrients | Required after trypsinization; not needed for Accutase [1] [6] |
| Matrigel | Basement membrane matrix | 3D culture substrate mimicking extracellular matrix | Promotes consistent spheroid formation; variable between batches [9] |
| GrowDex | Plant-derived nanofibrillar cellulose | Bioincompatible 3D scaffold for cell culture | Sustainable alternative to animal-derived matrices [9] |
| Anti-adherence Solution | Polymer coating | Prevents cell attachment for spheroid formation | Enables spheroid generation in standard plates at reduced cost [8] |
Future Directions in Adherent Cell Detachment Research
The field of adherent cell detachment is evolving toward methods that better preserve cellular integrity while improving scalability and reproducibility. Several promising directions are emerging from current research:
Electrochemically Enabled Biomanufacturing: The development of enzyme-free electrochemical detachment platforms represents a paradigm shift with potential to transform large-scale biomanufacturing [4]. By applying low-frequency alternating voltage to conductive biocompatible surfaces, this approach achieves high detachment efficiency (>95%) while maintaining excellent viability (>90%) and eliminating concerns associated with animal-derived enzymes [4]. The technology offers particular promise for automated, closed-loop cell culture systems in therapeutic manufacturing, especially for sensitive cell types like primary immune cells for CAR-T therapies where phenotype preservation is critical.
Advanced Biomimetic Surfaces: Research continues into smart culture surfaces with dynamically tunable properties that can be switched from adhesive to non-adhesive states through external stimuli including light, temperature, or electrical signals. These platforms would enable controlled cell release without enzymatic or mechanical stress, potentially revolutionizing both research-scale and industrial-scale culture processes.
Integration with AI-Driven Culture Management: The combination of automated monitoring systems with advanced detachment technologies creates opportunities for fully optimized culture workflows [10]. AI algorithms can precisely determine optimal detachment timing based on quantitative metrics rather than subjective assessment, then trigger appropriate detachment protocols tailored to specific cell types and applications, significantly improving consistency and efficiency across research and manufacturing environments.
These advancing methodologies collectively address the core challenges in adherent cell detachment—balancing efficiency with phenotype preservation, scaling processes while maintaining viability, and standardizing protocols for reproducibility—paving the way for more reliable and physiologically relevant cell-based research and therapies.
The Role of the Extracellular Matrix (ECM) and Adhesion Proteins in Cell Attachment
The Extracellular Matrix (ECM) is a dynamic, three-dimensional network of macromolecules that provides not only structural support to tissues but also critical biochemical and mechanical cues that regulate cellular behavior including adhesion, migration, differentiation, and signal transduction [11]. For adherent cells, the ECM serves as the physical scaffold for attachment, a process mediated primarily by specialized transmembrane receptors, most notably integrins [12]. The interaction between cells and the ECM is not a passive anchoring event but a complex, bidirectional signaling process essential for maintaining tissue homeostasis. Understanding these mechanisms is foundational to the field of adherent cell detachment research, which aims to develop techniques for harvesting cells with minimal damage to their functional and metabolic activity for applications in tissue engineering, regenerative medicine, and industrial cell manufacturing [13].
This technical guide will detail the core components of the ECM, the fundamental mechanisms of cell adhesion, and the experimental methodologies used to study these interactions. The content is framed within the context of advancing adherent cell detachment research, a field that must balance the need for efficient cell release with the imperative to preserve cell viability, function, and surface protein integrity [13].
Core Components of the Extracellular Matrix
The ECM's composition is highly variable across tissues, but its core structural and functional molecules can be categorized as follows:
Major Structural Proteins
- Collagens: The most abundant proteins in the human body, collagens are fibrous proteins with a unique triple-helix structure that provide tensile strength and structural integrity to tissues. Multiple types exist (e.g., I, II, III, IV), each with specialized functions and tissue distributions [14] [11].
- Elastin: A highly elastic protein that allows tissues such as skin, lungs, and blood vessels to stretch and recoil. It forms cross-linked networks that provide functional resilience and flexibility, working in concert with collagen to determine tissue mechanical properties [14] [11].
Specialized Glycoproteins and Polysaccharides
- Fibronectin: A high-molecular-weight glycoprotein that plays a crucial role in cell adhesion and migration. It contains specific domains, such as the Arg-Gly-Asp (RGD) sequence, which are recognized by cell surface integrins [11] [15].
- Laminin: A key component of the basement membrane, laminin is essential for forming networks that support cell adhesion, proliferation, and differentiation [15].
- Proteoglycans (PGs) and Glycosaminoglycans (GAGs): PGs are glycosylated proteins characterized by covalently attached GAG chains. GAGs are long, linear polysaccharides with repeating disaccharide units (e.g., heparan sulfate, chondroitin sulfate, keratan sulfate, hyaluronan) [14]. These molecules serve as dynamic regulators of tissue mechanics, modulate cellular responses to mechanical stimuli, and interact with growth factors and cell surface receptors [14]. Key PGs include syndecans, glypicans, and perlecans.
Table 1: Major ECM Components and Their Primary Functions
| ECM Component | Primary Function | Key Characteristics |
|---|---|---|
| Collagens | Provides tensile strength and structural integrity. | Triple-helix structure; most abundant protein in the body. |
| Elastin | Confers elasticity and recoil to tissues. | Hydrophobic amino acids (Gly, Ala); cross-linked networks. |
| Fibronectin | Mediates cell adhesion and migration. | Contains RGD integrin-binding motif. |
| Laminin | Basement membrane foundation; cell adhesion. | Forms sheet-like networks; crucial for epithelial cells. |
| Proteoglycans/GAGs | Regulates hydration, tissue mechanics, and signaling. | Highly negative charge; interacts with growth factors. |
The physical properties of the ECM—including its stiffness, viscoelasticity, and topology—are not passive traits but active regulators of cell behavior. For instance, ECM stiffness can influence cell proliferation, migration, and differentiation, with dysregulation implicated in diseases like cancer and fibrosis [11]. The stiffness of healthy tissues can range from <2 kPa in the brain to 40–55 MPa in bone, while pathological states like breast cancer tumors can exhibit a significant stiffening (e.g., ~4 kPa vs. 0.167 kPa in normal tissue) [11].
Fundamental Mechanisms of Cell Adhesion
Cell adhesion to the ECM is a multi-step process orchestrated by a complex machinery of transmembrane receptors, intracellular adaptors, and the cytoskeleton.
Integrins: The Primary Transmembrane Receptors
Integrins are the principal cell surface receptors that mediate adhesion to the ECM. They are α/β heterodimeric transmembrane proteins, with 18 α and 8 β subunits forming 24 distinct integrins in mammals [12]. They bind to specific motifs in ECM proteins:
- RGD-binding integrins (e.g., αvβ3, αvβ5, α5β1) recognize the Arg-Gly-Asp sequence found in fibronectin, vitronectin, and other proteins [12] [15].
- Collagen-binding integrins (e.g., α1β1, α2β1) recognize the GFOGER motif in collagens [12].
- Laminin-binding integrins (e.g., α3β1, α6β1, α6β4) bind to laminins in the basement membrane [12].
Integrin-mediated adhesion is a dynamic and regulated process. Integrins exist in inactive (bent) and active (extended) conformations. The switch to an active state, which increases affinity for ECM ligands, can be triggered by intracellular signals (inside-out signaling). Upon ligand binding, integrins cluster and initiate outside-in signaling, recruiting a plethora of cytoplasmic proteins to form adhesion plaques [12] [16].
Focal Adhesions and the Adhesome
The linkage of integrins to the actin cytoskeleton is facilitated by a multi-protein complex known as the adhesome, which assembles into focal adhesions (FAs) [16]. Key adaptor and signaling proteins include:
- Talin: Binds directly to the cytoplasmic tail of β-integrins, activating them and recruiting other proteins like vinculin [16].
- Vinculin: Strengthens the integrin-cytoskeleton linkage by simultaneously binding to talin and actin [16].
- Focal Adhesion Kinase (FAK): A critical non-receptor tyrosine kinase that transduces integrin-mediated signals, regulating cell spreading, migration, and survival [17] [15].
- Paxillin: An adaptor protein that recruits numerous signaling and structural molecules to focal adhesions [18] [15].
The following diagram illustrates the core mechanotransduction pathway from the ECM to the cytoskeleton and nucleus:
Diagram 1: Core ECM-integrin mechanotransduction pathway
Other Adhesion Structures
While integrins are central to cell-ECM adhesion, cells also form other specialized junctions:
- Adherens Junctions: Mediate cell-cell adhesion through cadherin receptors, which are linked to the actin cytoskeleton via catenins (β-catenin, α-catenin) [17].
- Tight Junctions: Form impermeable seals between cells, regulating paracellular transport and maintaining cell polarity. Key components include claudin and occludin [17].
Experimental Methodologies for Studying Cell Adhesion
Microcontact Printing and Patterned Substrates
Objective: To engineer defined areas of ECM proteins to study how spatially confined ECM ligands regulate cell adhesion initiation and strength [18].
Detailed Protocol:
- Fabrication of PDMS Stamps: A silicon mold with circular holes of defined diameters (e.g., 2 μm to 10 μm) is used to cast poly(dimethyl)siloxane (PDMS) micropillar stamps [18].
- Inking: The PDMS micropillars are "inked" with a solution of fluorescently labeled ECM protein (e.g., collagen I or fibronectin) [18].
- Printing: The inked stamp is brought into physical contact with a glass coverslip, transferring the protein onto the glass in a defined pattern [18].
- Passivation: The non-patterned glass surface is passivated with bovine serum albumin (BSA) or a non-integrin-binding protein fragment (e.g., FNIII7-10ΔRGD) to prevent non-specific cell attachment [18].
- Cell Seeding and Analysis: Cells are seeded onto the patterned substrate. Adhesion is assessed via confocal microscopy (e.g., staining for actin and paxillin) or functional adhesion assays [18].
Single-Cell Force Spectroscopy (SCFS)
Objective: To quantify the initiation and strength of cell adhesion at high temporal and force resolution [18].
Detailed Protocol:
- Cantilever Functionalization: An atomic force microscopy (AFM) microcantilever is coated with concanavalin A (ConA) to facilitate cell attachment [18].
- Cell Capture: A single, rounded cell is attached to the ConA-coated cantilever [18].
- Adhesion Initiation: The cell is positioned above a specific ECM pattern (created via microcontact printing) and brought into contact for a defined period (e.g., 5 to 360 seconds) to allow adhesion formation and strengthening [18].
- Force Quantification: The cantilever is retracted, and the force required to detach the cell from the substrate is measured. This provides a direct quantification of adhesion force and energy [18].
The following diagram visualizes this integrated experimental workflow:
Diagram 2: Microcontact printing and SCFS workflow
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Cell Adhesion and Detachment Research
| Research Reagent | Function and Application | Example Use-Case |
|---|---|---|
| Trypsin-EDTA | Enzymatic cell detachment; trypsin cleaves ECM proteins, EDTA chelates calcium. | Standard, robust method for harvesting adherent cells from culture vessels [13]. |
| Collagenase | Enzyme that specifically degrades native collagen fibrils in the ECM. | Isolation of cells from tissues rich in collagen, such as bone or tendon. |
| Cilengitide | Cyclic RGD peptide; selective inhibitor of αvβ3 and αvβ5 integrins. | Studying integrin function and reversing Cell Adhesion-Mediated Drug Resistance (CAMDR) [15]. |
| FNIII7-10ΔRGD | A fibronectin fragment lacking the integrin-binding RGD domain. | Used as a passivation agent to block non-specific cell adhesion on substrates [18]. |
| Anti-Integrin Antibodies | Block specific integrin subtypes to study their function or activate signaling. | Functional studies to dissect the role of specific integrin heterodimers in adhesion. |
| Recombinant ECM Proteins (Fibronectin, Laminin, Vitronectin) | Coat culture surfaces to study cell adhesion on specific ECM components. | Investigating CAMDR or creating defined microenvironments for cell culture [15]. |
Implications for Adherent Cell Detachment Research
The fundamental understanding of cell adhesion directly informs the challenges and opportunities in adherent cell detachment research. The primary goal is to disrupt the very adhesion mechanisms described above while preserving cell health and function [13].
- Enzymatic Detachment: The most common method uses proteolytic enzymes like trypsin to cleave ECM proteins and cell surface receptors. While effective and robust, it can damage critical cell surface proteins, boost apoptotic rates, and dysregulate protein expression, which is undesirable for therapeutic applications [13].
- Non-Enzymatic Detachment:
- Chelating Agents (e.g., EDTA): Bind calcium ions, disrupting calcium-dependent cell adhesion molecules like cadherins. They are less damaging than enzymes but may not be effective for all cell types and can introduce toxicity [13].
- Physical Methods: Scraping, thermal changes, or application of magnetic/electric fields. These are simple and avoid chemical residues but can be difficult to scale and control precisely [13].
- Stimuli-Responsive Surfaces: Advanced materials like thermo-responsive polymers change their properties (e.g., become hydrophilic/hydrophobic) with temperature shifts, allowing controlled cell release without enzymes. Challenges include cost, robustness, and the need for precise control over material characteristics [13].
A critical application is in bioreactor-based cell manufacturing using microcarriers. Developing robust, scalable detachment methods for these systems, such as microcarriers with stimuli-responsive coatings, is a rapidly growing area of research to meet the demands of the expanding cell manufacturing industry [13].
Furthermore, ECM adhesion has been directly linked to Cell Adhesion-Mediated Drug Resistance (CAMDR), a phenomenon where adhesion to ECM proteins like laminin, vitronectin, and fibronectin confers resistance to chemotherapeutic agents in cancer cells, including glioblastoma. This resistance is mediated through integrin αv and the FAK/paxillin/AKT signaling pathway, which suppresses p53-mediated apoptosis [15]. This underscores the therapeutic relevance of understanding and modulating cell-ECM interactions.
Table 3: Common Cell Detachment Methods and Their Characteristics
| Detachment Method | Mechanism of Action | Key Advantages | Key Limitations |
|---|---|---|---|
| Trypsin-EDTA | Proteolysis + Calcium Chelation | Highly effective, robust, low cost. | Damages cell surface proteins; can dysregulate metabolism [13]. |
| Collagenase | Degrades Collagen | Specific for collagen-rich matrices. | Limited to specific ECM contexts; potential for residual enzyme activity. |
| Chelate-Free Buffers | Ionic Disruption of Adhesion | Simpler formulation; no enzyme residuals. | May be less effective for strongly adherent cells [13]. |
| Thermo-Responsive Polymers | Temperature-induced surface hydration change | Non-enzymatic; allows harvest of intact cell sheets. | Requires precise material control; less robust; more expensive [13]. |
| Mechanical Scraping | Physical Shearing Force | Simple; no chemicals or enzymes. | Causes significant cell damage and death; not scalable [13]. |
Cell Detachment as a Critical Step in Subculturing and Cell Harvesting
Cell detachment is a fundamental laboratory procedure essential for the subculturing (passaging) of adherent cells and for harvesting cells for downstream experiments and applications. Adherent cell cultures are characterized by their need to attach to a solid, growth-promoting substrate to proliferate, a property known as "anchorage dependence" [1]. The process of detaching these cells from their culture surface is therefore a critical step that can significantly impact cell health, viability, and the reliability of subsequent experimental data. In the broader context of adherent cell detachment research, the central challenge lies in efficiently breaking the bonds between the cell and its substrate while minimizing damage to delicate cell membranes and functionally important surface proteins [13]. This guide provides an in-depth technical overview of the principles, methods, and quantitative analyses of cell detachment, framed for researchers, scientists, and drug development professionals.
Fundamental Mechanisms of Cell Adhesion and Detachment
The Biological Basis of Cell Adhesion
Understanding detachment first requires an understanding of how cells adhere. Cell adhesion to a substrate occurs primarily through interactions with the extracellular matrix (ECM), a three-dimensional network of proteins, proteoglycans, and glycosaminoglycans that serves as the foundational scaffold [13]. The key cell-matrix adhesion structures include:
- Focal Adhesions: These are specific sites on the cell membrane where transmembrane proteins called integrins cluster to form a mechanical link between the ECM outside the cell and the actin cytoskeleton inside the cell [13].
- Hemidesmosomes: These junctions provide stable adhesion by connecting intermediate filaments of the cytoskeleton to the ECM, particularly in epithelial cells [13].
The adhesion process is biphasic. The initial phase (within seconds to minutes of contact) is dominated by non-specific, rapid electrostatic interactions. The mature phase (developing over hours to days) is characterized by the formation of specific, protein-mediated bonds, such as those in focal adhesions, which are orders of magnitude stronger [19].
Theoretical Framework of Detachment Forces
The physical process of detaching a cell can be analyzed through the lens of cell mechanics. Research dedicated to comparing cell-cell detachment forces in different experimental setups—such as pipette-pipette, plate-plate, and plate-pipette assays—reveals that the measured detachment force is not an intrinsic property of the cell alone but a global property of the entire system [20] [21] [22].
Theoretical models based on Young-Laplace equations describe cell shape under an applied external force. In a pipette-pipette setup, for instance, the force ((F)) required to detach two identical adherent cells is given by: [ F{dc} = \frac{1}{2} \pi \gamma rH \cos^2 \thetac ] where (\gamma) is the cell surface tension, (rH) is the radius of curvature related to the cell's internal pressure, and (\theta_c) is the cell-cell contact angle determined by the adhesion energy [20] [21]. This model highlights that the measured detachment force depends not only on cell-specific parameters (adhesion tension, surface tension) but also on experimental setup parameters (e.g., pipette radius and pressure) [20] [21].
The following diagram illustrates the key interactions and forces at play during the adhesion and detachment processes.
Established Cell Detachment Methods: A Technical Comparison
A wide array of techniques has been developed to detach adherent cells, each with distinct mechanisms, advantages, and limitations. These can be broadly categorized into chemical and physical methods.
Chemical and Enzymatic Methods
Enzymatic Methods: These involve the use of proteases to cleave the proteins that facilitate cell adhesion.
- Trypsin: This protease is the most common enzymatic method, often used in combination with the chelating agent EDTA. It cleaves peptides after lysine or arginine residues, effectively degrading adhesion proteins and the ECM [1] [13]. Its drawbacks include potential damage to cell surface receptors, which can alter cell phenotype and function [13].
- Accutase: This is a blend of proteolytic and collagenolytic enzymes, widely considered a milder alternative to trypsin. However, recent studies show that it can compromise specific surface proteins, such as Fas ligands (FasL) and Fas receptors, cleaving them into fragments and significantly reducing their surface levels as measured by flow cytometry. This effect is reversible, but requires up to 20 hours for protein recovery [5].
- Collagenase: This enzyme specifically targets collagen, a major ECM component, and is often used for tissues or cell types with a dense collagenous matrix [13].
Non-Enzymatic Chemical Methods:
- Chelating Agents (e.g., EDTA, EGTA): These agents bind to calcium ions ((Ca^{2+})) and, to a lesser extent, magnesium ions ((Mg^{2+})), which are essential for the function of cell adhesion molecules like cadherins and integrins. By removing these ions, chelators weaken cell-cell and cell-matrix interactions [1] [13]. While gentle, EDTA alone is often insufficient for strongly adherent cells and may require mechanical assistance, such as scraping [5].
Physical and Emerging Methods
Mechanical Detachment: This includes physical scraping or pipetting to dislodge cells. While simple and cost-effective, these methods can cause significant cell damage and rupture, leading to low viability and are not suitable for sensitive applications [5] [13].
Novel Electrochemical Method (MIT Approach): A recent innovation involves using a low-frequency alternating current on a conductive biocompatible polymer surface. This method disrupts the cell-surface interface electrochemically without enzymes. It reports detachment efficiency of 95% with cell viability exceeding 90%, offering a promising path for automation and large-scale biomanufacturing by reducing waste and avoiding animal-derived enzymes [4] [23].
Other Physical Stimuli: Research is also exploring light-, magnetic-, and ultrasound-based methods to induce cell detachment in a controlled manner, though these are less established [13].
The table below provides a quantitative comparison of the most common detachment methods.
Table 1: Quantitative Comparison of Common Cell Detachment Methods
| Detachment Method | Typical Incubation Time | Reported Cell Viability | Key Advantages | Key Limitations/Damage |
|---|---|---|---|---|
| Trypsin-EDTA [1] [13] | ~2-10 min | >90% (if optimized) | Fast, effective, low-cost, robust | Cleaves surface receptors (e.g., CD4, CD8); can induce apoptosis |
| Accutase [5] | 10 min - 1 h | High (exact % not specified) | Considered milder than trypsin | Compromises FasL & Fas; requires 20h recovery |
| Chelators (e.g., EDTA) [5] [13] | ~20-30 min | High | Gentle on surface proteins; non-enzymatic | Often ineffective for strongly adherent cells alone |
| Mechanical Scraping [5] | N/A | Can be low | Simple, no chemicals | High physical stress, can tear cells, low viability |
| Electrochemical (MIT) [4] [23] | "Within minutes" | >90% | Enzyme-free, high viability, automatable | Emerging technology, requires specialized surfaces |
Standardized and Experimental Protocols
Standard Protocol for Passaging Adherent Mammalian Cells
The following is a detailed protocol for subculturing adherent cells using enzymatic dissociation, as derived from standard laboratory practice [1].
- Assessment: Before passaging, monitor cells to ensure they are in the log phase of growth and have a viability greater than 90%. Do not allow cells to become over-confluent.
- Media Removal: Aspirate and discard the spent cell culture media from the culture vessel.
- Washing: Gently wash the cell layer with a balanced salt solution without calcium and magnesium (e.g., PBS). Use approximately 2 mL per 10 cm² of surface area. This step removes residual serum, which contains trypsin inhibitors.
- Dissociation Reagent Application: Add a pre-warmed dissociation reagent (e.g., trypsin, TrypLE) to the side of the vessel. Use enough to cover the cell layer (approx. 0.5 mL per 10 cm²). Gently rock the vessel to ensure complete coverage.
- Incubation: Incubate the vessel at room temperature for approximately 2 minutes. The exact time varies by cell line.
- Monitoring: Observe cells under a microscope. If less than 90% of cells are detached, increase incubation time in 30-second increments, tapping the vessel gently to expedite detachment.
- Neutralization: When most cells are detached, add a volume of pre-warmed complete growth medium equivalent to at least twice the volume of the dissociation reagent used. Pipette the medium over the cell layer surface to disperse the cells and neutralize the enzyme.
- Centrifugation: Transfer the cell suspension to a centrifuge tube and pellet the cells at 200 x g for 5–10 minutes.
- Resuspension and Seeding: Resuspend the cell pellet in fresh, pre-warmed complete growth medium. Count the cells using a hemocytometer or automated cell counter, then dilute the suspension to the recommended seeding density and pipette into new culture vessels.
The workflow for this standard protocol, as well as for the novel electrochemical method, is summarized in the following diagram.
Experimental Analysis of Detachment Effects on Surface Proteins
Objective: To evaluate the impact of different detachment agents on the surface expression of Fas Ligand (FasL) and Fas receptor on macrophages (e.g., RAW264.7 cells) using flow cytometry [5].
Materials:
- Adherent macrophage cell line.
- Detachment solutions: Accutase, EDTA-based non-enzymatic solution (e.g., Versene).
- Complete growth medium.
- Phosphate-buffered saline (PBS).
- Antibodies for flow cytometry: Anti-FasL, Anti-Fas, and an isotype control.
- Flow cytometer.
Method:
- Cell Culture: Grow cells to approximately 80% confluency.
- Detachment: For each experimental group, perform the following:
- Aspirate media and wash with PBS.
- Add the recommended volume of Accutase or EDTA-based solution.
- Incubate for 10 minutes or 30 minutes at room temperature.
- For a control group, detach cells using a cell scraper.
- Neutralization and Collection: Neutralize enzymatic action with complete medium. Gently pipette to collect cells. Avoid vigorous pipetting.
- Cell Processing: Centrifuge cells, wash with PBS, and resuspend in FACS buffer.
- Staining: Aliquot cells and incubate with fluorescently-labeled antibodies against FasL and Fas (and appropriate isotype controls) for 30-60 minutes on ice or in the dark.
- Analysis: Wash cells to remove unbound antibody, resuspend in FACS buffer, and analyze on a flow cytometer. Compare the Mean Fluorescence Intensity (MFI) of FasL and Fas between the Accutase-treated and EDTA-treated groups.
Expected Results: Cells detached with Accutase will show a significant decrease (p < 0.001) in the MFI of surface FasL and Fas compared to cells detached with the EDTA-based solution or by scraping, indicating cleavage of these specific surface proteins [5].
Quantitative Data and Research Reagents
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for Cell Detachment
| Reagent / Solution | Primary Function | Key Considerations for Use |
|---|---|---|
| Trypsin-EDTA [1] [13] | Proteolytic enzyme cleaves adhesion proteins; EDTA chelates Ca²⁺/Mg²⁺. | Concentration and incubation time must be optimized per cell line to minimize surface protein damage. |
| Accutase [5] | Blend of enzymes for gentle dissociation. | Not universally "gentle"; can cleave specific proteins like FasL. Requires recovery time for surface protein re-expression. |
| Non-Enzymatic Dissociation Buffer (e.g., Versene) [5] | Chelates Ca²⁺/Mg²⁺ to disrupt integrin-mediated adhesion. | Ideal for preserving surface antigens for flow cytometry, but may be ineffective for some strongly adherent cells. |
| TrypLE [1] | A recombinant fungal trypsin-like enzyme. | A non-animal-derived alternative to trypsin, offering consistent performance and reduced risk of contamination. |
| Chelate-Free Solutions [13] | Often use cationic salts to disrupt electrostatic interactions. | Simple and avoid chelators, but may be less effective and require optimization for each application. |
| Collagenase [13] | Degrades native collagen in the ECM. | Essential for dissociating tissues or cells embedded in a collagen-rich matrix. |
Quantitative Analysis of Adhesion Strength
Single-cell force spectroscopy (SCFS) and FluidFM technology allow for the precise measurement of cell detachment forces across different stages of adhesion. A comparative study highlights the dramatic increase in adhesion strength over time [19].
Table 3: Quantified Detachment Forces and Energies in Early vs. Mature Adhesion
| Adhesion Phase | Contact Time | Maximum Detachment Force (MDF) | Detachment Energy | Dominant Interaction Type |
|---|---|---|---|---|
| Initial Adhesion [19] | 5 - 30 seconds | 0.5 - 4 nN | 1 - 40 fJ (femtojoules) | Non-specific electrostatic forces |
| Mature Adhesion [19] | 1 - 3 days | ~600 nN | ~10 pJ (picojoules) | Specific, protein-mediated bonds (e.g., integrin-ECM) |
This data demonstrates that adhesion forces can increase by approximately 150-fold as the contact matures from initial, non-specific attachment to the formation of specific focal adhesions [19]. This has critical implications for detachment protocols, as mature cultures require much harsher conditions to harvest, which inherently increases the risk of cellular damage.
Cell detachment is a critical, yet complex, step that bridges routine cell culture and downstream applications. The choice of detachment method is a significant variable that directly influences cell viability, phenotype, surface protein integrity, and consequently, experimental outcomes. While enzymatic methods like trypsin are robust and widely used, evidence of their detrimental effects on cells is growing [5] [13]. Non-enzymatic chemical methods are gentler but may lack efficiency.
The future of adherent cell detachment research is moving towards precise, controllable, and non-invasive technologies. The emergence of novel approaches, such as the electrochemical platform from MIT, points to a promising direction for automating biomanufacturing workflows for cell therapies and tissue engineering, reducing waste, and improving reproducibility [4] [23]. Furthermore, the development of smart biomaterials, such as microcarriers with stimuli-responsive coatings that release cells upon application of a specific trigger (temperature, light, pH), is a rapidly advancing field aimed at solving the scalability challenges in industrial cell manufacturing [13]. As the demand for high-quality cells in research and therapy continues to grow, the development and adoption of advanced, gentle detachment strategies will remain a central focus in biomedical science.
The process of adherent cell detachment is a fundamental yet critical procedure in biomedical research, industrial biomanufacturing, and therapeutic development. Adherent cells require physical attachment to solid surfaces for survival, growth, and reproduction, making detachment an essential step for cell passaging, harvesting for experiments, and production of cell-based therapies [4]. However, this necessary process inherently challenges cell integrity, creating a delicate balance between efficient cell release and preservation of cellular health. The transition from an attached to suspended state represents a period of significant cellular stress, potentially triggering adverse responses ranging from compromised viability to initiation of programmed cell death pathways [13].
Understanding the impact of detachment techniques on cell health is paramount for researchers and drug development professionals seeking to generate reliable, reproducible data and develop effective cellular products. Traditional enzymatic methods, while effective for detachment, can induce profound stresses through damage to delicate cell membranes and surface proteins, particularly in sensitive primary cells [4]. Even milder dissociation agents previously considered gentle can compromise specific surface markers and require substantial recovery periods, thereby influencing experimental outcomes [5]. As the field advances toward more sophisticated applications in regenerative medicine, cell therapy, and large-scale biomanufacturing, the development and selection of detachment strategies that minimize detrimental impacts on cell health have become increasingly crucial [4] [13].
Core Mechanisms: How Detachment Methods Influence Cellular Fate
Molecular Interactions During Detachment
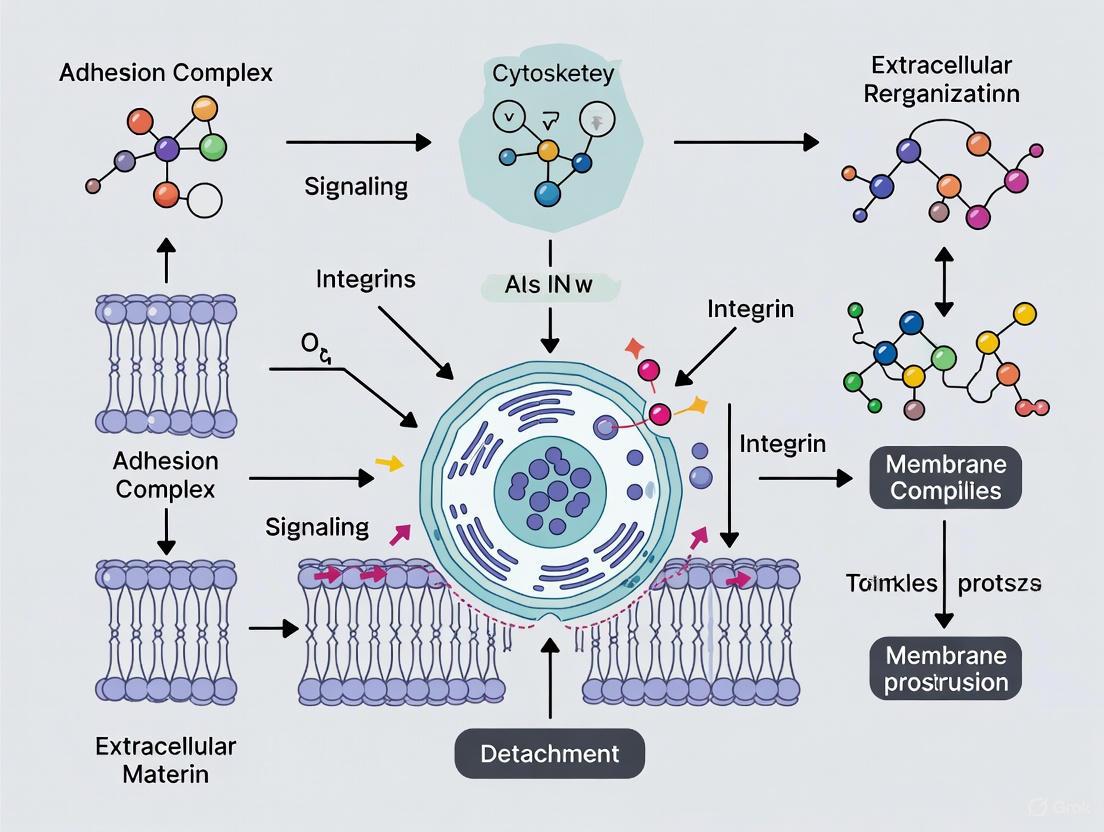
Cell adhesion to surfaces occurs through complex molecular interactions involving integrins, cadherins, and other adhesion receptors that connect the extracellular matrix (ECM) to the intracellular cytoskeleton [13]. These adhesion complexes not only provide physical attachment but also mediate critical survival signals. Disruption of these connections, which is the fundamental goal of cell detachment, inevitably interferes with these signaling pathways. The method of disruption directly influences the cellular response, determining whether cells remain viable and functional or enter states of stress, senescence, or apoptosis [13].
The extracellular matrix (ECM) forms a 3D fibrous network of proteins, proteoglycans, glycosaminoglycans, and metalloproteinases that serves as the foundation for cell attachment [13]. Cell-matrix adhesion occurs primarily through specialized structures such as focal adhesions, which involve integrin proteins that transmit both mechanical and chemical signals across the cell membrane. During detachment, the method used to disrupt these connections varies significantly:
- Enzymatic methods (e.g., trypsin, accutase) cleave specific peptide bonds in adhesion proteins [5] [13]
- Chelating agents (e.g., EDTA) remove divalent cations like calcium that are essential for integrin function [5] [13]
- Physical methods (e.g., bubbling, scraping) apply mechanical forces to break adhesions [4] [24]
- Stimuli-responsive surfaces (e.g., electrochemical, thermal) modify surface properties to reduce adhesion strength [4] [13]
Detachment-Induced Signaling Pathways
The disruption of adhesion complexes during detachment initiates intracellular signaling cascades that can ultimately influence cell fate. The diagram below illustrates key pathways through which detachment methods impact cell health, particularly through anoikis (a form of apoptosis triggered by detachment from the ECM) and membrane integrity damage.
The relationship between detachment mechanisms and cellular outcomes reveals that different approaches trigger distinct stress pathways. Enzymatic methods primarily cause surface protein damage, which can directly initiate apoptosis, while methods that disrupt adhesion complexes often trigger anoikis. Physical approaches like scraping may more directly compromise membrane integrity, leading to immediate viability loss [5] [13].
Comparative Analysis of Detachment Techniques and Their Impacts
Enzymatic Detachment Methods
Enzymatic approaches represent the most widely used detachment strategy in research and industrial applications. Trypsin, a protease that cleaves after lysine or arginine residues, effectively degrades most cell surface proteins and extracellular matrix components but causes significant damage to cell membranes and surface markers [4] [13]. This damage can extend beyond immediate viability reduction to longer-term effects including dysregulation of protein expression, enhanced apoptotic cell death, and altered metabolic pathways [13].
Accutase, often considered a milder enzymatic alternative, demonstrates reduced impact on many surface markers but shows specific vulnerability for certain proteins. Research has revealed that accutase significantly decreases surface levels of Fas ligands (FasL) and Fas receptors, cleaving the extracellular portion of FasL into fragments under 20 kD in size [5]. This specific protein damage required approximately 20 hours for recovery after accutase treatment, during which surface levels of these proteins gradually returned to normal [5]. Despite this specific damage, accutase demonstrated superior performance in maintaining overall cell viability compared to EDTA-based solutions or PBS buffers, particularly with extended exposure times up to 90 minutes [5].
Non-Enzymatic and Novel Detachment Strategies
Non-enzymatic methods offer alternatives that avoid proteolytic damage to cell surfaces. Chelating agents like EDTA function by removing calcium ions essential for integrin-mediated adhesion, providing a mild detachment approach but often requiring mechanical assistance for strongly adherent cells [5] [13]. Mechanical scraping, while effective, frequently causes membrane damage and cell tearing, reducing overall viability and increasing cellular debris [5].
Recent innovations have introduced advanced detachment strategies seeking to overcome limitations of traditional methods. MIT researchers have developed an electrochemical approach using alternating current on a conductive biocompatible polymer nanocomposite surface [4]. This method disrupts adhesion within minutes while maintaining over 90% cell viability, addressing key limitations of enzymatic and mechanical methods [4]. Another novel system utilizes electrochemically generated bubbles to create shear stress at the cell-surface interface, effectively removing cells without generating harmful bleach byproducts that can damage sensitive cells [24]. This technique has proven effective across multiple cell types, including algae, ovarian cancer cells, and bone cells, demonstrating its broad applicability [24].
Table 1: Quantitative Comparison of Cell Detachment Methods
| Method | Detachment Efficiency | Cell Viability | Impact on Surface Proteins | Recovery Time Required | Scalability |
|---|---|---|---|---|---|
| Trypsin | High (≈95%) [13] | Moderate (varies by cell type) [13] | Severe damage [4] [13] | 4-24 hours [13] | High [13] |
| Accutase | High (≈90%) [5] | High (>90%) [5] | Selective damage (e.g., FasL, Fas) [5] | ~20 hours [5] | Moderate [5] |
| EDTA | Low to Moderate [5] | High [5] | Minimal [5] | Minimal [5] | Low [13] |
| Scraping | High [5] | Low to Moderate [5] [25] | Physical damage [5] | Variable [5] | Low [13] |
| Electrochemical | High (95%) [4] | High (>90%) [4] | Minimal reported [4] | Minimal reported [4] | High [4] [24] |
Assessing Detachment Impacts: Viability and Apoptosis Methodologies
Technical Protocols for Cell Health Assessment
Accurately evaluating the impact of detachment methods on cell health requires robust assessment techniques. The following experimental protocols provide methodologies for quantifying cell viability and apoptosis following detachment procedures.
Modified EB/AO Apoptosis Assay in 96-Well Format
The Ethidium Bromide/Acridine Orange (EB/AO) staining method enables simultaneous quantification of live, apoptotic, and necrotic cell populations, making it particularly valuable for assessing detachment impacts [25]. This modified approach eliminates detaching and washing steps for adherent cells, minimizing additional damage and preserving fragile cell populations.
Materials and Equipment:
- Cell culture with appropriate detachment agent
- Ethidium bromide solution (100 μg/mL)
- Acridine orange solution (100 μg/mL)
- 96-well plate (preferably glass-bottom for optimal optical clarity)
- Centrifuge with 96-well plate carriers
- Fluorescence microscope with appropriate filters
Experimental Procedure:
- Cell Preparation: Following detachment by the test method, transfer cell suspension to 96-well plate (≈10,000-50,000 cells per well).
- Centrifugation: Centrifuge plate at 300×g for 5 minutes to sediment all cells, including floaters, to the well bottom.
- Staining: Add EB/AO working solution (1:1 mixture of EB and AO) directly to wells without removing supernatant.
- Incubation: Incubate for 5-10 minutes at room temperature protected from light.
- Immediate Analysis: Visualize using fluorescence microscope without washing steps:
- Live cells: Normal green nucleus with organized structure
- Early apoptotic: Bright green nucleus with condensed or fragmented chromatin
- Late apoptotic: Condensed and fragmented orange chromatin
- Necrotic: Structurally normal orange nucleus [25]
This method's advantage lies in its ability to maintain adherent cells in their cultured state, avoiding additional detachment stress that could alter apoptotic population distributions [25].
Annexin V/Propidium Iodide Flow Cytometry Protocol
The Annexin V/propidium iodide (PI) binding assay represents another widely used approach for detecting apoptosis and viability following cell detachment.
Materials and Equipment:
- Detached cell suspension
- Annexin V binding buffer (containing calcium ions)
- Fluorescently-labeled Annexin V probe
- Propidium iodide (PI) solution
- Flow cytometry staining buffer
- Benchtop centrifuge
- Flow cytometer
Experimental Procedure:
- Cell Preparation: Detach cells using test method and transfer 1×10⁶ cells per condition to microcentrifuge tubes.
- Washing: Pellet cells by centrifugation (300×g, 5 minutes), resuspend in Annexin binding buffer.
- Staining: Add fluorescent Annexin V probe, incubate 15 minutes at room temperature protected from light.
- Viability Staining: Add PI (final concentration 0.5-1.0 μg/mL) and incubate 5-20 minutes at room temperature.
- Analysis: Analyze by flow cytometry without additional washing:
- Annexin V⁻/PI⁻: Viable, non-apoptotic cells
- Annexin V⁺/PI⁻: Early apoptotic cells
- Annexin V⁺/PI⁺: Late apoptotic or necrotic cells [26]
Critical Considerations:
- Avoid chelating agents (EDTA, EGTA) in buffers as Annexin V requires calcium for phospholipid binding [26]
- Include controls: unstained, single stains, and apoptosis-induced positive control
- Process samples immediately after detachment for accurate assessment
Experimental Workflow for Detachment Method Comparison
The comprehensive evaluation of detachment methods requires a systematic approach comparing multiple cell health parameters. The following diagram illustrates an integrated workflow for assessing detachment impacts from initial processing through final analysis.
This comprehensive workflow enables researchers to systematically evaluate how different detachment methods affect immediate cell health, recovery trajectory, and long-term functionality. The time-course analysis is particularly important, as some surface protein damage requires extended recovery periods up to 20 hours [5].
The Research Toolkit: Essential Reagents and Technologies
Table 2: Research Reagent Solutions for Cell Detachment and Analysis
| Reagent/Category | Function & Mechanism | Key Applications | Considerations & Limitations |
|---|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme cleaves adhesion proteins; EDTA chelates calcium [13] | Routine cell culture, robust cell lines [13] | Damages surface proteins, requires serum inhibition [4] [13] |
| Accutase | Enzyme mixture with proteolytic and collagenolytic activity [5] | Sensitive cells, flow cytometry [5] [27] | Cleaves specific markers (FasL, Fas); requires recovery time [5] |
| Non-Enzymatic Dissociation Buffers | Chelate calcium/magnesium ions to disrupt cadherins and integrins [5] [13] | Surface marker preservation, sensitive applications [5] | Less effective for strongly adherent cells [5] |
| Annexin V Binding Buffers | Provide calcium-enriched environment for phosphatidylserine binding [26] | Apoptosis detection post-detachment [26] | Incompatible with EDTA; requires immediate analysis [26] |
| Viability Dyes (PI, EB, AO) | DNA intercalating agents distinguish membrane integrity [25] [26] | Viability assessment, apoptosis staging [25] | Varying specificity; requires appropriate controls [25] |
| Electrochemical Systems | Alternating current disrupts adhesion; bubble generation creates shear [4] [24] | High-value cells, automation-compatible processes [4] | Specialized equipment required; optimization needed [4] |
The impact of cell detachment on cellular health represents a critical consideration in experimental design and therapeutic development. Traditional enzymatic methods, while effective for cell release, impose significant stresses through surface protein damage, membrane disruption, and induction of apoptotic pathways [4] [5] [13]. The emerging recognition that even "gentle" enzymatic methods like accutase cause specific protein damage requiring extended recovery periods underscores the necessity of carefully matching detachment strategies to experimental goals [5].
Future directions in detachment research focus on developing enzyme-free systems that minimize cellular damage while enabling automation and scalability. Electrochemical approaches achieving over 90% viability and 95% detachment efficiency demonstrate the potential for integrated systems that maintain cell health throughout culture processes [4] [24]. As the cell dissociation market progresses—projected to grow from USD 455.03 million in 2025 to USD 1621.47 million by 2035—increased investment in innovative technologies addressing these challenges is anticipated [28]. For researchers and drug development professionals, selecting appropriate detachment methods and assessment protocols remains fundamental to generating reliable data and developing effective cellular therapies where preservation of cellular integrity is paramount.
A Guide to Cell Detachment Methods: From Classic Enzymatic to Novel Technologies
The culture of adherent cells is a cornerstone of biomedical research, drug development, and cell therapy. These cells require physical attachment to a solid surface to survive, grow, and reproduce [4]. A critical, yet challenging, step in working with these cells is the process of detachment and dissociation—releasing them from the culture surface while maintaining cellular integrity and function. Enzymatic dissociation remains one of the most robust and frequently used methods for harvesting adherent cells [13]. These methods function by selectively breaking down the proteins that facilitate cell adhesion to the culture substrate and the connections between adjacent cells.
The importance of choosing an appropriate detachment method cannot be overstated. Traditional techniques, while effective, can compromise cell viability by damaging delicate cell membranes and degrading crucial surface proteins, which in turn can alter cellular function and skew experimental results [29] [4] [13]. This is particularly critical for applications in regenerative medicine and cell therapy, where the preservation of cell health and function is paramount. This guide provides an in-depth technical examination of four key enzymatic agents—Trypsin, TrypLE, Accutase, and Collagenase—detailing their mechanisms, optimal use cases, and practical protocols to inform the work of researchers and drug development professionals.
Enzyme Mechanisms and Characteristics
Enzymatic detachment agents work by cleaving specific proteins involved in cell adhesion. The extracellular matrix (ECM) and cell-surface proteins like cadherins and integrins mediate the strong attachment of cells to the culture surface; these proteins often require calcium ions to maintain their adhesive function [30] [13]. Enzymes disrupt this adhesion through proteolytic activity.
The table below summarizes the core characteristics and primary applications of each enzyme.
Table 1: Key Characteristics of Enzymatic Cell Dissociation Agents
| Enzyme | Mechanism of Action | Key Characteristics | Primary Cell Type Applications |
|---|---|---|---|
| Trypsin | Cleaves peptide bonds after lysine or arginine residues; often used with EDTA to chelate calcium [29] [30] [13]. | Robust, cost-effective; can damage many surface proteins and boost apoptotic cell death [29] [13]. | Strongly adherent cell lines; high-density cultures (with collagenase) [30]. |
| TrypLE | A recombinant fungal protease with trypsin-like activity, cleaving after lysine and arginine [30]. | Animal-origin-free, consistent; gentler on cells than trypsin; direct protocol substitute for trypsin [30] [31]. | Strongly adherent cells; applications requiring animal-origin-free reagents [30]. |
| Accutase | A blend of collagenolytic and proteolytic enzymes, including trypsin-like protease XIV and thermolysin [29] [32]. | Ready-to-use, gentle; preserves cell surface antigens and viability; effective for delicate cells [29] [32]. | Pluripotent stem cells, neuronal cells, macrophages, and other sensitive primary cells [29] [32]. |
| Collagenase | Targets and degrades native collagen, a major component of the ECM and some tissues [30]. | Essential for breaking down structural collagen; often used in combination with other enzymes [30]. | Primary tissues rich in collagen; compact tissues [30]. |
Impact on Cell Surface Proteins and Recovery
A critical consideration when selecting a dissociation enzyme is its effect on cell surface markers. Research has demonstrated that even enzymes marketed as "gentle" can have significant, though often reversible, impacts.
A 2022 study revealed that Accutase can significantly decrease the surface levels of Fas ligands (FasL) and Fas receptors on macrophages compared to EDTA-based non-enzymatic detachment. The enzyme was found to cleave the extracellular region of FasL into small fragments. Fortunately, this effect was not permanent; the surface expression of these proteins required up to 20 hours to fully recover after accutase treatment. Notably, the surface levels of other markers, like F4/80 on murine macrophages, were not altered, indicating the effect is protein-specific [29].
This underscores the importance of allowing adequate recovery time after cell detachment and before proceeding with experiments where surface marker integrity is crucial, such as flow cytometry or functional immune assays.
Quantitative Data and Comparison
To aid in evidence-based reagent selection, the following table synthesizes quantitative data on enzyme performance from the literature.
Table 2: Quantitative Performance Comparison of Detachment Enzymes
| Enzyme | Typical Concentration | Incubation Time (at 37°C) | Reported Cell Viability | Key Experimental Findings |
|---|---|---|---|---|
| Trypsin | 0.25% (for tissue) [30] | 5-15 min (cells) [30]; 20-30 min (after cold incubation for tissue) [30] | >90% (standard protocol) [30] | Can dysregulate protein expression and enhance oncogene expression [13]. |
| TrypLE | Volume to cover monolayer (e.g., 5 mL/75 cm²) [30] | Until detachment observed [30] | >90% (standard protocol) [30] | 91% detachment efficiency of LNCaP cells from a specialized surface within 10 minutes [31]. |
| Accutase | Ready-to-use solution [32] | 10 min to 1 hour [29] | >90% [32] | Maintained significantly higher cell viability after 60 and 90 min treatment vs. EDTA [29]. Compromises surface FasL/Fas; requires 20h recovery [29]. |
| Collagenase | 50-200 U/mL [30] | 4-18 hours [30] | Varies by tissue and time | Effective for disaggregating whole tissue; often used with other enzymes like Dispase [30]. |
Detailed Experimental Protocols
Below are standardized protocols for the enzymatic dissociation of adherent cell monolayers and primary tissues. Always perform these procedures under sterile conditions.
General Enzymatic Dissociation for Adherent Cell Monolayers
This procedure is a template for Trypsin, TrypLE, and Accutase. Optimal conditions should be determined empirically for each cell line [30].
- Aspiration: Remove and discard the spent cell culture media from the flask or dish.
- Rinse: Wash the cell monolayer using a balanced salt solution without calcium and magnesium (e.g., DPBS). Add the wash solution to the side of the vessel opposite the cells, rock gently for 1-2 minutes, and aspirate the solution. This step removes residual calcium and serum that would inhibit the enzyme.
- Enzyme Application: Add the pre-warmed dissociation solution to the vessel, ensuring complete coverage of the cell monolayer. Use approximately 2-3 mL per 25 cm² of surface area [30].
- Incubation: Incubate the vessel at 37°C. Rock the flask gently periodically. Monitor the cells under an inverted microscope. Most cells detach within 5-15 minutes, but this can vary.
- Neutralization: When cells are completely detached, add complete growth medium (containing serum) to the flask. For serum-free workflows, use an inhibitor like soybean trypsin inhibitor (note: not recommended for TrypLE [30]). Gently pipette the suspension to disperse any clumps.
- Centrifugation: Transfer the cell suspension to a conical tube and centrifuge at approximately 100-200 × g for 5-10 minutes.
- Resuspension: Discard the supernatant and resuspend the cell pellet in fresh, pre-warmed complete medium.
- Counting and Seeding: Determine viable cell density and percent viability using an automated cell counter or hemocytometer. Seed, incubate, and subculture according to your specific protocol [30].
Primary Tissue Dissociation Protocols
Trypsin-Based Tissue Disaggregation
This method is effective for many tissues but requires careful timing [30].
- Mincing: After dissection, mince the tissue into 3-4 mm pieces with sterile instruments.
- Washing: Wash the tissue pieces several times in a balanced salt solution without calcium and magnesium, allowing pieces to settle between washes.
- Cold Penetration: Place the container on ice, remove supernatant, and add 0.25% trypsin solution (1 mL per 100 mg tissue). Incubate at 4°C for 6-18 hours to allow enzyme penetration.
- Enzyme Activation: Decant and discard the trypsin. Incubate the tissue pieces with the residual trypsin at 37°C for 20-30 minutes.
- Dispersion: Add warm complete media and disperse the tissue by gentle pipetting. For serum-free media, add a soybean trypsin inhibitor.
- Filtration and Seeding: Filter the cell suspension through a sterile 100-200 µm mesh, count cells, and seed for culture [30].
Collagenase-Based Tissue Disaggregation
Ideal for tissues rich in structural collagen, such as connective tissues [30].
- Mincing and Washing: Mince tissue into 3-4 mm pieces and wash several times with HBSS containing calcium and magnesium.
- Enzyme Application: Submerge the tissue in HBSS with calcium and magnesium and add collagenase to a final concentration of 50-200 U/mL. Supplementing with 3 mM CaCl₂ can increase efficiency.
- Digestion: Incubate at 37°C for 4-18 hours on a rocker platform.
- Dispersion and Washing: Disperse the cells by passing through a sterile mesh. Wash the dispersed cells several times by centrifugation in HBSS without collagenase.
- Seeding: Resuspend the final cell pellet in culture medium, determine viable cell density, and seed the cells [30].
The Scientist's Toolkit: Essential Reagent Solutions
Table 3: Key Reagents for Enzymatic Cell Dissociation Workflows
| Reagent / Material | Function in the Protocol |
|---|---|
| DPBS (without Ca2+/Mg2+) | A balanced salt solution used to rinse the cell monolayer prior to dissociation, removing inhibitory divalent cations and serum [30]. |
| Trypsin-EDTA | A classic, potent enzymatic cocktail for detaching strongly adherent cell lines. EDTA chelates calcium, weakening integrin-mediated adhesion [30] [13]. |
| TrypLE Express | A recombinant, animal-origin-free enzyme that functions as a direct substitute for trypsin in existing protocols, offering greater consistency and gentler action [30]. |
| Accutase | A gentle, ready-to-use blended enzyme solution ideal for dissociating sensitive cells like stem cells and neurons while preserving surface markers [29] [32]. |
| Cell Dissociation Buffer | A non-enzymatic, EDTA-based solution used for lightly adherent cells or when the integrity of all surface proteins is critical [30]. |
| Soybean Trypsin Inhibitor | Used to quench trypsin activity after cell detachment in serum-free cell culture workflows [30]. |
| Complete Growth Medium | Used to neutralize enzymatic activity after detachment (via serum) and to resuspend the cell pellet for counting and seeding [30]. |
Visualizing Workflows and Mechanisms
Enzymatic Cell Detachment Mechanism
This diagram illustrates the general mechanism by which enzymatic and non-enzymatic agents facilitate cell detachment.
Standardized Cell Detachment Experimental Workflow
This flowchart outlines the key steps in a general enzymatic cell detachment protocol.
Enzymatic methods using Trypsin, TrypLE, Accutase, and Collagenase provide a powerful and versatile toolkit for harvesting adherent cells. The choice of enzyme is a critical experimental parameter that balances detachment efficiency against the preservation of cell viability, surface markers, and functionality. While enzymatic methods are currently the most robust, they are not without their limitations, including the potential degradation of surface proteins and the introduction of animal-derived components [4] [13].
The field of adherent cell detachment research is actively evolving to address these challenges. Future directions focus on developing non-enzymatic, stimuli-responsive strategies that offer greater precision and reduce cellular damage. Promising approaches include:
- Electrochemical Detachment: Novel platforms using alternating electrochemical current on conductive polymer surfaces can disrupt adhesion within minutes while maintaining over 90% cell viability, presenting a potential pathway for automated biomanufacturing [4].
- Advanced Microfluidic Systems: Sensor-based microfluidic extraction systems can detect a cell's "pre-detachment moment" by monitoring its oscillatory response to hydrodynamic forces. This allows for dynamic adjustment of suction parameters, enabling gentle, non-invasive extraction of single cells with minimal stress [33].
- Smart Microcarriers: For large-scale bioreactor cultures, the development of microcarriers with stimuli-responsive coatings (e.g., thermoresponsive polymers) allows for cell harvesting without enzymes, which is crucial for the expanding cell manufacturing industry [13].
As research progresses, the integration of these advanced, controlled detachment technologies will be essential for improving the reproducibility, scalability, and quality of cells used in therapeutic applications and sophisticated in vitro models.
The research and development of adherent cell detachment techniques are crucial for advancing biomedical applications, including cell therapy, regenerative medicine, and drug development. Within this field, non-enzymatic chemical methods represent a growing area of interest aimed at overcoming the limitations of enzymatic approaches. Ethylenediaminetetraacetic acid (EDTA) stands as a principal chelating agent used to facilitate cell detachment by targeting the divalent cations that are essential for cell adhesion processes [34] [35]. Unlike proteolytic enzymes such as trypsin, which digest adhesion proteins, EDTA operates through a non-proteolytic mechanism that minimizes damage to critical cell surface markers and receptors [13]. This technical guide provides an in-depth examination of the role, mechanism, and application of EDTA and analogous chelating agents within the context of modern cell culture and bioprocessing, highlighting their strategic importance in the broader landscape of adherent cell detachment research.
The Fundamental Role of Cell Adhesion and Its Reversal
To understand the mechanism of chelating agents, one must first appreciate the biological process of cell adhesion. Adherent cells attach to the extracellular matrix (ECM) and their neighbors through specialized junctions and adhesion complexes. Cell-matrix adhesion,
particularly through focal adhesions, is heavily dependent on transmembrane proteins called integrins [13]. These integrins require divalent cations, notably calcium (Ca²⁺) and magnesium (Mg²⁺), to stabilize their conformation and facilitate their binding to specific sequences (e.g., RGD motifs) in ECM proteins like fibronectin and laminin [34] [13]. These cation-dependent protein-protein and protein-carbohydrate interactions form the foundation of adherent cell culture. The process of cell detachment, therefore, necessitates the disruption of these stable adhesion complexes. While enzymatic methods cleave the proteins themselves, non-enzymatic chemical methods aim to disrupt the ionic environment that is critical for their stability, offering a gentler alternative for cell harvesting.
EDTA: The Paradigmatic Chelating Agent
Chemical Structure and Mechanism of Action
EDTA (C₁₀H₁₆N₂O₈) is an aminopolycarboxylic acid characterized by a structure that features multiple electron-donating oxygen atoms [35] [36]. This structure allows EDTA to act as a hexadentate ligand, meaning it can form six coordination bonds with a single metal ion [36]. Its primary mechanism in cell detachment is chelation—the formation of stable, water-soluble complexes with di- and trivalent metal ions.
In the context of cell culture, EDTA's binding to calcium (Ca²⁺) and magnesium (Mg²⁺) is paramount. By sequestering these ions from the cell culture environment, EDTA effectively:
- Destabilizes Integrin Binding: The binding of integrins to their ECM ligands is cation-dependent. Chelating these ions causes integrins to revert to a low-affinity state, weakening cell-substrate attachment [34] [13].
- Disrupts Cell-Cell Junctions: Calcium is essential for the formation and maintenance of adherence junctions and other intercellular connections. EDTA chelation thus promotes the dissociation of cell sheets into individual cells [34].
The following diagram illustrates this core mechanism of action.
Diagram 1: EDTA Chelation Mechanism in Cell Detachment
Synergy with Other Agents
While EDTA is effective alone for some lightly-adherent cell types, it is most famously used in synergy with proteolytic enzymes like trypsin [34] [30] [37]. In this combined approach, EDTA's role is to prime the system by chelating ions and weakening the overall adhesion structure, thereby allowing trypsin to more efficiently access and cleave the peptide bonds in the now-exposed adhesion proteins [34]. This synergy reduces the required concentration and exposure time to trypsin, mitigating its damaging effects. However, for applications where intact surface proteins are critical, EDTA can be used as a standalone, non-enzymatic dissociation agent.
Comparative Analysis of Cell Detachment Techniques
The selection of a detachment method is a critical experimental design choice. The table below summarizes key characteristics of EDTA-based and other prominent non-enzymatic detachment methods, highlighting their relative advantages and challenges.
Table 1: Comparison of Non-Enzymatic Cell Detachment Methods
| Method | Mechanism of Action | Typical Detachment Efficiency | Key Advantages | Primary Limitations |
|---|---|---|---|---|
| EDTA (Chelating Agent) | Sequesters Ca²⁺ & Mg²⁺ ions, destabilizing adhesion complexes [34] [13] | Varies by cell type; often high for loosely adherent cells [30] | Preserves surface proteins; simple protocol; low cost [13] | May be ineffective for strongly adherent cells; can affect cellular metabolism [13] [30] |
| Thermo-Responsive Polymers (e.g., PNIPAM) | Polymer hydration/swelling generates mechanical disjoining pressure below LCST (~32°C), pushing cells off [38] [13] [39] | High (>90%) for suitable cell-surface combinations [38] [39] | Enzyme-free; allows for harvest of intact cell sheets; high viability post-harvest [38] [13] | Requires specialized, expensive surfaces; precise control of polymer properties needed; slower detachment [13] |
| Electrochemical Redox | Applies alternating current to a conductive surface to disrupt the ionic microenvironment at the cell-surface interface [4] [23] | ~95% (reported for osteosarcoma and ovarian cancer cells) [4] [23] | High efficiency and viability (>90%); potential for full automation and scalability [4] [23] | Emerging technology; requires specialized instrumentation and conductive surfaces [4] |
| Mechanical Scraping | Physical force shears cells from the substrate [13] [30] | High, but with potential for selective detachment | Rapid; low cost; no chemical exposure [30] | Significant cell damage and death; not suitable for sensitive applications [13] |
Experimental Protocols for EDTA-Based Cell Detachment
Standard Protocol for Monolayer Dissociation with EDTA
The following step-by-step protocol is adapted from established cell culture baselines and is suitable for dissociating monolayer cultures using a commercial, non-enzymatic cell dissociation buffer, which typically contains EDTA as the active component [30].
Objective: To gently detach adherent cells from a culture vessel while maximizing viability and preserving cell surface proteins. Principle: Chelation of divalent cations to disrupt cell-substrate and cell-cell adhesions.
Reagents and Materials:
- Pre-warmed Cell Dissociation Buffer (e.g., Gibco, containing EDTA) or a solution of EDTA in a balanced salt solution [30].
- Pre-warmed (37°C) phosphate-buffered saline (PBS), without calcium and magnesium [30].
- Pre-warmed complete growth medium.
- T-75 culture flask or 100 mm dish containing the adherent cell monolayer.
- Centrifuge tubes, pipettes, and a cell counter.
Procedure:
- Preparation: Warm all reagents (PBS, Cell Dissociation Buffer, growth medium) to 37°C prior to use to minimize thermal shock to cells [30].
- Rinsing:
- Aspirate and discard the spent cell culture medium.
- Gently rinse the cell monolayer twice with 5-10 mL of pre-warmed PBS (without Ca²⁺ and Mg²⁺) to remove residual media containing divalent cations. Rock the vessel gently for 30-60 seconds during each rinse and completely aspirate the solution [30].
- Dissociation:
- Add approximately 5 mL of pre-warmed Cell Dissociation Buffer to the T-75 flask, ensuring it covers the entire cell layer.
- Gently rock the vessel at room temperature for 1-2 minutes. Aspirate and discard this initial volume (this step further ensures removal of residual cations).
- Add another 5 mL of fresh, pre-warmed Cell Dissociation Buffer.
- Tap the flask firmly against the palm of your hand to dislodge cells.
- Observe under an inverted microscope. If cells remain adherent, allow the flask to sit at room temperature for 2-5 more minutes, then tap again. Repeat as necessary for strongly adherent cells [30].
- Neutralization and Collection:
- Once the majority of cells are detached (appearing round and refractive under the microscope), add at least 5 mL of complete growth medium to neutralize the dissociation process.
- Pipette the cell suspension gently over the surface of the flask to ensure complete cell recovery. Avoid vigorous pipetting to maintain cell viability [30].
- Post-Harvest Processing:
- Transfer the cell suspension to a centrifuge tube and pellet the cells by centrifugation at 100 × g for 5-10 minutes.
- Discard the supernatant and resuspend the cell pellet in an appropriate volume of fresh, pre-warmed complete medium.
- Determine viable cell density and percent viability using an automated cell counter or manual counting with trypan blue exclusion [30].
The following workflow summarizes the key steps.
Diagram 2: EDTA-Based Cell Detachment Workflow
Critical Parameters for Optimization
- Cell Type: The efficacy of EDTA is highly cell-type dependent. Lightly adherent cells (e.g., some epithelial lines) may detach readily, while strongly adherent cells (e.g., fibroblasts) may require the addition of a low concentration of trypsin (e.g., 0.05% trypsin-EDTA) [30] [37].
- Incubation Time: Minimize the exposure time to EDTA. Prolonged incubation can lead to unintended effects on cell physiology and reduce viability [13] [37].
- Temperature: While the protocol can be performed at room temperature, performing the dissociation at 37°C can accelerate the process for more resistant cells [37].
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of EDTA-based detachment methods requires a set of specific reagents and materials. The following table details the key components of a research toolkit for this application.
Table 2: Essential Research Reagent Solutions for EDTA-Based Cell Detachment
| Reagent/Material | Function/Description | Example Usage Notes |
|---|---|---|
| Cell Dissociation Buffer | A ready-to-use, non-enzymatic solution, typically containing EDTA in a balanced salt solution. | Ideal for gentle dissociation; preserves sensitive cell surface epitopes for flow cytometry or immunotherapy applications [30]. |
| EDTA Solution | A stock solution of EDTA (e.g., 0.5 M, pH 8.0) that can be diluted in PBS or other calcium-free buffers. | Offers flexibility for customizing concentration and buffer conditions for specific experimental needs [34]. |
| PBS (without Ca²⁺ & Mg²⁺) | A balanced salt solution used for rinsing cells prior to dissociation. | Critical for removing serum and divalent cations from the culture medium that would otherwise inhibit EDTA's action [30] [37]. |
| Complete Growth Medium | Standard culture medium containing serum. | Used to neutralize the dissociation process, as serum proteins act as a natural chelator and stop the reaction [30]. |
| Soybean Trypsin Inhibitor | A specific inhibitor of trypsin. | Required for neutralizing trypsin-EDTA mixtures in serum-free cultures, where serum proteins are absent [30] [37]. |
EDTA remains a cornerstone reagent in the field of non-enzymatic cell detachment, valued for its simple, cost-effective, and non-destructive mechanism of action. Its role in basic research and industrial biomanufacturing is secure, particularly for applications where the integrity of the cell surface proteome is paramount. The ongoing research in adherent cell detachment is increasingly focused on developing integrated and scalable technologies that combine the gentleness of chemical methods with the precision and automation potential of physical stimuli [4] [13] [23]. While novel platforms like electrochemical redox-cycling and advanced smart polymers show immense promise for the future of cell therapy manufacturing, EDTA and other chelating agents continue to provide a fundamental, reliable, and well-understood tool for the scientist's arsenal. The choice of method ultimately depends on a careful balance between the requirements for cell viability, functionality, yield, and the practical constraints of scalability and cost.
Adherent cell detachment is a critical, yet challenging, step in cell culture processes for biopharmaceutical applications and basic biological research [40]. While enzymatic digestion, notably with trypsin, has been the historical standard, these methods inherently damage cell membrane proteins and extracellular matrix components, compromising viability and subsequent experimental results [41] [5]. Physical and mechanical detachment methods present compelling alternatives by leveraging direct force to dislodge cells, thereby avoiding enzymatic damage. These methods—including scraping, shaking, and the application of fluid shear forces—are indispensable for applications requiring high cell viability, intact surface proteins, or the harvest of cell sheets. This whitepaper provides an in-depth technical guide to these physical methods, detailing their principles, applications, and specific protocols to equip researchers with the knowledge to implement these techniques effectively.
Core Principles and Methodologies
Cell Scraping
Cell scraping is a fundamental mechanical method involving a physical spatula to dislodge cells from a culture surface [40].
- Mechanism of Action: The rubber or plastic blade of the scraper applies direct mechanical force to the cell monolayer, breaking the adhesive bonds between the cells and the substrate. This method is quick and avoids chemical reagents or complex surface modifications [40].
- Typical Applications: Suitable for laboratory-scale detachment from standard culture vessels like T-flasks. It is particularly useful when the subsequent use of the cells does not require high viability or when preserving cell-cell interactions in a sheet is necessary [40].
- Key Limitations: The procedure is disruptive and can result in significant cell death and damage. It is challenging to automate and is generally not feasible for industrial-scale processes or complex culture supports like microcarriers [40].
Experimental Protocol: Cell Scraping
- Preparation: Aspirate and discard the spent cell culture medium from the culture vessel [1].
- Wash: Gently wash the cell layer with a balanced salt solution without calcium and magnesium (e.g., DPBS) to remove any residual serum that can inhibit detachment [1].
- Scraping: Add a small volume of fresh, pre-warmed culture medium or a buffered solution to the vessel. Using a sterile, disposable cell scraper, firmly and evenly scrape the entire growth surface to dislodge the cells.
- Collection: Pipette the medium containing the detached cells into a sterile collection tube. Rinse the surface with additional medium to collect any remaining cells.
- Processing: The cell suspension can then be centrifuged, and the pellet resuspended for counting and subsequent passaging or experimentation [1].
Manual Shaking and Agitation
Shaking and agitation employ rhythmic motion to generate hydrodynamic forces that detach cells.
- Mechanism of Action: The motion of the culture vessel or the fluid within it creates shear stress at the cell-surface interface. When this force exceeds the cell's adhesive strength, detachment occurs. This method can be used alone for weakly adherent cells or in combination with mild chemical treatments for more robust adhesion [40].
- Advanced Application: Intermittent ultrasonic traveling waves represent a sophisticated form of this method. An ultrasonic transducer creates acoustic pressure and medium "sloshing" within a standard culture dish, enabling enzyme-free detachment [41].
Experimental Protocol: Intermittent Ultrasonic Traveling Waves
- System Setup: The system is composed of an ultrasonic transducer, a glass plate, and a piezoelectric ceramic ring to hold a standard cell culture dish. Glycerol is used as an acoustic couplant between the transducer and the dish [41].
- Cell Culture and Medium Switch: Culture cells (e.g., CHO cells) in serum-supplemented medium (SSM) to promote adhesion. Before detachment, switch to a serum-free medium (SFM) for a defined period (e.g., 42h SSM / 6h SFM), as the absence of adherence proteins facilitates release [41].
- Ultrasound Application: Activate the ultrasonic transducer with optimized parameters (e.g., input voltage of 2EA, frequency range of 29–31 kHz, modulation period of 0.02 s) for an exposure duration of 5 minutes [41].
- Cell Collection: Following ultrasound treatment, transfer the medium containing the detached cells to a collection tube for further use. This method has been shown to detach 96.2% of cells with less membrane damage and faster post-detachment adhesion compared to trypsinization [41].
Application of Controlled Shear Forces
The use of controlled shear forces in specialized devices provides a quantifiable and reproducible means to study cell adhesion strength and detach cells.
- Mechanism of Action: A unidirectional fluid flow is passed over adherent cells, inducing a lateral wall shear stress (τ). Cells detach when the applied hydrodynamic force exceeds the total adhesive force of the cells to the culture surface [42].
- Typical Device: A parallel plate flow chamber is a widely accepted microfluidic device for this purpose. It creates a single channel of fluid flow between two plates, with cells adhered to the bottom plate [42].
- Research Application: This method is powerful for quantifying differences in adhesive function between cell types. For instance, cancer cell lines with high metastatic potential (e.g., MDA-MB-231) demonstrate greater detachment under shear stress than their low-metastatic counterparts (e.g., MCF-7), providing a functional metric for cancer aggression [42].
Experimental Protocol: Parallel Plate Flow Chamber
- Cell Seeding: Seed and culture the cells of interest on a substrate that fits within the flow chamber (e.g., a glass slide coated with an extracellular matrix molecule) [42].
- Assembly: Assemble the flow chamber with the cell-coated substrate forming one wall of the flow channel.
- Perfusion: Connect the chamber to a perfusion system containing an appropriate buffer or medium. Precisely control the flow rate using a pump to generate the desired wall shear stress. Computational Fluid Dynamics (CFD) simulations are often used to characterize the relationship between flow rate and shear stress in the system [43].
- Observation and Quantification: Observe the cells under a microscope during perfusion. The percentage of cells detached at specific time points and shear stress levels (e.g., τ ≥ 100 × 10⁻⁵ N cm⁻² can cause detachment in the absence of proteolytic reagent) is quantified to assess adhesion strength [43] [42].
- Cell Collection: The effluent from the chamber can be collected to harvest the detached cells.
Quantitative Comparison of Physical Detachment Methods
The table below summarizes key performance metrics and characteristics of the primary physical and mechanical detachment methods.
Table 1: Quantitative Comparison of Physical and Mechanical Cell Detachment Methods
| Method | Detachment Efficiency | Impact on Viability/ Function | Key Quantitative Parameters | Spatial Resolution | Process Scalability |
|---|---|---|---|---|---|
| Cell Scraping | High (for accessible surfaces) | Low viability; causes physical damage [40] | N/A (manual force) | Low (whole surface) [40] | Low (laboratory scale) [40] |
| Shaking & Agitation | Variable (cell-line dependent) | Milder than scraping; can be combined with enzymes [40] | Shaking speed, duration, vessel geometry | Low (whole vessel) | Moderate (bioreactors) |
| Intermittent Ultrasound | 96.2% (CHO cells) [41] | High viability; preserves surface proteins & pseudopodia [41] | Input voltage (e.g., 2EA), frequency (29-31 kHz), exposure (5 min) [41] | Low (whole dish) | To be determined |
| Shear Force (Flow Chamber) | Quantifiable by % detached at set τ [42] | Can be harsh, causing damage or death [40] | Wall Shear Stress (τ); e.g., 8.2x10⁻⁵ to 100x10⁻⁵ N cm⁻² [43] [42] | High (controlled environment) [42] | Low (primarily analytical) |
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of physical detachment methods requires specific reagents and equipment. The following table details a core set of materials for the protocols described in this guide.
Table 2: Essential Research Reagent Solutions and Materials for Physical Detachment
| Item | Function/Application | Example Protocol Usage |
|---|---|---|
| Balanced Salt Solution (without Ca²⁺/Mg²⁺) | Washes cells pre-detachment; removes serum ions that inhibit detachment [1] [44]. | Standard step in scraping, shaking, and shear force protocols [1]. |
| Serum-Free Medium (SFM) | Medium without adherence proteins; used to facilitate cell release in acoustic detachment [41]. | Replacing SSM before ultrasonic wave application [41]. |
| Complete Growth Medium | Used to resuspend and dilute cells after detachment; contains nutrients and serum for cell health [1]. | Resuspending cell pellets after scraping or collection from flow chamber [1]. |
| Extracellular Matrix (ECM) Molecules | Coats culture surfaces to promote and standardize cell adhesion prior to detachment studies [44] [42]. | Coating slides for parallel plate flow chamber adhesion assays [42]. |
| Cell Scraper | Manual tool with a rubber or plastic blade for physically dislodging cells from a culture surface [40]. | Primary tool for the cell scraping protocol [40]. |
| Parallel Plate Flow Chamber | Microfluidic device that generates controlled, unidirectional fluid flow to apply quantifiable shear stress [42]. | Core device for the shear force detachment protocol [42]. |
| Ultrasonic Transducer System | Generates intermittent traveling waves for enzyme-free cell detachment via acoustic pressure and sloshing [41]. | Core device for the intermittent ultrasonic wave protocol [41]. |
Experimental Workflow and Decision Pathway
The following diagram illustrates a generalized experimental workflow for implementing and analyzing physical detachment methods, from initial preparation to data interpretation. It integrates the key protocols and decision points discussed in this guide.
Diagram 1: Experimental workflow for physical cell detachment.
Physical and mechanical methods for adherent cell detachment offer a versatile and powerful toolkit for modern cell biology and biopharmaceutical development. From the simple scraper to sophisticated ultrasonic and shear stress devices, these techniques enable researchers to overcome the limitations of enzymatic digestion. The choice of method must be guided by the experimental goals, whether for routine passaging, harvesting sensitive cells with high viability, or quantitatively probing the fundamental mechanisms of cell adhesion. As the field advances, further refinement and scaling of these physical methods, particularly ultrasound and controlled shear, will undoubtedly enhance their utility in producing the high-quality cell populations essential for therapeutic and diagnostic breakthroughs.
Adherent cell detachment is a critical, yet often disruptive, step in most cell culture protocols, particularly for applications in tissue engineering, regenerative medicine, and large-scale biomanufacturing where preserving cell viability, surface proteins, and extracellular matrix (ECM) is paramount [13]. Traditional enzymatic methods, such as trypsinization, are effective but inherently damaging, as they cleave anchoring proteins and cell surface receptors, which can dysregulate protein expression, boost apoptotic cell death, and impair cellular functions [13] [5]. The growing demand for high-quality cells in advanced therapies has driven the development of stimuli-responsive surfaces that enable cell attachment and on-demand detachment in response to external cues, thereby minimizing cellular damage and preserving complex cell sheets [13] [45]. This whitepaper reviews three advanced stimuli-responsive methods—thermo-responsive, pH-responsive, and electrochemical surfaces—framed within the broader research objectives of achieving precise, non-invasive, and scalable cell harvesting technologies.
Thermo-Responsive Surfaces
Mechanism of Action
Thermo-responsive cell detachment primarily utilizes polymers that undergo a reversible change in hydration state and conformation at a specific critical temperature. The most extensively studied polymer is poly(N-isopropylacrylamide) (pNIPAAm) [45]. Above its lower critical solution temperature (LCST) of approximately 32°C, pNIPAAm is hydrophobic and relatively collapsed, permitting cell adhesion and proliferation. When the temperature is reduced below the LCST, the polymer chain hydrates and expands, becoming hydrophilic. This switch creates a physical barrier that disrupts cell-substrate interactions, promoting the spontaneous detachment of intact cell sheets without the need for proteolytic enzymes [45]. This process preserves vital cell-cell junctions and the deposited ECM, which is crucial for the subsequent success of engineered tissues in applications such as cardiac repair and corneal regeneration [45].
Experimental Protocols
The foundation of thermo-responsive cell culture is the fabrication of modified surfaces, with electron beam (EB) polymerization being a widely used and reliable method.
- Surface Fabrication via EB Polymerization: Tissue culture polystyrene (TCPS) dishes are uniformly coated with a NIPAAm monomer solution in 2-propanol and irradiated with a 0.3 MGy electron beam. Post-irradiation, non-grafted monomers are rinsed off with deionized water, leaving a covalently grafted pNIPAAm layer [45].
- Critical Parameter - Film Thickness: The thickness of the grafted pNIPAAm layer is a critical determinant of success. Research indicates an optimal thickness between 15 nm and 20 nm [45]. Layers thinner than this may not facilitate effective detachment, while thicker layers (e.g., ~29 nm) can prevent cell adhesion altogether, even at 37°C [45].
- Cell Seeding and Detachment Protocol:
- Surface Preparation: Sterilize the pNIPAAm-grafted dishes under UV light.
- Cell Seeding: Seed adherent cells onto the surface and culture at a standard 37°C (above the LCST) until confluence is reached.
- Temperature Reduction: For cell sheet harvesting, reduce the culture temperature to around 20°C (below the LCST). This is typically achieved by moving the culture dish to a lower-temperature incubator or by exchanging the medium with cool, fresh medium.
- Harvesting: Cell sheet detachment is often complete within 30 minutes to 2 hours, after which the sheet can be gently pipetted or transferred for further processing [45].
Table 1: Fabrication Methods for Thermo-Responsive pNIPAAm Surfaces
| Fabrication Method | Underlying Substrate | Key Advantages | Key Limitations | Detachment Time |
|---|---|---|---|---|
| Electron Beam Polymerization | Tissue Culture Polystyrene (TCPS) | Robust, well-characterized, uniform grafting | Requires specialized, expensive equipment | ~1 hour at 20°C [45] |
| Plasma Polymerization | Silica, Glass, TCPS | Solvent-free, one-step vapor phase coating | Potential monomer fragmentation; film thickness less critical | ~2 hours at 20°C [45] |
| UV Irradiation | Polydimethylsiloxane (PDMS), Poly(ethylene terephthalate) | Uses common laboratory equipment | Potential for incomplete polymerization | ~30 minutes [45] |
| Spin-Coating | Glass, Si Wafer | Low-cost, no specialized equipment | Film dissolution can cause cytotoxicity; detachment in clumps | ~2 minutes at 20°C [45] |
Signaling Pathways in Thermo-Responsive Detachment
The following diagram illustrates the cellular signaling cascade triggered by the swelling of a thermo-responsive polymer, leading to cell detachment.
pH-Responsive Surfaces
Mechanism of Action
pH-responsive systems leverage chemical groups that accept or donate protons in response to changes in environmental pH, leading to a structural or property change in the material. Common functional groups include carboxylic acids (-COOH) and primary amines (-NH₂) [13] [46]. For example, a surface decorated with polycarboxylic acids is neutral and relatively hydrophobic at low pH. When the pH increases, the carboxylic acids deprotonate to form carboxylate anions (-COO⁻), rendering the surface hydrophilic and charged, which disrupts electrostatic interactions with cells and induces detachment [13]. Conversely, surfaces with basic groups can become protonated and positively charged at lower pH. This principle is also exploited in drug delivery, where pH-sensitive linkages (e.g., using 2-propionic-3-methylmaleic anhydride) can be designed to degrade in the slightly acidic tumor microenvironment [47].
Experimental Protocols
A typical protocol for harvesting cells from a pH-responsive surface involves the use of a weak acid or base to modulate the local environment.
- Surface Fabrication: One approach involves grafting pH-responsive polymers like polyacrylic acid (PAA) or chitosan onto culture substrates. This can be achieved through various methods, including plasma treatment, UV-induced grafting, or chemical coupling.
- Cell Seeding and Culture: Cells are seeded and cultured on the pH-responsive surface under standard physiological conditions (pH ~7.4) where the surface properties support adhesion.
- Detachment Induction:
- Medium Exchange: Remove the standard culture medium and gently rinse with a neutral buffer like phosphate-buffered saline (PBS).
- pH Shift: Add a mild, biocompatible buffer at a pre-optimized pH. For an anionic system like PAA, a slightly alkaline buffer (e.g., pH 8.0-8.5) is used to ionize the surface. A CO₂-independent medium can also be used to stabilize the pH.
- Incubation and Monitoring: Incubate the culture for a defined period (typically 15-60 minutes) at 37°C. Cells detach as the surface becomes hydrated and charged.
- Harvesting: Gently pipette the buffer across the surface to assist in detaching any remaining cells and collect the cell suspension [13].
Table 2: Comparison of Stimuli-Responsive Cell Detachment Methods
| Method | Stimulus | Key Mechanism | Key Advantages | Key Challenges |
|---|---|---|---|---|
| Thermo-responsive | Temperature | Hydration/dehydration switch of polymer (e.g., pNIPAAm) | Preserves ECM and cell junctions; well-established | Slow detachment; precise polymer synthesis required [13] [45] |
| pH-responsive | pH | Protonation/Deprotonation inducing charge & wettability change | Rapid; uses simple chemical stimulus | Requires medium exchange; potential cytotoxicity from extreme pH [13] |
| Electrochemical | Alternating Current | Redox cycling disrupts cell-adhesive motifs | Rapid (<10 min); high viability (>90%); enzyme-free; scalable [4] | Requires conductive substrate; optimization of voltage/frequency needed [4] |
| Photo-responsive | Light (e.g., UV) | Cleavage of photolabile groups (e.g., o-nitrobenzyl) | High spatiotemporal precision | Potential for UV-induced cell damage; limited penetration depth [13] |
Electrochemical Surfaces
Mechanism of Action
Electrochemical cell detachment is a novel, enzyme-free strategy that uses electrical currents to disrupt cell adhesion. A platform developed by Varanasi and colleagues at MIT uses low-frequency alternating current (AC) on a conductive biocompatible polymer nanocomposite surface [4]. The applied voltage drives alternating redox (reduction-oxidation) reactions at the interface. This electrochemical cycling is thought to degrade or modify the cell-adhesive motifs present on the surface or generate local changes in the ionic microenvironment that disrupt the electrostatic and chemical bonds holding the cells to the substrate. This process effectively releases cells without the use of enzymes, preserving delicate cell membranes and surface proteins, which is especially valuable for sensitive primary cells and immune cells used in CAR-T therapies [4].
Experimental Protocols
This method requires a specialized conductive substrate and precise control of electrical parameters.
- Surface Fabrication: The culture surface is a conductive biocompatible polymer nanocomposite. The exact composition can vary, but it is designed to be non-cytotoxic and support robust cell growth under standard conditions while allowing for the application of an electrical potential.
- Cell Culture: Cells are seeded and cultured to confluence on the conductive surface, similar to traditional methods.
- Electrochemical Detachment Protocol:
- Setup: Place the cell culture vessel into an appropriate holder connected to a power source capable of delivering low-frequency alternating voltage.
- Parameter Application: Apply an optimized low-frequency AC voltage. In the MIT study, identifying the optimal frequency was critical, increasing detachment efficiency from 1% to 95% for human osteosarcoma and ovarian cancer cells [4].
- Process Duration: The detachment process is rapid, typically completing within minutes.
- Cell Collection: Once detachment is confirmed under a microscope, the cell suspension is collected. The researchers reported cell viability exceeding 90% post-detachment [4].
Experimental Workflow for Electrochemical Detachment
The workflow for harvesting cells using an electrochemical surface involves the following key stages:
The Scientist's Toolkit: Research Reagent Solutions
The following table details key materials and reagents essential for working with advanced stimuli-responsive detachment systems.
Table 3: Essential Research Reagents for Stimuli-Responsive Cell Detachment
| Item Name | Function/Description | Key Considerations |
|---|---|---|
| N-Isopropylacrylamide (NIPAAm) Monomer | The primary monomer for synthesizing thermo-responsive pNIPAAm surfaces [45]. | Requires precise control over polymerization and grafting density to ensure proper performance and cell adhesion/detachment. |
| Conductive Polymer Nanocomposite | Serves as the electroactive culture substrate for electrochemical detachment [4]. | Must be biocompatible, support cell growth, and have stable electrical properties. Often requires custom fabrication. |
| Polyacrylic Acid (PAA) or Chitosan | pH-responsive polymers used to fabricate surfaces that respond to pH shifts [13] [46]. | PAA is used for anionic systems; Chitosan, a natural polymer, is used for cationic systems. |
| CO₂-Independent Medium | Used during pH detachment to stabilize the medium pH outside a CO₂ incubator [13]. | Crucial for maintaining the precise pH required to trigger the detachment mechanism. |
| Low-Frequency AC Power Source | Provides the controlled electrical stimulus for electrochemical detachment [4]. | Requires fine control over voltage, frequency, and waveform; optimal parameters are cell-type specific. |
Thermo-responsive, pH-responsive, and electrochemical surfaces represent a significant leap forward in adherent cell detachment technology, moving beyond the destructive nature of conventional enzymatic methods. Each technique offers distinct advantages: thermo-responsive systems excel in harvesting intact, ECM-preserved cell sheets; pH-responsive methods provide a simple, chemically triggered approach; and electrochemical platforms enable rapid, automated, and highly viable cell harvesting with great potential for industrial scale-up [13] [4] [45]. The choice of method depends on the specific application, considering factors such as the need for preserved ECM, sensitivity of the cell type, required speed, and scalability. Future research will focus on enhancing the robustness and scalability of these systems, particularly for bioreactor-based microcarrier culture, and on developing multi-stimuli responsive platforms that offer greater precision and control for the next generation of cell-based therapies and biomanufacturing processes [13].
The transition from laboratory-scale flasks to industrial bioreactors represents a critical bottleneck in the manufacturing of cell-based therapies, vaccines, and cellular agriculture products. Adherent cells, which require a surface for attachment, proliferation, and function, present unique scaling challenges as demands increase from millions to billions of cells [48]. Traditional two-dimensional (2D) culture systems, rooted in plastic dishes and flasks, become prohibitively inefficient due to surface area constraints, labor-intensive handling, and insufficient process control [48]. Microcarrier technology has emerged as a transformative solution to these limitations, offering high surface-to-volume ratios within controlled bioreactor environments [48] [49]. However, the process of efficiently detaching cells from these microcarriers at the end of the expansion phase remains a significant research and operational hurdle. This guide examines the core methodologies, parameters, and emerging technologies for scaling adherent cell cultures, with a specific focus on detachment strategies within the broader context of adherent cell detachment research.
Microcarrier Selection for Scalable Cell Expansion
Microcarriers are tiny beads, typically 100-300 micrometers in diameter, that provide a surface for adherent cells to attach and grow in suspension within a bioreactor [48]. The choice of microcarrier is paramount, as its physical and chemical properties directly impact cell attachment, proliferation, phenotype, and—crucially—the efficiency of subsequent cell detachment and harvest.
Table 1: Common Microcarrier Types and Their Characteristics
| Microcarrier Type | Material | Key Features | Typical Applications | Detachment Considerations |
|---|---|---|---|---|
| Collagen-coated | Polystyrene + ECM | Supports cell adhesion via protein coating | Mesenchymal stem cells (MSCs), fibroblasts [48] | Enzymatic digestion of coating is typically required [48] |
| Native collagen | Collagen | Biodegradable, highly biocompatible | Chondrocytes, skin cells [48] | Enzymatic digestion is typically required [48] |
| Charge-modified | Polystyrene (C, DEAE) | Promotes electrostatic cell attachment | Vero, hybridoma, insect cells [48] | Strong electrostatic bonding can make cell recovery challenging [50] |
| Thermoresponsive | GelMA hydrogel with PNIPAM brushes | Coating enables enzyme-free detachment via temperature shift | Human dermal fibroblasts, MSCs [51] | Low-temperature (e.g., 4°C) incubation yields >95% viability [51] |
The selection of a microcarrier is a balance between facilitating initial cell attachment and enabling efficient final harvest. For instance, while positively charged surfaces promote excellent initial adhesion, they can lead to lower overall cell recoverability due to strong electrostatic interactions that are difficult to disrupt without compromising cell viability [50]. Conversely, collagen-coated and novel thermoresponsive carriers often provide more favorable detachment profiles [51] [50].
Cell Detachment Methodologies: From Enzymatic to Novel Solutions
Detaching cells from microcarriers is a critical unit operation that can significantly impact cell yield, viability, and functionality. Research in this domain is focused on minimizing stress, preserving phenotype, and enabling scalability.
Conventional Enzymatic Detachment
The standard method for cell detachment involves proteolytic enzymes like trypsin or animal-origin-free alternatives (e.g., TrypLE) to cleave the adhesion proteins that bind cells to the microcarrier surface [52] [51]. Key operational parameters must be optimized for a successful harvest [52]:
- Enzyme Volume and Activity: The concentration and activity of the enzyme solution must be sufficient for complete dissociation. Studies have demonstrated feasibility in reducing trypsin volume to 50 mL per gram of microcarrier while maintaining activity above 380 USP/mL for complete cell dissociation [52].
- Agitation Speed: Gentle agitation is critical to ensure uniform enzyme exposure and prevent microcarrier settling, but it must not be so vigorous as to shear cells. An optimized speed is often defined as a multiple of the minimum just-suspended speed (Njs) [52].
- Incubation Time: The duration of enzyme exposure must be long enough to detach cells but minimized to avoid damage. Incubation times can extend up to 60 minutes [52].
- Washing Steps: Implementing multiple washing steps (e.g., two times with PBS) post-medium removal is crucial for eliminating serum that would otherwise inhibit enzyme activity [52].
Despite its widespread use, enzymatic detachment has drawbacks, including potential damage to delicate cell membranes and surface proteins, high cost, introduction of animal-derived components, and generation of significant biological waste [4] [51].
Emerging Enzyme-Free Strategies
Recent research has produced novel, enzyme-free detachment methods that aim to overcome the limitations of enzymatic processes.
- Electrochemical Detachment: A novel platform uses a conductive biocompatible polymer nanocomposite surface and applies a low-frequency alternating current to disrupt cell adhesion. This method has reported detachment efficiencies of 95% while maintaining cell viability exceeding 90% for osteosarcoma and ovarian cancer cells [4] [23]. This approach is promising for enabling automated, closed-loop biomanufacturing systems with minimal waste [4].
- Thermoresponsive Microcarriers: These carriers are fabricated from materials like gelatin methacryloyl (GelMA) and grafted with a polymer (e.g., PNIPAM) that changes hydration state with temperature. A simple shift to a lower temperature (e.g., 4°C) causes the polymer to expand, physically pushing the cells off the surface. This method has achieved up to 69% detachment efficiency for MSCs with >95% post-detachment viability while using 10-fold less enzyme than conventional methods, or even enabling entirely enzyme-free harvest [51].
Optimization and Monitoring of the Detachment Process
Successful scale-up requires rigorous optimization and real-time monitoring of critical process parameters to ensure consistency and efficiency.
Table 2: Key Parameters for Optimizing In-Situ Cell Detachment
| Parameter | Optimization Goal | Experimental Approach | Impact on Process |
|---|---|---|---|
| Agitation Speed | Maintain uniform suspension without shear damage [52] | Test multiples of Njs (e.g., 1.25 x Njs) [52] | Ensures efficient enzyme/carrier contact; prevents cell damage [52] |
| Enzyme Volume & Activity | Minimize volume while ensuring complete dissociation [52] | Titrate enzyme concentration; measure activity (USP/mL) [52] | Reduces cost and waste; maintains high cell viability [52] |
| Incubation Time | Shorten exposure while achieving full detachment [52] | Time-course sampling to monitor detachment efficiency [52] | Prevents prolonged exposure that can damage cells [52] |
| Washing Steps | Remove serum inhibitors effectively [52] | Compare single vs. multiple PBS wash steps [52] | Maximizes enzyme efficiency for reliable detachment [52] |
| Cell Confluence | Detach at optimal growth phase | Monitor confluence via microscopy or sensors | Ensures maximum yield and cell health [53] |
Real-Time Process Monitoring
The integration of Process Analytical Technology (PAT) tools is crucial for advanced bioprocess control. Capacitance sensors (e.g., Hamilton's Incyte sensor) have been effectively used to monitor viable cell density in real-time during the growth phase and are now being applied to the detachment process [52] [54].
These sensors work on the principle of frequency-dependent polarization, detecting cells with intact membranes. During enzymatic detachment, the permittivity signal drops sharply as cells detach from microcarriers. Research has shown that a signal decline below 10 pF/cm indicates complete detachment [54]. Furthermore, predictive models can use signal data from the first 3-7 minutes of trypsinization to forecast the final detachment outcome, enabling rapid, data-driven process decisions without the need for offline sampling [52] [54]. This non-invasive, real-time monitoring significantly enhances process control and is particularly valuable for scaling up.
Diagram: Real-time monitoring workflow for cell detachment.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Microcarrier-Based Cell Culture
| Reagent/Material | Function | Example Use Case |
|---|---|---|
| Cytodex 1 | Dextran-based microcarrier with positive charge for cell attachment [52]. | Expansion of MA 104 and Vero cells for vaccine production [52]. |
| Trypsin-EDTA (0.25%) | Proteolytic enzyme solution for dissociating cells from microcarriers and surfaces [52]. | Standard enzymatic detachment; optimized at 50 mL per gram of microcarriers [52]. |
| TrypLE | Animal-origin-free, recombinant enzyme for cell detachment [51]. | Alternative to trypsin to reduce animal-derived component risk [51]. |
| BrushGel Microcarriers | Thermoresponsive GelMA-based microcarriers with PNIPAM brush coating [51]. | Enzyme-minimized expansion and detachment of MSCs and fibroblasts [51]. |
| Conductive Polymer Nanocomposite | Biointerface for applying electrochemical signals for cell detachment [4] [23]. | Enzyme-free detachment of sensitive cells (e.g., CAR-T, primary cells) [4]. |
| Incyte Capacitance Sensor | PAT tool for real-time, in-line monitoring of viable cell density and detachment status [54]. | Monitoring cell growth and predicting detachment efficiency in bioreactors [54]. |
Scaling adherent cell cultures from lab flasks to industrial bioreactors is a multifaceted challenge centered on the critical step of cell detachment. While optimized enzymatic protocols remain a workhorse, the future of scalable, cost-effective, and gentle cell manufacturing lies in innovative solutions. Electrochemical and thermoresponsive detachment strategies represent a significant paradigm shift toward enzyme-free or enzyme-reduced workflows, minimizing cell damage and process variability [4] [51]. Furthermore, the integration of advanced PAT tools like capacitance sensors for real-time monitoring is transforming process control from an empirical art to a data-driven science [52] [54]. As the fields of cell therapy and cellular agriculture continue to advance, these technologies will be foundational to achieving the robust, automated, and scalable bioprocesses required to meet future demand. Adherent cell detachment research, therefore, is not merely a technical step but a central discipline enabling the next generation of biological manufacturing.
Solving Common Detachment Problems and Optimizing Cell Yield and Quality
Adherent cell detachment is a fundamental step in cell culture, directly impacting cell yield, viability, and the integrity of cellular components for downstream applications. Efficient detachment preserves critical surface proteins and extracellular matrix (ECM) components, which is essential for accurate experimental outcomes in drug development, regenerative medicine, and basic biological research [13]. The process involves disrupting cell-surface adhesions, primarily achieved through enzymatic, non-enzymatic (chelating agents), or physical (mechanical scraping) methods [55] [56]. However, researchers frequently face challenges with poor detachment efficiency, often resulting from suboptimal manipulation of three core parameters: digestion time, reagent concentration, and temperature. This guide details the interplay of these factors, providing evidence-based strategies to optimize protocols, enhance data reliability, and advance cell-based research.
Core Parameters Governing Detachment Efficiency
The efficiency of cell detachment is not governed by a single factor but by the precise interplay of several physical and chemical conditions. Mastering these parameters is key to developing robust and reproducible protocols.
Digestion Time
Digestion time must be carefully calibrated; insufficient time leaves cells adherent, while over-digestion damages cell membranes and degrades surface proteins [56]. The optimal duration is highly dependent on cell type. For instance, a monolayer of standard adherent cells may detach with 0.25% trypsin in 1-5 minutes at 37°C, whereas primary cells or tissues require longer incubation [56]. A study on macrophage detachment revealed that a 10-minute treatment with the enzyme accutase significantly decreased surface levels of Fas ligand (FasL) and Fas receptor compared to mechanical scraping, with the effect being reversible only after a 20-hour recovery period [29]. For isolating bovine adipose tissue-derived mesenchymal stromal cells (MSCs), a 3-hour incubation with Liberase proved optimal for achieving the highest cell yield [57].
Reagent Concentration
The concentration of the detachment reagent directly influences its activity and potential cytotoxicity. Higher concentrations generally accelerate dissociation but increase the risk of damaging surface proteins [29] [13]. Recombinant trypsin is typically used at a concentration of 0.1 mg/ml to 0.3 mg/ml (approximately 500 to 1500 USP units/ml) for cell detachment, with the lower end suitable for most cell lines and the higher end recommended for primary tissues [56]. In the optimization of bovine MSC isolation, a 0.1% enzyme concentration consistently yielded more cells than a 0.04% concentration across various enzymes [57]. Furthermore, chelating agents like EDTA are often used in combination with enzymes (e.g., Trypsin-EDTA) to enhance efficacy by binding calcium and magnesium ions, thereby weakening cell-cell adhesions [55] [56].
Temperature
Temperature is a critical determinant of enzymatic reaction rates. Most enzymatic detachment processes, including those using trypsin, are conducted at 37°C to maximize enzyme activity [55] [56]. However, alternative methods utilize temperature shifts to control detachment. Temperature-responsive culture dishes (TRCDs) are coated with poly(N-isopropylacrylamide (PIPAAm); cells adhere and proliferate at a standard culture temperature (e.g., 37°C) and spontaneously detach as an intact sheet when the temperature is reduced, typically to around 20°C or 32°C, avoiding enzymatic damage [58]. This underscores how temperature can be leveraged as a primary stimulus for detachment.
Table 1: Optimization Guide for Key Detachment Parameters
| Parameter | Typical Range | Effect of Insufficient | Effect of Excessive | Cell Type Example |
|---|---|---|---|---|
| Digestion Time | 1-5 min (trypsin for cell lines) to 3 hours (Liberase for tissue) [57] [56] | Incomplete detachment, low cell yield | Cell death, surface protein degradation (e.g., FasL) [29] [56] | Macrophages, bovine AT-MSCs [29] [57] |
| Reagent Concentration | 0.1-0.3 mg/ml recombinant trypsin; 0.04-0.1% tissue dissociation enzymes [57] [56] | Incomplete detachment | Cytotoxicity, damage to surface markers | Most continuous cell lines, primary tissues [57] |
| Temperature | 37°C (enzymatic); 20-32°C (TRCD detachment) [58] [56] | Slow, inefficient detachment | Increased enzyme toxicity, cell stress | Cells for sheet engineering (via TRCD) [58] |
Experimental Optimization Protocols
Protocol 1: Systematic Optimization for a Novel Cell Line
When working with a new or finicky cell type, a systematic approach is required to identify the ideal detachment conditions.
- Preparation: Pre-warm HBSS (without Ca2+/Mg2+), trypsin (or TrypLE), and complete growth medium to 37°C.
- Initial Setup: Plate cells in multiple identical wells and culture until 80-90% confluent.
- Parameter Testing:
- Concentration Gradient: Prepare a range of trypsin concentrations (e.g., 0.05%, 0.125%, 0.25%) in a balanced salt solution.
- Time-Course: For each concentration, aspirate the medium, add the dissociation reagent, and incubate at 37°C. Observe cells under a microscope every 30-60 seconds.
- Termination: Once ~90% of cells are rounded and begin to detach (gently tap the vessel), neutralize the enzyme with serum-containing medium.
- Assessment: Centrifuge the cell suspension, resuspend, and count cells using an automated counter or hemocytometer. Assess viability via Trypan Blue exclusion.
- Analysis: Plot cell yield and viability against concentration and time. The optimal condition is the one that provides the highest yield with >95% viability in the shortest time [56].
Protocol 2: High-Yield MSC Isolation from Adipose Tissue
This protocol, derived from a study comparing 32 isolation conditions, is optimized for maximum MSC yield from bovine adipose tissue, a relevant source for cultured meat research [57].
- Tissue Preparation: Mince subcutaneous adipose tissue (AT) finely with sterile scissors.
- Enzymatic Digestion: Add 0.1% Liberase in a balanced salt solution to the tissue (e.g., 1 mL per 100 mg tissue).
- Incubation: Incubate the tissue-enzyme mixture for 3 hours at 37°C with constant agitation.
- Separation: Centrifuge the digested suspension to separate the stromal vascular fraction (pellet) from adipocytes and debris (supernatant).
- Plating and Culture: Resuspend the pellet in complete growth medium and plate the cells in a culture vessel.
- Validation: Plastic-adherent, spindle-shaped cells should be observable within 2 days and ready for passage within 4-7 days. This protocol achieved an average cell yield ranging from 30.48 × 10^6 to 67.1 × 10^6 cells per gram of tissue [57].
The following workflow diagram summarizes the key decision points and steps for optimizing cell detachment.
The Scientist's Toolkit: Essential Reagents and Materials
Selecting the appropriate dissociation reagent is fundamental to experimental success. The choice depends on cell type, adherence strength, and the need to preserve surface markers.
Table 2: Research Reagent Solutions for Cell Detachment
| Reagent / Material | Primary Function | Key Applications & Considerations |
|---|---|---|
| Trypsin (Porcine) [55] | Proteolytic enzyme cleaves adhesion proteins. | General purpose for continuous cell lines. May damage surface proteins; requires inactivation with serum or inhibitors. |
| TrypLE (Recombinant) [55] | Animal-origin-free recombinant enzyme with trypsin-like activity. | Gentler alternative to trypsin; stable at room temperature; inactivated by dilution. Ideal for bioproduction. |
| Accutase [29] | Mild enzyme mixture of proteases and collagenases. | Considered gentle, but shown to cleave specific surface proteins (e.g., FasL). Requires recovery time for surface marker studies. |
| Collagenase [55] [57] | Digests native collagen in connective tissues. | Essential for isolating cells from tissues (e.g., adipose, liver). Types I, II, and IV are selected based on tissue. |
| EDTA / Versene [55] [56] | Chelating agent binds Ca2+/Mg2+ ions, disrupting integrin-mediated adhesion. | Mild, non-enzymatic method for weakly adherent cells (e.g., epithelial cells). Often mixed with trypsin to enhance efficacy. |
| Liberase [57] | Highly purified, defined blend of collagenase and neutral protease enzymes. | Provides high yield and viability for challenging isolations, such as MSCs from adipose tissue. |
| Dispase [55] | Neutral protease that cleaves fibronectin and collagen IV. | Gentle harvesting of cell sheets and dissociation of delicate primary cells, with minimal impact on membrane integrity. |
| Temperature-Responsive Surfaces [58] | PIPAAm-coated dishes allow cell attachment at 37°C and detachment upon cooling. | Harvesting intact cell sheets for tissue engineering without enzymatic damage. High cost can be a limitation. |
| Cell Scrapers [55] | Mechanical detachment by physically scraping cells from the surface. | Simple and cost-effective, but can cause significant cell damage and is not suitable for single-cell suspension. |
Achieving optimal adherent cell detachment is a cornerstone of reproducible and reliable cell culture. As research progresses toward more complex applications like the production of cell sheets for regenerative medicine [58] or the scalable manufacturing of cultured meat [57], the precision control of digestion time, reagent concentration, and temperature becomes paramount. By understanding the mechanisms of cell adhesion and the impact of these key parameters, researchers can move beyond standardized protocols to develop tailored, efficient, and gentle detachment strategies. This systematic approach mitigates the common challenge of poor detachment efficiency, ultimately ensuring the integrity of cellular samples and bolstering the validity of downstream scientific data.
The culture of adherent cells is a cornerstone of biomedical research, tissue engineering, and biomanufacturing. A critical yet often disruptive step in these processes is the detachment of cells from culture surfaces, a procedure that inherently challenges cellular integrity. Conventional detachment methods, while effective at releasing cells, frequently compromise key physiological attributes by inducing plasma membrane damage, triggering apoptotic signaling cascades, and degrading functionally important surface proteins. This technical guide examines the mechanisms of detachment-induced cellular stress and presents advanced methodologies designed to preserve cell viability, functionality, and phenotypic stability. Within the broader context of adherent cell detachment research, the evolution from enzymatic and mechanical methods towards precisely controlled, biomimetic, and physical approaches represents a paradigm shift aimed at supporting the rapidly advancing fields of cell therapy, regenerative medicine, and high-throughput drug screening.
Conventional Cell Detachment Methods and Their Limitations
Enzymatic and Mechanical Detachment
Traditional cell harvesting primarily relies on enzymatic and mechanical strategies, each presenting significant challenges for viability and phenotype preservation.
Trypsinization: The most widely used enzymatic method, trypsin is a potent protease that cleaves peptides after lysine or arginine residues. While effective for detachment, it non-specifically degrades cell surface proteins and receptors, potentially damaging delicate membrane structures and altering cellular phenotypes [5]. The required incubation time must be carefully optimized, as over-digestion severely reduces viability.
Accutase: Often considered a milder enzymatic alternative, accutase is a mixture of proteolytic and collagenolytic enzymes. However, evidence shows it can specifically cleave certain surface proteins, notably Fas ligands (FasL) and Fas receptors, while leaving other markers like the macrophage-specific F4/80 unaffected [5]. This selective degradation can compromise critical signaling pathways, such as those mediating immune cell cytotoxicity.
Non-Enzymatic Chelators: Ethylenediaminetetraacetic acid (EDTA) solutions work by chelating calcium ions required for integrin-mediated adhesion. This is a gentler, non-proteolytic approach that better preserves surface markers. However, its efficacy is limited for strongly adherent cell types, often requiring supplemental mechanical force such as scraping, which can itself cause physical membrane damage [5].
Mechanical Scraping: This method physically dislodges cells but risks significant membrane tearing and cell lysis, leading to low yields and the release of intracellular components that can contaminate the culture [5].
Table 1: Impact of Different Detachment Methods on Cell Surface Markers and Viability
| Detachment Method | Mechanism of Action | Effect on Surface FasL/Fas | General Cell Viability | Key Limitations |
|---|---|---|---|---|
| Trypsin | Proteolytic cleavage of adhesion peptides | Significant decrease [5] | Moderate (dose-dependent) | Non-specific protein degradation; broad surface marker damage |
| Accutase | Proteolytic & collagenolytic activity | Significant decrease (reversible) [5] | High post-detachment [5] | Selective cleavage of specific surface markers (e.g., FasL, Fas) |
| EDTA-based Solutions | Chelation of Ca²⁺ ions | Minimal decrease [5] | Moderate to High | Weak efficacy on strongly adherent cells; may require scraping |
| Mechanical Scraping | Physical dislodgement | Minimal decrease [5] | Low | High risk of membrane rupture and physical damage |
Initiating Apoptotic Signaling
Beyond immediate physical damage, detachment stress can activate programmed cell death pathways. Apoptosis can be triggered through two principal routes:
The Extrinsic Pathway: Initiated by extracellular death ligands binding to cell surface death receptors (e.g., Fas). Notably, some detachment enzymes like accutase directly cleave FasL, potentially disrupting this pathway and creating artifacts in apoptosis studies [5].
The Intrinsic (Mitochondrial) Pathway: Activated by internal cell stress signals. Detachment from the extracellular matrix (anoikis), chemical toxicity from detachment agents, or mechanical shear stress can cause an imbalance in the Bcl-2 family of proteins, leading to Mitochondrial Outer Membrane Permeabilization (MOMP) and the release of pro-apoptotic factors like cytochrome c [59] [60]. This, in turn, activates a cascade of caspases—initiator caspases (e.g., caspase-8, -9) and executioner caspases (e.g., caspase-3, -7)—that systematically dismantle the cell [59].
The following diagram illustrates these key apoptotic pathways and their potential activation by detachment stressors.
Advanced Techniques for High-Viability Cell Detachment
Electrochemical Detachment
A novel enzyme-free strategy uses alternating low-frequency electrochemical current on a conductive biocompatible polymer nanocomposite surface [4]. This approach disrupts cell adhesion without enzymatic digestion or significant mechanical force.
- Mechanism: The application of alternating voltage dynamically shapes the ionic microenvironment at the cell-surface interface, likely disrupting electrostatic and other non-covalent interactions that mediate adhesion.
- Performance: This method achieves a detachment efficiency of up to 95% while maintaining over 90% cell viability, effectively overcoming the primary limitations of enzymatic and mechanical methods [4].
- Advantages:
- Animal-derived component-free: Eliminates compatibility concerns for human therapies.
- Scalable and automatable: Suitable for large-scale biomanufacturing like CAR-T cell production [4].
- Reduced waste generation: Addresses the environmental burden of an estimated 300 million liters of annual cell culture waste from traditional methods [4].
The experimental workflow for implementing and validating this electrochemical detachment method is outlined below.
Quantitative Analysis of Detachment Forces
Understanding the physical forces involved in cell detachment is crucial for optimizing techniques. Research comparing different measurement setups (pipette-pipette, plate-plate, plate-pipette) reveals that the measured detachment force is not an intrinsic cellular property but a global system property dependent on the experimental setup [61] [62]. For instance, the detachment force ( F_{dc} ) for two identical cells can be analytically expressed as:
[ F{dc} = \frac{1}{2}\pi \gamma \,rH \cos^2\theta_c ]
where ( \gamma ) is cell tension, ( rH ) is a curvature parameter, and ( \thetac ) is the contact angle [61]. This formula highlights that the force required is influenced by both cell-specific parameters and the geometry of the holding apparatus. Crucially, the same cells will experience different forces and potentially different outcomes in different experimental or processing setups.
Table 2: Comparison of Cell Detachment and Viability Assessment Techniques
| Technique | Principle | Throughput | Key Metric | Reported Performance/Value |
|---|---|---|---|---|
| Electrochemical Detachment [4] | Alternating current on conductive polymer | Potentially High (Automable) | Detachment Efficiency & Viability | 95% Efficiency, >90% Viability |
| Micropipette Force Measurement [61] | Direct physical pulling via micropipette | Low (Single-cell) | Detachment Force ( ( F_{dc} ) ) | Setup-dependent (nN to μN range) |
| Geometric Viability Assay (GVA) [63] | Colony distribution in a cone (pipette tip) | Very High (1200/day) | Viable Cell Concentration | Correlates with CFU (Pearson r=0.98) |
| Traditional Colony-Forming Unit (CFU) [63] | Colony growth on a plate | Low | Viable Cell Concentration | Gold Standard, but time/resource intensive |
Experimental Protocols for Validation and Analysis
Protocol: Electrochemical Cell Detachment
Objective: To harvest adherent cells using an electrochemical platform to maximize viability and minimize membrane damage [4].
Materials:
- Conductive biocompatible polymer nanocomposite culture surfaces
- Low-frequency alternating current power source
- Biocompatible electrolyte buffer (e.g., PBS)
- Standard cell culture reagents
Procedure:
- Cell Culture: Culture adherent cells (e.g., human osteosarcoma or ovarian cancer cells) to confluence on the conductive polymer surface.
- System Setup: Place the culture surface in a suitable chamber and immerse in a biocompatible buffer.
- Application of Current: Apply a low-frequency alternating voltage. The optimal frequency must be determined empirically (e.g., identified in the study for high efficiency).
- Monitoring and Harvesting: Monitor detachment visually. The process typically completes within minutes. Gently agitate or pipette the buffer to suspend released cells.
- Cell Collection: Transfer the cell suspension to a collection tube and centrifuge gently to pellet cells. Resuspend in fresh culture medium or buffer for downstream applications.
Validation:
- Viability Assessment: Use trypan blue exclusion or fluorescent live/dead stains to confirm viability exceeds 90%.
- Functionality Tests: Assess post-harvest functionality through re-plating efficiency, specific surface marker expression (via flow cytometry), or functional assays relevant to the cell type.
Protocol: Assessing Viability with the Geometric Viability Assay (GVA)
Objective: To rapidly and accurately quantify viable cell concentration after detachment with minimal waste and time [63].
Materials:
- Transparent pipette tips
- Low-melt agarose
- Appropriate culture medium (e.g., LB for bacteria, specific medium for mammalian cells)
- Triphenyl tetrazolium chloride (TTC) for contrast (optional)
- Incubator
- Imaging setup (e.g., custom-built with Canon camera) [63]
Procedure:
- Sample Preparation: Mix the cell suspension thoroughly with melted, cooled (≤55°C) agarose containing culture medium and TTC to a final agarose concentration of 0.5%.
- Loading: Pipette the cell-agarose mixture into a standard pipette tip. Avoid air bubbles.
- Solidification: Eject the solidified agarose plug from the tip into a sterile container, or allow it to solidify directly within the tip placed in a rack.
- Incubation: Incubate the tips or plugs at the appropriate temperature and conditions (e.g., 37°C overnight for many bacterial cells) to allow colony formation.
- Imaging and Analysis: Image the entire plug/tip. Measure the perpendicular distance (x) of each colony from the tip. Calculate the viable cell concentration using the formula: [ \text{CFUs/mL} = \frac{N(x)}{V \int{x1}^{x2} \frac{3x^2}{h^3} dx} ] where ( N(x) ) is the number of colonies counted between positions ( x1 ) and ( x_2 ), ( V ) is the volume of the cone, and ( h ) is the total cone length [63].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Research Reagent Solutions for Cell Detachment and Viability Analysis
| Reagent/Material | Function | Key Considerations |
|---|---|---|
| Conductive Polymer Nanocomposite [4] | Surface for electrochemical detachment | Must be biocompatible and support normal cell growth prior to detachment. |
| Accutase [5] | Enzymatic detachment solution | Effective but requires validation for specific surface markers; allows high post-detachment viability. |
| EDTA-based Solution (Versene) [5] | Non-enzymatic, calcium-chelation detachment | Mild but may be insufficient for strongly adherent cells; best for preserving surface proteins. |
| Cell Preparation Tubes (CPTs) [64] | Integrated tube for PBMC isolation | Enables automated processing; contains anticoagulant, density medium, and a gel barrier. |
| Low-Melt Agarose [63] | Matrix for embedding cells in GVA | Must be cooled appropriately before mixing with cells to maintain viability. |
| Triphenyl Tetrazolium Chloride (TTC) [63] | Stain for visualizing colonies in GVA | Metabolically reduced by growing cells to a red formazan, increasing contrast. |
The pursuit of gentle, efficient, and high-fidelity cell detachment methods is a critical focus within adherent cell research, directly enabling advancements in cell-based therapies and precision medicine. The limitations of conventional enzymatic and mechanical techniques—membrane damage, induction of apoptosis, and alteration of surface phenotypes—are now being systematically addressed by innovative approaches. Electrochemical detachment offers a scalable, enzyme-free alternative that preserves viability, while sophisticated assays like GVA allow for rapid, high-throughput validation. As the field progresses, the integration of these advanced methods into automated, closed-loop biomanufacturing systems will be essential for producing the high-quality cells required for the next generation of regenerative medicine and therapeutic applications. Future research will likely focus on further refining the specificity and gentleness of these techniques, particularly for sensitive primary and immune cells, ensuring that the process of harvesting cells does not undo the careful work of culturing them.
Within the broader field of adherent cell detachment research, a central challenge is the harvesting of cells without altering their native biological state, particularly for subsequent analytical techniques like flow cytometry. The integrity of cell surface proteins is paramount; they are the primary targets for immunophenotyping, a cornerstone of immunology, oncology, and drug development [65]. These proteins, including Cluster of Differentiation (CD) antigens, are not merely passive markers but are often functionally critical mediators of cell-cell interaction and signal transduction [65]. Unfortunately, the very process of disrupting cell-adhesion mechanisms to harvest adherent cells can inflict significant damage on these delicate surface epitopes.
Routine enzymatic treatments, such as trypsinization, are fraught with challenges. Proteases like trypsin work by cleaving proteins of the extracellular matrix and, non-specifically, cell surface receptors, potentially damaging the very epitopes that antibodies are designed to recognize [13]. This can lead to downregulation of protein expression, boosted apoptotic cell death, and aberrant experimental results [13]. Consequently, selecting a detachment method that preserves surface protein integrity and understanding the necessary post-harvest recovery period are essential for generating reliable, high-quality flow cytometry data. This guide provides a detailed framework for method selection and protocol optimization to achieve this goal.
Cell Detachment Methods: A Comparative Analysis
The choice of detachment strategy represents a fundamental trade-off between efficiency and the preservation of cellular physiology. The following table summarizes the key characteristics of common detachment methods.
Table 1: Comparison of Adherent Cell Detachment Methods for Flow Cytometry
| Method | Mechanism of Action | Impact on Surface Proteins | Recommended Recovery Time | Best Use Cases |
|---|---|---|---|---|
| Enzymatic (Trypsin) [1] [13] | Proteolytic cleavage of adhesion proteins. | High Risk: Cleaves surface proteins and receptors, potentially destroying epitopes. | 6-10 hours on a rocker platform to allow receptor regeneration [66]. | Routine sub-culturing where surface protein integrity is not a primary concern. |
| Enzymatic (TrypLE, Accutase) [27] [65] | Broad-spectrum proteolytic activity. | Moderate Risk: Milder action than trypsin, but epitope damage remains possible. | At least 30 minutes, but requires optimization for specific cell-antibody pairs. | A gentler alternative to trypsin for sensitive cells. |
| Non-Enzymatic (Chelators like EDTA) [1] [13] | Binds calcium and magnesium, disrupting calcium-dependent cell adhesion. | Lower Risk: Does not cleave proteins; primarily affects integrin function. | Minimal to 30 minutes [66]. However, chelators can cause unexpected cell detachment during staining, so washing is crucial. | Ideal for cells that adhere via Ca²⁺-dependent mechanisms when epitope preservation is critical. |
| Mechanical (Scraping) [27] | Physical dislodgement of cells. | Variable Risk: Can cause significant physical damage and shear stress, leading to uncontrolled protein loss or activation. | Not typically applicable, as physical damage is not reversible via incubation. | A last resort for cells extremely sensitive to all chemical methods; not recommended for flow cytometry. |
| Advanced Non-Enzymatic (Electrochemical) [4] | Alternating electrochemical current disrupts adhesion on a smart surface. | Very Low Risk: Enzyme-free process avoids proteolytic damage, maintaining over 90% cell viability and surface protein integrity. | Minimal, as the method is designed to be non-destructive. | High-value applications like CAR-T therapy manufacturing, sensitive primary cells, and automated bioprocessing. |
Optimized Experimental Protocols for Flow Cytometry
Cell Harvesting with Surface Protein Preservation
The following protocol is optimized to minimize surface protein damage during the harvesting of adherent cells for flow cytometry.
Materials Required:
- Flow Cytometry Staining Buffer: A phosphate-buffered saline (PBS) solution containing 0.5-1% bovine serum albumin (BSA) or 2-10% fetal calf serum (FCS) [67] [66].
- Detachment Reagent: A non-enzymatic option like EDTA (e.g., 0.5 mM or 10 mM in PBS) is recommended for optimal epitope preservation [27] [66].
- Fc Receptor Blocking Reagent: Such as human IgG, mouse anti-CD16/CD32, or goat serum, to prevent non-specific antibody binding [67] [66].
Procedure:
- Wash: Remove and discard the spent culture media. Gently wash the cell layer with a balanced salt solution without calcium and magnesium (e.g., PBS) to remove any traces of serum that can inhibit detachment reagents [1].
- Detach: Add pre-warmed (37°C) EDTA solution to cover the cell layer. Incubate at 37°C for the minimum time required for detachment (typically 2-10 minutes), gently tapping the vessel to aid detachment [1] [66].
- Neutralize and Harvest: Once ≥90% of cells are detached, promptly add twice the volume of pre-warmed complete growth medium or flow cytometry staining buffer to neutralize the EDTA. Gently pipette to create a single-cell suspension and transfer to a conical tube [1].
- Wash and Count: Centrifuge the cells at ~200-500 x g for 5 minutes at 4°C. Discard the supernatant and resuspend the pellet in flow buffer. Perform a cell count and viability analysis [67] [27]. Viability should ideally be 90-95% [67].
- Post-Harvest Recovery (if required): If enzymatic detachment was unavoidable, a recovery incubation is essential. Resuspend the cell pellet in complete growth medium and incubate on a rocker platform for 6-10 hours to allow surface proteins to regenerate [66]. After recovery, proceed to staining.
Staining Protocol for Surface Antigens
This protocol assumes a single-cell suspension with high viability has been prepared.
- Aliquot Cells: Transfer up to (1 \times 10^6) cells per sample into a FACS tube or a well of a 96-well plate [67] [66].
- Fc Blocking (Critical): To prevent non-specific antibody binding, resuspend the cell pellet in an Fc receptor blocking buffer and incubate for 15-30 minutes in the dark at 4°C [67] [66]. Do not wash after this step.
- Surface Antibody Staining: Add a pre-titrated amount of fluorochrome-conjugated antibody directly to the tube. Vortex gently and incubate for 30 minutes in the dark at 4°C [66].
- Wash: Add 2 mL of cold flow cytometry staining buffer and centrifuge at 200-500 x g for 5 minutes. Discard the supernatant carefully. Repeat this wash step two more times to remove unbound antibody completely [67] [66].
- Fixation (Optional): If the cells do not need to be kept alive for subsequent experiments, resuspend the pellet in a fixative such as 1-4% paraformaldehyde for 15-20 minutes on ice to preserve the stained sample [67]. Wash once after fixation.
- Resuspend and Analyze: Resuspend the final cell pellet in 200–400 µL of flow cytometry staining buffer for acquisition on the flow cytometer [66].
Diagram 1: Cell harvest and recovery workflow.
The Scientist's Toolkit: Essential Reagents for Surface Staining
Table 2: Key Research Reagent Solutions for Flow Cytometry
| Reagent / Material | Function / Purpose | Example |
|---|---|---|
| Flow Cytometry Staining Buffer [66] | Provides an isotonic, protein-supplemented environment for antibody dilution and washing to maintain cell stability and reduce background noise. | PBS with 0.5% BSA or 2% FCS, often containing sodium azide. |
| Fc Receptor Blocking Reagent [67] [66] | Blocks Fc receptors on immune cells to prevent non-specific binding of antibodies, thereby reducing false positives. | Human IgG, Mouse anti-CD16/CD32, Goat serum. |
| Viability Dye [67] [68] | Distinguishes live from dead cells during analysis. Dead cells bind antibodies non-specifically and must be excluded by gating. | 7-AAD, DAPI, Propidium Iodide (PI), or fixable amine-reactive dyes. |
| Cell Strainer [27] | Removes cell clumps and debris from the single-cell suspension to prevent clogging the flow cytometer and reduce analysis artifacts. | Nylon mesh filters (e.g., 70 µm). |
| Fixative [67] | Preserves cell structure and stabilizes the antibody-antigen complex for delayed analysis, but can alter epitopes and scatter properties. | 1-4% Paraformaldehyde (PFA). |
Advanced Techniques and Emerging Technologies
The field of adherent cell detachment is rapidly evolving beyond traditional methods. Research is increasingly focused on stimuli-responsive surfaces that allow for highly controlled, non-invasive cell release.
- Thermo-responsive Polymers: These surfaces, often coated with polymers like poly(N-isopropylacrylamide), allow for cell attachment and proliferation at 37°C. When the temperature is reduced, the polymer undergoes a hydrophilic transition, prompting cells to detach spontaneously along with their deposited extracellular matrix, forming an intact "cell sheet" [13]. This method entirely avoids proteolytic damage.
- Electrochemical Platforms: A groundbreaking approach uses alternating electrochemical current on a conductive biocompatible polymer surface. This method disrupts cell adhesion without enzymes, achieving over 95% detachment efficiency while maintaining over 90% cell viability, as demonstrated with human cancer cells [4]. This technology is particularly promising for automating cell therapy manufacturing workflows.
Diagram 2: Emerging non-enzymatic detachment technologies.
The journey from an adherent cell culture to a reliable flow cytometry dataset is fraught with potential pitfalls that can compromise surface protein integrity. As detailed in this guide, the selection of a gentle, non-enzymatic detachment method like EDTA, coupled with a rigorous staining protocol that includes Fc receptor blocking and adequate post-harvest recovery when needed, is critical for success. The ongoing research in smart, stimuli-responsive biomaterials heralds a future where cell harvesting can be both highly efficient and exceptionally gentle, thereby enhancing the reproducibility and biological relevance of data in drug development and cellular research.
Managing Contamination and Process Compatibility in Biopharmaceutical Workflows
In biopharmaceutical manufacturing, the processes of adherent cell detachment and contamination control are intrinsically linked. Effective management of this intersection is paramount for producing safe and efficacious therapies, especially advanced modalities like cell and gene therapies. Anchorage-dependent cells require physical attachment to a surface to grow and reproduce, making the detachment step—the separation of these cells from their culture surface—a critical part of the production workflow [4]. Unfortunately, this necessary step often presents significant opportunities for microbial contamination, which can compromise product quality and patient safety.
Traditional cell detachment methods, particularly those using animal-derived enzymes, are fraught with challenges. They can damage delicate cell membranes and surface proteins, involve slow, multi-step processes that increase contamination risk, and generate substantial biological waste—estimated at 300 million liters of cell culture waste annually [4]. Furthermore, these methods can introduce compatibility concerns for human therapies and limit scalability in modern biomanufacturing. This technical guide examines advanced strategies and innovative technologies designed to mitigate contamination risks while maintaining process compatibility across biopharmaceutical workflows, with a specific focus on implications for adherent cell detachment research.
Contamination Risks in Cell Culture and Detachment Processes
A risk-based analysis of bioprocessing reveals multiple vectors for contamination, each requiring specific control strategies:
- Raw Materials: Cell lines themselves are a primary source, with studies indicating 5-35% of bioproduction cell lines harbor mycoplasma contamination. Even test reagents, such as DNA-extraction kits or Bovine Serum Albumin (BSA) used in assays, can be contamination sources [69].
- Human Operators: Employees represent a critical control point, historically accounting for a significant percentage of Good Manufacturing Practice (GMP) deviations. Although automation and process improvements have reduced this, the human element remains a substantial variable [69].
- Manufacturing Environments: Airflow, water systems, and cleanroom surfaces can all introduce contaminants. Approximately 10% of process contamination originates from cleanroom airflow systems, highlighting the importance of proper HVAC maintenance and monitoring [69].
- Process Equipment: Single-use systems (SUS) with assembly defects or damage can introduce contamination during processing. Additionally, endogenous viral gene sequences present in approximately 5% of Chinese hamster ovary (CHO) cell lines represent a less obvious but significant risk [69].
Special Considerations for Cell Therapy Manufacturing
The challenges of contamination control are magnified in cell therapy manufacturing, particularly for autologous products where each batch is patient-specific and timing is critical. For treatments like CAR-T cell therapies, which are often the last option for severely ill patients, there is typically only one opportunity to manufacture the product successfully. A contamination event necessitates discarding the product and restarting manufacturing, with significant implications for cost and patient health [70]. The manual processes common in small-scale cell therapy production, often conducted in open biosafety cabinets rather than fully closed systems, further elevate contamination risks compared to traditional biomanufacturing with closed systems [70].
Comprehensive Contamination Control Strategies
Foundations of an Effective Control Strategy
A comprehensive contamination control strategy extends beyond traditional microbiology testing to encompass prevention, monitoring, and systematic response protocols. Key elements include:
- Preventive Design: Process systems should be designed with contamination risks in mind, incorporating proper gowning procedures, isolators or enclosed production systems, appropriate air filtration and pressure controls, and regular disinfection protocols [70].
- Environmental Monitoring (EM): Ongoing EM activities with data trending help identify potential contamination concerns before they impact manufacturing. These programs should be risk-based regarding sampling location, frequency, and methodology [70].
- Investigation and Response: When contamination events or microbial data deviations occur, comprehensive investigations must address root cause analysis, process impact assessment, and appropriate corrective and preventive actions [70].
Decontamination Methodologies
Effective decontamination is achieved through manual or automated approaches, each with distinct advantages:
Table 1: Comparison of Automated Decontamination Methods
| Contamination Control Method | Advantages | Disadvantages |
|---|---|---|
| UV Irradiation | Speed; no requirement to seal enclosure | Prone to shadowing; may not kill spores; efficacy decreases with distance from light source |
| Chlorine Dioxide | Highly effective at killing microbes; can be quick at high concentrations | Highly corrosive, causing equipment damage; high consumables cost; high toxicity requires building evacuation |
| Aerosolized Hydrogen Peroxide | Good material compatibility; effective at killing microbes | Liquid droplets prone to gravity; relies on direct line of sight; longer cycle times; more suitable for small isolators |
| Hydrogen Peroxide Vapor | Highly effective; excellent distribution; good material compatibility; quick cycle times with active aeration; typically includes safety sensors | Requires specialized equipment [70] |
While manual decontamination requires minimal capital investment, automated systems provide superior consistency, repeatability, reduced disinfection time, and lower operator resource requirements [70]. For cell therapy facilities, many developers implement a transition timeline, beginning with manual processes and progressing to automated systems as products approach approval.
The following workflow illustrates the relationship between cell detachment methods and contamination control strategies in a biopharmaceutical workflow:
Advanced Cell Detachment Methods and Process Compatibility
Limitations of Traditional Detachment Methods
Traditional enzymatic cell detachment using trypsin or other proteases, while widely employed, presents significant limitations for modern biomanufacturing. These methods can cleave anchoring proteins and essential surface receptors, leading to dysregulation of protein expression and metabolic pathways [13]. Trypsinization has been shown to boost apoptotic cell death rates and enhance oncogene expression, creating unwanted problems for cell transplantation therapy, tissue engineering, and regenerative medicine [13].
Innovative Enzyme-Free Detachment Technologies
Recent research has focused on developing alternative detachment methods that minimize cellular damage while reducing contamination risks:
Electrochemical Detachment: MIT researchers have developed a novel enzyme-free strategy using alternating electrochemical current on a conductive biocompatible polymer nanocomposite surface. This approach disrupts adhesion within minutes while maintaining over 90% cell viability, overcoming limitations of enzymatic and mechanical methods [4]. The method applies low-frequency alternating voltage to create electrochemically mediated redox reactions at the cell-surface interface, enabling rapid detachment without enzymatic damage.
Stimuli-Responsive Surfaces: Various smart materials have been investigated for cell detachment, including thermo-responsive polymers that change properties with temperature shifts, pH-responsive materials that react to environmental acidity changes, and light-sensitive coatings that release cells upon irradiation [13]. While promising, these approaches often require precise control over material characteristics and can face challenges in robustness and scalability.
Experimental Protocol: Electrochemical Cell Detachment
The following detailed methodology outlines the enzyme-free electrochemical detachment process developed by MIT researchers [4]:
Surface Preparation: Culture cells on a conductive biocompatible polymer nanocomposite surface specifically engineered for electrochemical responsiveness.
Culture Conditions: Maintain cells under standard conditions (37°C, 5% CO₂) until they reach desired confluency, typically 70-90%.
Solution Replacement: Replace standard culture medium with an electrochemically compatible buffer solution optimized for charge transfer.
Voltage Application: Apply low-frequency alternating voltage (specific optimal frequency identified by researchers) using a specialized electrochemical instrument. Typical parameters range from 0.1-10 Hz at voltages maintaining cell viability.
Process Monitoring: Observe detachment progress microscopically. The process typically requires several minutes for substantial cell release.
Cell Collection: Gently rinse the surface with buffer to collect detached cells once ≥90% detachment is achieved.
Viability Assessment: Evaluate cell viability using standard methods (Trypan blue exclusion or automated cell counting), consistently demonstrating >90% viability.
Further Processing: Transfer cells to subsequent manufacturing steps without the enzyme neutralization required in traditional methods.
This protocol represents a significant advancement in contamination-conscious workflow design by eliminating animal-derived enzymes, reducing processing steps, and maintaining high cell viability.
Process Compatibility and Scalability Considerations
Integration with Upstream and Downstream Processes
Successful implementation of any detachment method requires seamless integration with both upstream and downstream bioprocessing stages:
Upstream Compatibility: Upstream processing focuses on cell cultivation and growth, requiring detachment methods that maintain high viability and phenotype stability for subsequent expansion or direct therapeutic use [71]. Advanced methods like electrochemical detachment show particular promise for preserving these qualities.
Downstream Compatibility: Downstream processing involves separation, purification, and concentration of the final product [71]. Detachment methods must not interfere with purification technologies, particularly for sensitive applications like cell therapies where traditional purification steps may be limited.
Microcarrier Harvesting: For large-scale bioreactor cultures using microcarriers, detachment faces additional challenges. Enzymatic harvesting remains common, but stimuli-responsive microcarriers with smart coatings are emerging as alternatives, though they face similar scalability and robustness challenges as other advanced methods [13].
Automation and Closed-System Integration
A critical advantage of advanced detachment methods like the electrochemical approach is their compatibility with automation and closed-system manufacturing. As noted by researchers, "Because this method can be applied uniformly across large areas, it's ideal for high-throughput and large-scale applications like cell therapy manufacturing. We envision it enabling fully automated, closed-loop cell culture systems in the near future" [4]. This compatibility addresses a fundamental contamination vector—human intervention—while supporting the scale-up requirements of commercial biomanufacturing.
The following diagram illustrates the compatibility framework between cell detachment methods and biomanufacturing requirements:
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Research Reagent Solutions for Cell Detachment and Contamination Control
| Reagent/Technology | Function | Application Notes |
|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme chelating agent combination for enzymatic cell detachment | Effective but can damage surface proteins; requires neutralization; animal-derived sources pose contamination risk [1] [13] |
| TrypLE | Proprietary enzyme-free dissociation reagent | Reduced enzyme activity concerns; functionally similar to trypsin but with potentially gentler action [1] |
| Electrochemical Surfaces | Conductive biocompatible polymer nanocomposites for electrical detachment | Enable enzyme-free detachment with >90% viability; compatible with automation [4] |
| Thermo-responsive Polymers | Surface coatings that change properties with temperature shifts | Allow cell sheet harvesting without enzymatic treatment; require precise control of film characteristics [13] |
| Hydrogen Peroxide Vapor | Automated decontamination method for rooms and enclosures | Highly effective with excellent distribution and material compatibility; features quick cycle times with active aeration [70] |
| Membrane Chromatography | Purification technology for large biomolecules | Faster processing with higher throughput; ideal for advanced therapies; uses convective flow rather than diffusion [72] |
The integration of advanced cell detachment technologies with comprehensive contamination control strategies represents a paradigm shift in biopharmaceutical manufacturing. Methods like electrochemical detachment that eliminate animal-derived enzymes while maintaining high cell viability offer compelling advantages for both contamination reduction and process compatibility. When combined with robust contamination control strategies—including automated decontamination, closed-system processing, and rigorous environmental monitoring—these approaches significantly enhance manufacturing reliability and product safety.
Future advancements will likely focus on further automation and single-use technologies to minimize human-dependent contamination vectors. Additionally, the continued development of stimuli-responsive materials and precision detachment methods will enable more selective cell harvesting with minimal phenotypic impact. As the biopharmaceutical industry evolves toward more personalized therapies and smaller batch sizes, the flexibility, safety, and compatibility of detachment methods will become increasingly critical factors in manufacturing success. By adopting these advanced approaches, researchers and manufacturers can address the dual challenges of contamination control and process compatibility while advancing the development of next-generation biotherapeutics.
The process of detaching adherent cells is a critical, yet often disruptive, phase in cell culture workflows, directly impacting cell viability, phenotypic stability, and experimental reproducibility. This whitepaper provides an in-depth technical guide for researchers and drug development professionals on optimizing three foundational culture conditions—medium composition, pH stability, and serum selection—to mitigate the stresses of detachment and support robust post-detachment health. Framed within the broader context of adherent cell detachment research, this review synthesizes current protocols and data, emphasizing how pre- and post-detachment culture strategies can preserve cellular integrity and function for downstream applications in biomanufacturing and therapy development.
Adherent cell detachment is an essential step in subculturing, cell harvesting for therapy, and various analytical procedures. However, the very process of breaking cell-substrate adhesions induces significant cellular stress. Traditional enzymatic methods, while effective, can damage delicate cell membranes and cleave crucial surface proteins, receptors, and ion channels [5] [13]. Non-enzymatic methods, including chelating agents and novel physical stimuli, present alternatives but come with their own sets of challenges regarding efficacy and scalability [4] [13].
The "post-detachment health" of a cell population is not merely a measure of viability but encompasses the preservation of functional integrity, including metabolic activity, surface marker profiles, and proliferative capacity. A growing body of research indicates that the culture conditions before and immediately after detachment are pivotal in determining these outcomes. The medium provides essential nutrients and growth factors, serum (or its alternatives) supplies adhesion proteins and mitogens, and tightly regulated pH maintains optimal enzymatic and metabolic function. Instability in any of these components can exacerbate detachment-induced stress, leading to aberrant phenotypes, activated stress-response pathways, and compromised experimental or therapeutic value [5] [73]. This guide details the optimization of these core elements to foster a culture environment that actively supports cell recovery and functional maintenance.
Optimizing Culture Medium Formulation
The culture medium is the most critical component of the environment, providing necessary nutrients, growth factors, and hormones, while also regulating pH and osmotic pressure [74]. Selecting and managing the medium appropriately is fundamental to pre-detachment cell health and post-detachment recovery.
Basal Media Selection and Nutrient Supplementation
The choice of basal medium should be tailored to the specific cell type, as nutritional requirements can vary significantly. For instance, MEM is a common starting point for many adherent cells like fibroblasts, while DMEM/F12 is often chosen for its rich composition, supporting a wider variety of cells, including in serum-free applications [74].
- Pre-detachment Conditioning: Ensure cells are in the log phase of growth with viability greater than 90% prior to detachment. Depleted nutrients or accumulated metabolic waste (e.g., high lactate, ammonia) in over-confluent cultures can weaken cells, making them more susceptible to detachment damage [1] [73].
- Post-detachment Recovery: Resuspend the detached cell pellet in fresh, pre-warmed complete growth medium immediately after centrifugation. This provides immediate access to essential nutrients and energy for membrane repair and recovery of surface proteins [1]. Using spent or expired medium can hinder recovery and reduce seeding efficiency in the new vessel.
Strategic Use of Serum-Free Media (SFM) and Supplements
The shift towards serum-free media (SFM) is driven by the need for defined, consistent, and xeno-free conditions, particularly for clinical applications. However, SFM formulations are highly specialized and their performance can vary.
- Performance and Cost Considerations: A 2025 study highlights that not all commercially available SFM support optimal cell growth equally. While some SFM supported mesenchymal stem cell (MSC) expansion well, others did not. In contrast, all human platelet lysate (hPL) preparations in the study supported growth. The study also concluded that the cost-performance balance currently favors hPL over SFM [75].
- Hidden Animal-Derived Components: The same study revealed a critical finding: some commercially marketed SFM were found to contain significant levels of human blood-derived components like myeloperoxidase, glycocalicin, and fibrinogen. This can misleadingly reclassify them and impact their regulatory status for cell therapy [75]. Researchers must rigorously vet SFM suppliers.
- Supplementation Strategies: Reduced-serum media are basal formulations enriched with nutrients and animal-derived factors, which lower the required serum percentage. For SFM, supplements like bovine serum albumin (BSA), lipids, and specific growth factors are used to create a defined environment that can be selective for specific cell types [74].
Table 1: Comparison of Common Basal Media for Adherent Cells
| Culture Medium | Full Name | Typical Applications | Key Features |
|---|---|---|---|
| MEM | Minimum Essential Medium | General cell culture, adherent cell lines, primary cells, cytotoxicity assays | A simple, widely used medium; available with different salt formulations [74]. |
| DMEM | Dulbecco's Modified Eagle Medium | Fibroblasts, neurons, glial cells, HUVECs, cancer research | Available in high and low glucose formulations; can be supplemented with pyruvate [74]. |
| DMEM/F12 | Dulbecco's Modified Eagle Medium: Nutrient Mixture F-12 | Mammalian cells, serum-free growth of CHO cells, hybridomas | Combines the rich nutrients of DMEM and F-12; supports a wide variety of cells [74]. |
| RPMI 1640 | Roswell Park Memorial Institute 1640 Medium | Lymphocyte culture, hybridoma cells, immunological studies | Originally developed for suspension cells but used for some adherent lines; often supplemented with HEPES [74]. |
Maintaining pH Homeostasis
pH fluctuation is a primary source of stress in cell culture, directly affecting enzyme activity, cellular metabolism, and the integrity of the cell membrane. Maintaining stable pH before, during, and after detachment is therefore critical.
Causes and Impacts of pH Fluctuation
Most cell lines grow optimally at a pH of 7.2–7.4 [73]. Significant deviations can trigger cell detachment and death.
- Metabolic Acidification: As cells metabolize, they produce and release acidic metabolites (e.g., lactic acid, CO₂), causing the medium's pH to drop. This is visually indicated by a color change of phenol red from red (pH ~7.4) to yellow (pH <7.0) [73].
- Alkalization: An increase in pH, indicated by a purple hue in the medium, can result from insufficient CO₂ in the incubator or poor gas exchange in tightly sealed culture vessels [73].
- Detachment-Reagent Induced Shifts: The addition of dissociation reagents like trypsin or EDTA, which are not typically pH-balanced, can cause a sudden, localized shift in pH, further stressing cells during the vulnerable detachment process.
Protocols for pH Control and Monitoring
- Buffering Systems:
- Bicarbonate/CO₂ System: This is the most common buffering system. The equilibrium between sodium bicarbonate in the medium and CO₂ in the incubator atmosphere (typically 5–10%) maintains pH. It is crucial to ensure the incubator CO₂ concentration is calibrated and stable [76] [74].
- Organic Buffers (e.g., HEPES): Adding 10–25 mM HEPES provides additional buffering capacity, which is especially useful during procedures outside the incubator, such as cell detachment, washing, and counting [74].
- Medium Replacement Strategy: For batch cultures, medium should be replaced every 2–3 days, or more frequently if it turns yellow quickly, to prevent toxic metabolite accumulation and pH drift [73]. In advanced bioreactor systems, a perfusion strategy that continuously replaces spent medium with fresh medium based on pH feedback provides a more homeostatic environment, minimizing stress cycles [76].
- Protocol: Routine pH Monitoring and Adjustment:
- Visual Check: Daily observe the color of the medium using phenol red as an initial guide [73].
- Incubator Calibration: Regularly verify and calibrate the CO₂ and temperature sensors of the incubator.
- Equipment Check: Ensure that the incubator's door is properly sealed and that gas-permeable caps on flasks are not over-tightened.
- Use Pre-warmed Media: Always warm the complete medium to 37°C before use to avoid dissolved gas loss and pH shift upon addition to cells [73].
Selecting and Managing Serum and Supplements
Serum has been the standard supplement for cell culture media, but its use is associated with significant challenges, including batch-to-batch variability, ethical concerns, and risk of contamination. Selecting the right supplement and managing it properly is vital for consistent cell attachment and post-detachment recovery.
Comparative Analysis of Serum Supplements
Table 2: Quantitative Comparison of Culture Media Supplements (Based on [75])
| Supplement Type | Growth Factor Content (IGF-1, PDGF-AB, etc.) | Impact on MSC Growth Kinetics | Cost Consideration | Key Advantages | Key Challenges |
|---|---|---|---|---|---|
| Fetal Bovine Serum (FBS) | Reference Standard | Supports growth | Moderate | Rich, complex mixture; works for many cell types | Ethical concerns; high batch-to-batch variability; risk of zoonoses [75]. |
| Human Platelet Lysate (hPL) | Significant differences between categories, but all supported MSC growth | All tested hPL preparations supported growth | Lower than SFM | Xeno-free; reduced immunogenicity; high growth factor content [75]. | Batch consistency and supply can be challenging [75]. |
| Serum-Free Media (SFM) | Varies by product; some contained human blood components | Some supported growth well, others did not | Significantly higher than hPL | Defined composition; consistent performance; no animal components [75] [74]. | Cell-type specific; can require higher reagent purity; slower growth initially; high cost [75] [74]. |
Best Practices for Serum and Supplement Use
- Avoid Frequent Brand/Batch Changes: The composition of different serum batches and brands can vary. Avoid frequent switches. If a change is necessary, first conduct a trial test. If effective, purchase a large supply (e.g., a 6-month or 1-year stock) to minimize batch-to-batch variability [73].
- Proper Handling and Storage:
- Coating for Weakly Adherent Cells: For cells with inherently weak adhesion (e.g., LNCaP FGC, U-87 MG), coating culture surfaces with poly-L-lysine, gelatin, collagen, or extracellular matrix mixtures can significantly improve initial attachment and stability, preventing pre-detachment detachment and improving post-detachment recovery upon seeding [73].
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key reagents and materials essential for optimizing culture conditions and managing cell detachment.
Table 3: Research Reagent Solutions for Cell Culture and Detachment
| Item | Function/Application | Key Considerations |
|---|---|---|
| Trypsin/EDTA | Proteolytic enzyme/chelator combination for enzymatic cell detachment. | Effective but can damage surface proteins; concentration and incubation time must be optimized per cell line [1] [13]. |
| Accutase | A mild, enzymatic blend of proteases and collagenases for cell detachment. | Considered gentler than trypsin, but has been shown to cleave specific surface proteins like FasL and Fas receptor, requiring a 20-hour recovery period [5]. |
| Non-Enzymatic Cell Dissociation Solution (e.g., Versene) | EDTA-based solution that chelates calcium and magnesium, disrupting integrin-mediated adhesion. | A mild, non-proteolytic method ideal for preserving surface antigens for flow cytometry, but may be insufficient for strongly adherent cells [5]. |
| HEPES Buffer | Organic chemical buffer added to media (10-25 mM) to maintain pH during outside-incubator work. | Crucial for maintaining pH stability during cell detachment and washing steps [74]. |
| Human Platelet Lysate (hPL) | Xeno-free serum supplement rich in growth factors, used at 5-10% (vol/vol). | An effective FBS alternative for clinical-grade cell manufacturing; may require heparin addition to prevent clotting [75]. |
| Poly-L-Lysine | A synthetic polymer used to coat culture surfaces, enhancing cell attachment. | Promotes attachment for weakly adherent cells; available in D and L forms [73]. |
Optimizing culture conditions is not a passive background activity but an active strategy to enhance the resilience of adherent cells through the inherently stressful process of detachment. As the field moves towards automated, large-scale biomanufacturing for cell therapies and regenerative medicine, the principles outlined here—selecting a physiologically appropriate medium, enforcing strict pH control, and making informed, consistent choices about serum and supplements—become increasingly critical. Future advancements will likely involve the integration of real-time, in-line sensors in bioreactors to dynamically adjust these parameters, and the continued development of highly defined, cost-effective, and efficacious serum-free formulations. By viewing pre- and post-detachment culture not as separate phases but as a continuous supportive continuum, researchers can significantly improve data quality, process efficiency, and ultimately, the success of therapeutic cell products.
Appendix: Diagrams and Workflows
Cell Detachment Stress and Recovery Pathways
The following diagram illustrates the key stressors during cell detachment and the cellular pathways activated during recovery, highlighting points where culture condition optimization is critical.
Experimental Workflow for Optimizing Conditions
This workflow provides a methodological guide for systematically testing and validating the impact of different culture conditions on post-detachment cell health.
Comparing Detachment Techniques: Impact on Cell Phenotype and Functional Outcomes
Adherent cell detachment is a critical unit operation in biotechnology and pharmaceutical development, enabling the passaging of cells for scale-up and the harvesting of cellular products. This process directly influences cell viability, yield, phenotypic stability, and the reproducibility of subsequent experiments and production batches [77]. The fundamental challenge lies in disrupting the strong bonds between the cell and its growth surface—often mediated by integrins and cadherins—while minimizing damage to the delicate cell membrane and functionally important surface proteins [77] [5].
Research in this field is increasingly focused on benchmarking traditional and novel methods not merely by the speed of detachment, but by a comprehensive set of Critical Quality Attributes (CQAs) including post-detachment viability, proliferation recovery, the integrity of surface markers, and the overall yield of functional cells. This guide provides a detailed technical framework for conducting such a benchmarking analysis, providing researchers with standardized metrics and methodologies to objectively evaluate detachment performance within the broader context of advancing cell-based research and manufacturing.
Cell Detachment Methodologies: Mechanisms and Trade-offs
The selection of a detachment method is a balance between efficiency and cellular integrity. The following table summarizes the core characteristics, mechanisms, and inherent trade-offs of the primary detachment categories.
Table 1: Comparison of Primary Adherent Cell Detachment Methods
| Method Category | Specific Examples | Mechanism of Action | Key Advantages | Key Limitations & Cellular Impacts |
|---|---|---|---|---|
| Enzymatic | Trypsin/EDTA [1] [78] | Proteolytic cleavage of adhesion proteins. | Robust, effective for strongly adherent cells [77]. | Damages cell surface proteins; requires precise control to avoid irreversible harm [77] [5]. |
| Enzymatic (Milder) | Accutase, TrypLE, Detachin [79] [5] [80] | Blended enzymatic activities (protease, collagenase) in a gentler formulation. | Higher viability for delicate cells; considered a gentler alternative to trypsin [79]. | Can still compromise specific surface proteins (e.g., Fas ligand) requiring a recovery period [5]. |
| Non-Enzymatic (Chelators) | EDTA-based Cell Dissociation Buffer [5] [78] | Chelates Ca²⁺ and Mg²⁺ ions, disrupting integrin-mediated adhesion. | Preserves surface protein integrity; gentle [5]. | Often insufficient for strongly adherent cells; may require mechanical assistance like scraping [5] [78]. |
| Physical/Mechanical | Scraping, Vigorous Pipetting [78] | Physically shears cells from the surface. | Fast; avoids chemical/enzymatic stress. | Can cause significant cell rupture and damage, reducing yield of viable cells [78]. |
| Stimuli-Responsive Surfaces | Thermo-responsive Polymers [77] | Cells detach upon temperature shift, altering surface hydrophobicity. | Extremely gentle; no enzymatic or chemical treatment. | Less robust; requires specialized, expensive cultureware; detachment efficiency can vary [77]. |
Quantitative Benchmarking of Detachment Performance
A rigorous viability and yield analysis requires monitoring key metrics throughout the detachment process. Quantitative data from recent studies highlights the performance differentials between methods.
Impact on Cell Surface Markers and Recovery
Research has demonstrated that the choice of detachment agent can significantly alter the cell surface phenotype. A 2022 study found that accutase treatment significantly decreased the mean fluorescence intensity (MFI) of surface Fas ligand (FasL) and Fas receptor on macrophages compared to EDTA-based solutions, while the surface marker F4/80 remained unaffected [5]. This effect was reversible, but required approximately 20 hours of post-detachment culture for the surface proteins to fully recover. Western blot analysis confirmed that accutase cleaved the extracellular portion of FasL, explaining its transient removal from the cell surface [5]. This underscores the necessity of allowing a recovery period for cells before using them in experiments where surface marker integrity is critical.
Advanced Monitoring of Detachment Kinetics
The process of enzymatic detachment can be quantitatively monitored using lens-free imaging (LFI) to establish robust, non-subjective endpoints. One study defined a novel feature, the "percentage of detached cell regions," which could be automatically extracted from LFI intensity images [80]. Using this method, researchers determined that the optimal time to inhibit the enzymatic reaction was when approximately 92.5% of the cells were detached. The study reported that overconfluent cultures detached much slower, with inhibition times of 16.2–17.2 minutes, compared to 9.6–11.1 minutes for medium- to high-density cultures [80]. This objective approach to monitoring enhances reproducibility compared to manual inspection of cell "rounding-up."
Table 2: Key Metrics for Benchmarking Detachment Performance
| Performance Metric | Description | Benchmarking Method | Typical Target Range |
|---|---|---|---|
| Detachment Time (Kinetics) | Time from reagent addition until ≥90-95% of cells are detached. | Microscopic observation or automated imaging (e.g., LFI) [1] [80]. | Cell-line specific; minimize to reduce stress. |
| Post-Detachment Viability | Percentage of viable cells in the harvested suspension. | Trypan blue exclusion assay via hemocytometer or automated cell counter [1]. | >90% [1] [78]. |
| Functional Yield | Total number of viable cells recovered per cm² of culture surface. | Cell counting post-detachment and viability assessment [1]. | Maximize and ensure consistency between batches. |
| Surface Marker Integrity | Preservation of specific proteins of interest (e.g., FasL, receptors). | Flow cytometry (Mean Fluorescence Intensity) [5]. | Varies by protein; compare to gentle control (e.g., EDTA). |
| Proliferation Recovery | The ability of harvested cells to re-attach and proliferate at a normal rate. | Seeding cells and monitoring doubling time or confluency over 24-72 hours. | Comparable to pre-detachment rates. |
Experimental Protocols for Benchmarking Analysis
To ensure reproducible and comparable results across studies, standardized experimental workflows are essential.
Generalized Protocol for Enzymatic Detachment and Analysis
This protocol, adapted from industry standards, can be applied to benchmark various enzymatic reagents like trypsin, TrypLE, and accutase [1] [78].
- Preparation: Pre-warm complete growth medium, balanced salt solution (e.g., PBS without Ca²⁺/Mg²⁺), and the dissociation reagents to 37°C. Pre-label culture vessels and new recipient vessels with warmed growth medium.
- Initiation: Aspirate and discard the spent medium from the adherent cell culture.
- Wash: Gently rinse the cell layer with the balanced salt solution (approx. 2 mL per 10 cm²) to remove serum and divalent cations that inhibit enzyme activity. Aspirate and discard the wash solution [1].
- Detachment: Add a pre-determined volume of the enzymatic dissociation reagent to cover the cell layer (approx. 0.5 mL per 10 cm²). Gently rock the vessel to ensure complete coverage [1].
- Incubation & Monitoring: Incubate the vessel at room temperature or 37°C. Monitor the detachment process under a microscope approximately every 2 minutes. Tap the vessel gently if needed to expedite detachment [1]. For quantitative benchmarking, use automated imaging systems like LFI to track kinetics [80].
- Termination: Once ≥90% of cells are detached, add at least 2 volumes of complete growth medium (containing serum or inhibitors) to neutralize the enzyme. Gently pipette the medium across the surface to disperse any remaining cells and create a single-cell suspension [1].
- Centrifugation & Analysis: Transfer the cell suspension to a conical tube and centrifuge at 200 x g for 5–10 minutes. Resuspend the cell pellet in a known volume of fresh, pre-warmed growth medium [1].
- Assessment: Remove a sample for cell counting and viability analysis using a hemocytometer or automated cell counter. Calculate the key metrics outlined in Table 2.
Protocol for Assessing Surface Protein Integrity Post-Detachment
This specialized protocol is critical for experiments where the preservation of surface receptors is paramount, such as in immunology or flow cytometry-based assays [5].
- Detach Cells: Harvest cells using the test detachment method (e.g., accutase, trypsin, EDTA) and the standard protocol above.
- Split and Culture: After resuspending the cell pellet in complete medium, split the cell suspension into two parts:
- "Immediate Analysis" Sample: Process these cells immediately for analysis.
- "Recovery" Sample: Seed these cells into a new culture vessel with fresh, pre-warmed complete medium and return them to the incubator for a defined recovery period (e.g., 2, 6, 20 hours).
- Analyze by Flow Cytometry:
- At each time point (immediate and post-recovery), harvest the "Recovery" sample cells using a gentle, non-enzymatic method like an EDTA-based buffer or careful scraping to avoid confounding the results.
- Follow standard staining procedures for the surface markers of interest (e.g., FasL, Fas) and appropriate isotype controls.
- Analyze the cells using a flow cytometer and compare the Mean Fluorescence Intensity (MFI) of the immediate and recovered samples to the control (e.g., cells detached with EDTA-only) to assess the extent of protein cleavage and subsequent recovery [5].
Diagram 1: Experimental Workflow for Benchmarking Cell Detachment Methods
The Scientist's Toolkit: Essential Reagents and Materials
Successful detachment and analysis require a suite of specialized reagents and tools.
Table 3: Essential Research Reagent Solutions for Detachment Benchmarking
| Tool/Reagent Category | Specific Examples | Function & Application Note |
|---|---|---|
| Proteolytic Enzymes | Trypsin-EDTA (0.05-0.25%) [1] [78] | Standard enzyme for robust detachment of common cell lines; requires careful timing. |
| Gentle Enzyme Blends | Accutase [5], TrypLE [80], Detachin [79] | Milder enzymatic activity for sensitive cells (e.g., stem cells, primary cells). Helps maintain high viability. |
| Non-Enzymatic Solutions | EDTA Solution, Cell Dissociation Buffer [5] [78] | Chelates ions to disrupt adhesion. Ideal for preserving surface protein integrity for flow cytometry. |
| Balanced Salt Solutions (BSS) | DPBS, HBSS (without Ca²⁺/Mg²⁺) [1] [81] | Used for washing cells pre-detachment to remove inhibitory serum and ions. |
| Inactivation Media | Complete Growth Medium with Serum [1] [81] | Serum contains inhibitors (e.g., α1-antitrypsin) that neutralize trypsin and other proteases. |
| Viability Assay | Trypan Blue Stain [1] | Dye exclusion method to distinguish live (unstained) from dead (blue) cells for counting. |
| Advanced Monitoring | Lens-free Imaging (LFI) Set-up [80] | Enables quantitative, label-free monitoring of detachment kinetics and objective endpoint determination. |
Critical Considerations for Experimental Design
Beyond selecting a method, several factors are crucial for ensuring reliable and reproducible benchmarking data.
- Cell Line Variability: The optimal detachment method is highly cell-type specific. Strongly adherent cells (e.g., fibroblasts) may require trypsin, while delicate primary cells or neurons may need a gentler alternative like Detachin or a non-enzymatic buffer [79] [82]. Always consult literature or supplier recommendations for your specific cell line.
- Confluency and Culture Age: The density of the culture directly impacts detachment kinetics. Overconfluent cultures have formed more complex cell-cell and cell-matrix interactions, often requiring longer detachment times, as quantified in LFI studies [80]. Conversely, passaging cells at very low density can also be detrimental to health [82].
- Recovery Period for Downstream Assays: As evidenced by research on surface proteins, cells can be stressed immediately after detachment. For any functional assay (e.g., signaling studies, transfection, transplantation), it is critical to allow a post-detachment recovery period of several hours to overnight for cells to re-equilibrate their metabolism and re-synthesize any cleaved surface proteins [5].
- The Scraping Compromise: While mechanical scraping is a quick, chemical-free method, it should be used as a last resort for benchmarking. The physical shearing forces cause significant cell rupture and death, drastically reducing the yield of viable, functional cells and confounding viability data [78].
Diagram 2: Decision Logic for Selecting a Cell Detachment Method
A systematic approach to benchmarking adherent cell detachment methods is fundamental to the integrity of cell-based research and development. By moving beyond simple viability checks to a multi-parametric analysis that includes yield, surface marker integrity, and functional recovery, scientists can make informed, reproducible choices. The growing toolkit—from gentle reagent blends like Detachin to advanced monitoring with lens-free imaging—enables this rigorous analysis. Integrating these methodologies ensures that the detachment process becomes a controlled, optimized step that supports, rather than compromises, the scientific and manufacturing goals of modern biomedicine.
In the field of adherent cell detachment research, the integrity of cell surface markers is paramount for accurate experimental and diagnostic outcomes. Cell surface markers, including receptors, ligands, and glycoproteins, serve as essential identifiers of cell type, state, and function. They are indispensable for flow cytometry, immunophenotyping, single-cell analysis, and the isolation of specific cell populations for therapeutic applications [83]. The process of detaching adherent cells from their substrate, however, presents a significant technical challenge, as the method chosen can profoundly impact the preservation of these critical surface structures. Within the context of a broader thesis on adherent cell detachment, this review synthesizes current evidence quantifying how enzymatic and non-enzymatic dissociation methods affect surface receptor expression, providing a technical guide for researchers and drug development professionals.
The fundamental challenge lies in the fact that the very structures researchers aim to study—proteins on the cell surface—are vulnerable to the techniques used to prepare single-cell suspensions. Enzymatic methods, which use proteases to cleave adhesion proteins, can simultaneously degrade the epitopes recognized by antibodies, leading to false-negative results and a misrepresentation of the cellular landscape [84] [5]. Non-enzymatic alternatives, which typically rely on chelating agents to disrupt calcium-dependent adhesion, offer a gentler approach but may be insufficient for dissociating strongly adherent cell types, potentially requiring mechanical force that can reduce cell viability [85]. Understanding these trade-offs is essential for designing robust and reproducible experiments, particularly in immunology and cancer research where subtle differences in surface marker expression define cellular identity and function.
Comparative Analysis of Detachment Methods
Mechanisms of Action and Cellular Impact
The choice of cell detachment method directly influences the structural integrity of surface markers and overall cell health. The following diagram illustrates the key mechanisms and consequences of different dissociation approaches.
Enzymatic methods work through proteolytic cleavage of cell-surface and cell-matrix proteins. Trypsin, a serine protease, cleaves after lysine or arginine residues, making it a potent dissociating agent but also highly destructive to a broad range of surface proteins [85]. Accutase, a mixture of proteolytic and collagenolytic enzymes, is often considered gentler than trypsin. However, studies show it can specifically cleave certain surface markers. For instance, accutase significantly reduces the surface levels of Fas ligand (FasL) and Fas receptor on macrophages, potentially by cleaving the extracellular domain, while leaving the macrophage marker F4/80 unaffected [5]. This indicates that the impact of enzymatic treatments is highly marker-dependent.
In contrast, non-enzymatic methods like EDTA-based buffers work by chelating calcium and magnesium ions, which are essential for cadherin-mediated cell-cell adhesion and integrin-mediated cell-matrix adhesion [5] [85]. This mechanism avoids proteolytic damage but may be inefficient for strongly adherent cells. Mechanical scraping, another non-enzymatic method, applies direct physical force and has been shown to preserve surface markers like FasL better than enzymatic treatments, but it carries a higher risk of plasma membrane damage and can reduce cell viability [5]. Advanced non-enzymatic techniques, such as acoustic dissociation, use controlled acoustic energy to dissociate tissues without enzymes or elevated temperatures, demonstrating superior preservation of sensitive epitopes like CD19, CD45, and CD138 in immune cells [86].
Quantitative Comparison of Surface Marker Integrity
The following table summarizes quantitative findings from key studies on the effects of various detachment methods on specific surface markers.
Table 1: Impact of Cell Detachment Methods on Surface Marker Expression
| Surface Marker | Cell Type | Detachment Method | Quantified Effect | Reference |
|---|---|---|---|---|
| FasL (Fas Ligand) | RAW264.7 Macrophages | Accutase (30 min) | Significant decrease in MFI* vs. scraping or EDTA | [5] |
| EDTA-based solution (30 min) | Slight decrease in MFI vs. scraping | [5] | ||
| Scraping | Highest level of surface FasL preserved | [5] | ||
| Fas Receptor | RAW264.7 Macrophages | Accutase (30 min) | Significant decrease in MFI | [5] |
| CD55 | Various Cell Lines | Trypsin & Accutase | Significant differences in detection quality vs. other methods | [84] |
| CD19 | Murine Kidney B Cells | Enzymatic (Collagenase/Dispase) | Significantly lower MFI | [86] |
| Acoustic Dissociation | Significantly higher MFI (*p < 0.001) | [86] | ||
| CD138 | Murine Spleen B Cells | Enzymatic Digestion | Marked reduction in MFI | [86] |
| Acoustic Dissociation | Highest MFI preserved | [86] | ||
| CD45 | Murine Spleen Leukocytes | Enzymatic Digestion | Notable reduction in MFI | [86] |
| Acoustic Dissociation | Strong signal intensity preserved | [86] | ||
| Viability | Human Mesenchymal Stem Cells (MSC) | Trypsin | 93.2% ± 3.2% viability | [85] |
| Enzyme-free Dissociation Buffer | 68.7% ± 5.0% viability | [85] |
*MFI: Mean Fluorescence Intensity, a flow cytometry measure of marker abundance.
The data reveals a clear trend: enzymatic methods frequently reduce the detection levels of specific surface markers. The effect is not uniform, as seen with accutase's specific cleavage of FasL/Fas but not F4/80 [5]. This highlights the importance of method validation for each target marker. Furthermore, cell viability can also be affected, with one study showing significantly higher viability for mesenchymal stem cells dissociated with trypsin compared to an enzyme-free chelating buffer [85]. This suggests a trade-off between surface marker preservation and overall cell health, which must be considered based on the downstream application.
Experimental Protocols for Assessing Marker Integrity
Flow Cytometry-Based Evaluation Protocol
A standard method for quantifying surface marker integrity post-detachment is flow cytometry. The following workflow outlines a typical experiment comparing different detachment methods.
Detailed Protocol:
Cell Culture and Harvesting: Seed cells in multi-well plates and allow them to reach approximately 80% confluence [84]. Wash the monolayers with phosphate-buffered saline (PBS) to remove serum and dead cells. Apply different pre-warmed detachment methods (e.g., trypsin, accutase, EDTA-based buffer) to parallel wells for controlled time intervals (e.g., 5-15 minutes for trypsin, 15-30 minutes for accutase and EDTA) at 37°C [5] [85]. Gently pipette periodically to aid dissociation. For mechanical detachment, use a rubber or plastic scraper [84] [5].
Cell Processing and Staining: Neutralize enzymatic activity using serum-containing medium. For non-enzymatic methods, centrifugation is sufficient. Collect the cell suspension and centrifuge (e.g., 200-500 × g for 5-10 minutes) [84] [85]. Resuspend the pellet in a staining buffer (e.g., PBS with 0.1% Tween20 and 3% BSA) and incubate with fluorescently-labeled antibodies targeting the surface markers of interest (e.g., anti-CD55, anti-FasL) for 45 minutes at 4°C in the dark [84] [5]. Include isotype controls for background subtraction.
Data Acquisition and Analysis: Analyze the stained cells using a flow cytometer. Gate on live cells based on forward/side scatter properties and exclude debris [84]. The key metric for surface marker integrity is the Median Fluorescence Intensity (MFI). A significant reduction in MFI in one treatment group compared to others (e.g., scraping or acoustic dissociation) indicates loss or damage of the surface epitope due to the detachment method [5] [86].
Recovery and Functional Assay Protocol
Some surface proteins can recover after enzymatic cleavage if cells are given time to regenerate them. The following protocol assesses this recovery and the functional viability of cells post-detachment.
Detailed Protocol:
Detachment and Recovery Culture: Detach cells using the method of interest (e.g., accutase for 30 minutes). After washing, reseed the dissociated cells in complete culture medium and maintain them under standard culture conditions [5].
Time-Course Analysis: Harvest cells at various time points post-seeding (e.g., 0, 2, 8, 20 hours) using a gentle, non-enzymatic method like an EDTA-based buffer to avoid re-cleaving the markers. Then, process and stain the cells for flow cytometry as described in Section 3.1 to track the return of MFI over time [5].
Functional Viability Assessment (MTT Assay): To assess the ability of dissociated cells to reattach and metabolically function, seed a known number of newly dissociated cells into a new culture dish. After 24 hours, wash off unattached cells and perform an MTT assay. This measures the metabolic activity of the reattached, viable cells. The absorbance values can be compared to a non-dissociated control to calculate the percentage of viable cells that successfully reattached [85].
The Scientist's Toolkit: Essential Reagents and Solutions
Table 2: Key Reagents for Cell Detachment and Surface Marker Analysis
| Reagent/Solution | Primary Function | Key Considerations |
|---|---|---|
| Trypsin-EDTA | Serine protease that cleaves adhesion proteins. EDTA chelates cations to weaken adhesions. | Potent and fast-acting. Can broadly degrade surface proteins and reduce cell viability if over-exposed. |
| Accutase | A mixture of proteolytic and collagenolytic enzymes. | Often considered gentler than trypsin, but has been shown to specifically cleave markers like FasL and Fas [5]. |
| EDTA-based Buffer | A non-enzymatic, isotonic solution that chelates Ca²⁺ and Mg²⁺. | Preserves surface epitopes but may be slow and inefficient for strongly adherent cells. Can reduce reattachment viability [85]. |
| Recombinant Trypsin | A recombinant fungal protease with trypsin-like activity. | Animal-origin free, reducing pathogen transmission risk. Activity and specificity similar to porcine trypsin. |
| Fluorochrome-conjugated Antibodies | Bind specifically to surface epitopes for detection by flow cytometry. | Antibody clones should be validated for the specific application. Tandem dyes may be sensitive to enzyme activity. |
| Annexin V Binding Buffer | Provides appropriate Ca²⁺ concentration for Annexin V to bind to phosphatidylserine. | Critical for apoptosis assays post-detachment, as enzymes can cause transient phosphatidylserine exposure [84]. |
The selection of a cell detachment method is a critical experimental variable that directly influences data fidelity in surface marker analyses. Evidence consistently shows that enzymatic methods, while efficient, carry a high risk of cleaving specific surface receptors, leading to inaccurate phenotyping and potentially flawed conclusions. Non-enzymatic methods, particularly advanced techniques like acoustic dissociation, demonstrate superior preservation of epitope integrity.
To ensure reliable results, researchers should:
- Pilot Multiple Methods: Test enzymatic and non-enzymatic methods for new cell types or markers.
- Use the Gentlest Effective Method: Base the choice on the specific marker and downstream application.
- Allow Recovery Time: For enzymatically-dissociated cells used in functional assays, allow 20 hours for surface protein recovery [5].
- Report Methodology in Detail: Clearly state the detachment protocol, including enzyme type, concentration, and incubation time, to ensure experimental reproducibility.
By adopting a validated, evidence-based approach to cell dissociation, researchers can minimize artifacts and ensure their data truly reflects the biological reality of the cell surface landscape.
Adherent cell detachment is a fundamental yet critical step in cell culture processes, with profound functional consequences for downstream applications in therapy, regenerative medicine, and drug screening. Traditional enzymatic detachment methods, while widely used, present significant challenges that can compromise cell integrity and functionality. These methods can damage delicate cell membranes and surface proteins, particularly in primary cells, and often require multiple steps that create slow, labor-intensive workflows [4]. Furthermore, enzymatic treatments relying on animal-derived components introduce compatibility concerns for human therapies and generate substantial biological waste, estimated at 300 million liters of cell culture waste annually [4]. The limitations of conventional approaches have driven innovation in cell detachment technologies, with emerging methods focusing on preserving cell viability, functionality, and molecular integrity throughout the detachment process. This technical guide examines the functional consequences of cell detachment methodologies within the broader context of adherent cell detachment research, providing researchers with advanced protocols and analytical frameworks for optimizing downstream application outcomes across biomedical domains.
Consequences of Conventional Cell Detachment Methods
Impact on Cell Viability and Functionality
The functional integrity of detached cells is paramount for successful downstream applications. Conventional enzymatic detachment methods, particularly those utilizing trypsin, can proteolytically cleave not only adhesion proteins but also functionally critical surface markers and receptors. This non-specific proteolytic activity can alter cell signaling capabilities, migration potential, and differentiation capacity—attributes essential for therapeutic efficacy and predictive drug screening. Research indicates that enzymatic treatments can reduce cell viability and proliferation rates post-detachment, directly impacting their performance in regenerative applications [4] [87]. The mechanical stress associated with supplemental scraping procedures exacerbates these issues, further compromising membrane integrity and increasing apoptotic signaling in sensitive cell populations.
Implications for Specific Downstream Applications
Cell Therapy and Regenerative Medicine
In cell therapy and regenerative medicine, maintaining cell potency and differentiation potential is essential. Mesenchymal stem cells (MSCs) subjected to prolonged enzymatic detachment exhibit reduced multilineage differentiation capacity and altered secretion of paracrine factors that mediate their therapeutic effects [88]. For induced pluripotent stem cell (iPSC)-derived therapies, preserving surface markers is crucial for accurate characterization and predictable in vivo behavior. Current research highlights how enzymatic damage to surface proteins can impair homing and engraftment efficiencies when cells are administered therapeutically [89] [88].
Drug Screening and Toxicity Testing
In drug screening and toxicity testing, the physiological relevance of cell-based assays depends heavily on the preservation of native cell states. Enzymatic detachment can artificially alter receptor densities and signaling pathways, potentially leading to false positives or negatives in compound screening [90]. Research utilizing advanced models like tissue chips and organoids has demonstrated that cells recovered using harsh detachment methods show compromised barrier function, polarized secretion, and transporter expression—critical features for accurate pharmacokinetic and toxicological assessment [90] [91].
Table 1: Functional Consequences of Conventional Enzymatic Detachment Methods on Downstream Applications
| Downstream Application | Impact on Cell Viability | Functional Consequences | Long-term Effects |
|---|---|---|---|
| Cell Therapy | Viability reductions of 15-25% in sensitive primary cells [4] | Impaired homing and engraftment; Altered differentiation potential [88] | Reduced therapeutic efficacy in clinical applications [89] |
| Regenerative Medicine | Viability maintained but functionality compromised | Disrupted secretion of trophic factors; Modified extracellular matrix production [88] | Limited tissue integration and regenerative capacity |
| Drug Screening | Standard cell lines show minimal viability impact | Altered receptor signaling; Compromised barrier function [90] [91] | Reduced predictive accuracy for in vivo responses |
| Bioprocessing | Variable impacts based on cell type | Reduced growth kinetics and product yields [4] | Inconsistent manufacturing outcomes |
Advanced Solutions: Enzyme-Free Detachment Technologies
Electrochemical Cell Detachment Platform
Recent innovation has yielded a novel enzyme-free strategy for detaching cells from culture surfaces using alternating electrochemical current on a conductive biocompatible polymer nanocomposite surface [4]. This approach applies low-frequency alternating voltage to disrupt adhesion while maintaining over 90% cell viability, effectively overcoming the limitations of enzymatic and mechanical methods [4]. The platform operates through controlled electrochemical redox cycling at the biointerface, enabling rapid detachment (within minutes) without the damage associated with proteolytic enzymes. This technology has demonstrated remarkable efficiency across multiple cell types, with detachment efficiency increasing from 1% to 95% for human cancer cells including osteosarcoma and ovarian cancer, while simultaneously preserving viability exceeding 90% [4].
The electrochemical mechanism involves creating a dynamically tunable ionic microenvironment that gently disrupts cell-surface interactions without damaging membrane integrity or surface proteins. This preservation of cellular components is particularly valuable for applications requiring precise surface marker presentation, such as immune cell therapies and receptor signaling studies. Additionally, as this method utilizes defined electrical parameters rather than biological reagents, it eliminates batch-to-batch variability associated with enzyme preparations, enhancing experimental reproducibility and manufacturing consistency [4].
Experimental Protocol: Electrochemical Cell Detachment
Materials Required:
- Conductive biocompatible polymer nanocomposite culture surfaces
- Programmable electrochemical station with alternating current capability
- Sterile electrolyte solution (physiologically compatible)
- Cell culture reagents standard for the cell type being cultured
- Environmental chamber maintaining 37°C and 5% CO₂
- Viability assessment reagents (e.g., trypan blue, calcein AM/ethidium homodimer)
Methodology:
- Culture adherent cells to 70-80% confluence on specialized conductive polymer nanocomposite surfaces under standard conditions.
- Replace culture medium with sterile, temperature-equilibrated electrolyte solution optimized for electrochemical compatibility.
- Program electrochemical station to deliver low-frequency alternating voltage (optimal frequency determined empirically for specific cell type, typically 1-100 Hz range).
- Apply alternating current for predetermined duration (typically 2-10 minutes) while maintaining environmental conditions at 37°C.
- Monitor detachment progress microscopically; gentle agitation may be applied after initial detachment period to facilitate complete release.
- Transfer cell suspension to collection vessel and neutralize electrochemical activity through dilution with complete culture medium.
- Quantify detachment efficiency: (Number of detached cells / Total number of cells) × 100%.
- Assess cell viability using standardized methodology (e.g., flow cytometry with viability dyes, automated cell counting with trypan blue exclusion).
- Proceed to downstream applications with detached cells.
Validation Metrics:
- Detachment efficiency should exceed 90% for most applications
- Post-detachment viability should maintain >90% of pre-detachment levels
- Functional assessment specific to downstream application (e.g., differentiation potential, surface marker expression, drug response)
Diagram 1: Electrochemical Detachment Workflow. This protocol enables high-efficiency cell detachment while preserving viability and functionality.
Application-Specific Validation Frameworks
Validation for Regenerative Medicine Applications
The functional validation of detached cells for regenerative medicine requires comprehensive assessment of differentiation potential, paracrine signaling capacity, and in vivo functional integration. For mesenchymal stem cells (MSCs), this involves multilineage differentiation assays (osteogenic, chondrogenic, adipogenic) following detachment, comparing the extent and quality of differentiation to undetached controls [88]. For iPSC-derived therapies, validation should include teratoma formation assays to confirm maintained pluripotency or directed differentiation efficiency toward target lineages such as dopaminergic neurons for Parkinson's disease treatment [89] [92].
Recent advances in stem cell research have identified key signaling pathways critical for maintaining regenerative potential, including Notch, Wnt, and Hedgehog pathways [92]. Post-detachment validation should confirm that cells retain appropriate responsiveness to these developmental cues. For example, the Notch signaling pathway regulates cell development, organogenesis, and tissue homeostasis through interactions between Notch receptors (Notch1–4 in mammals) and their ligands (Jagged1, Jagged2, Delta-like ligands) [92]. Preservation of these signaling capabilities indicates minimal detachment-induced functional impairment.
Table 2: Key Signaling Pathways and Functional Markers for Validating Regenerative Potential Post-Detachment
| Cell Type | Critical Signaling Pathways | Functional Differentiation Markers | In Vivo Functional Assays |
|---|---|---|---|
| Mesenchymal Stem Cells (MSCs) | Notch, Wnt/β-catenin, BMP [92] | Osteocalcin (osteogenic), Aggrecan (chondrogenic), PPARγ (adipogenic) [88] | Bone defect repair, Cartilage formation in vivo models [88] |
| iPSC-Derived Neural Progenitors | Notch, Hedgehog, TGF-β [92] | Tyrosine hydroxylase, β-III-tubulin, MAP2 [89] | Functional recovery in Parkinson's or spinal cord injury models [89] |
| iPSC-Derived Cardiomyocytes | Wnt, BMP, FGF [92] | Cardiac troponin T, α-actinin, Connexin 43 [92] | Electrophysiological function, Engraftment in myocardial infarction models |
| Hematopoietic Stem Cells (HSCs) | Notch, CXCR4/SDF-1 [88] | CD34, CD45, CD133 [89] | Bone marrow reconstitution in immunodeficient models [89] |
Validation for Drug Screening Applications
With the FDA's recent movement toward reducing animal testing requirements for drug development, particularly for monoclonal antibodies and other therapeutics, advanced in vitro models have gained increased importance in the drug development pipeline [90] [91]. The validation of cells for drug screening applications must confirm preservation of native physiological responses, including receptor signaling, transporter activity, and metabolic functions.
For organoid-based screening platforms, which more faithfully recapitulate the phenotypic and genetic features of original tumors [91], post-detachment validation should include:
- Phenotypic stability: Confirmation that detachment and re-aggregation processes maintain original tissue architecture and cellular composition.
- Drug response profiling: Benchmarking against established response patterns to standard therapeutics.
- Barrier function: For epithelial organoids, confirmation that tight junctions and polarized functions remain intact.
- Transcriptomic consistency: RNA sequencing to verify that detachment doesn't induce stress-related gene expression patterns that might alter drug sensitivity.
The growing implementation of tissue chips and microphysiological systems for drug screening adds another dimension to validation, requiring confirmation that detached cells can successfully integrate into these complex microenvironments and establish appropriate cell-cell and cell-matrix interactions [90].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Platforms for Advanced Cell Detachment and Downstream Applications
| Reagent/Platform | Function | Application Context |
|---|---|---|
| Conductive Polymer Nanocomposite Surfaces | Provides electrochemical interface for enzyme-free detachment [4] | Large-scale biomanufacturing, automated cell culture systems |
| StemRNA Clinical Seed iPSC Clones | Standardized, clinically-compliant iPSC starting materials [89] | Regenerative medicine, cell therapy development |
| Organoid Culture Matrices | 3D extracellular matrix environment for complex tissue modeling [91] | Drug screening, disease modeling, personalized medicine |
| Tissue Chip Platforms | Microfluidic devices mimicking human organ physiology [90] | Predictive drug toxicity and efficacy testing |
| Defined Non-Enzymatic Detachment Solutions | Chemical disruption of cell adhesion without proteolytic activity [87] | Routine cell culture, therapeutic cell harvesting |
| PDX-Derived Cell Lines and Organoids | Clinically relevant models maintaining patient tumor characteristics [91] | Oncology drug discovery, biomarker identification |
Regulatory and Commercial Translation Considerations
The translation of advanced cell detachment technologies from research to clinical and commercial applications requires careful attention to regulatory frameworks and manufacturing standards. Recent FDA approvals of cell-based therapies, including Ryoncil (remestemcel-L) as the first MSC therapy for pediatric steroid-refractory acute graft-versus-host disease (December 2024) and Omisirge (omidubicel-onlv) for hematologic malignancies (April 2023), demonstrate the evolving regulatory landscape for cell therapies [89]. These approvals highlight the importance of maintaining consistent cell quality and functionality throughout manufacturing processes, including detachment.
For regenerative medicine applications, the preservation of critical quality attributes (CQAs) during cell detachment is essential for regulatory compliance. The FDA's expedited designation programs, including Regenerative Medicine Advanced Therapy (RMAT) and Fast Track, often require comprehensive process validation data, including demonstration that detachment methods don't alter product characteristics [89]. Similarly, in drug screening, the qualification of novel models for regulatory decision-making requires rigorous demonstration of reproducibility and predictive value, which can be compromised by variable cell detachment methodologies.
Commercial translation of advanced detachment platforms must address scalability and integration with closed-system biomanufacturing platforms. The electrochemical detachment approach shows particular promise for automated, high-throughput applications due to its compatibility with programmable, standardized protocols that minimize operator-dependent variability [4]. As the field moves toward increasingly personalized medicine approaches, the ability to gently and reliably process patient-specific cells at various scales will be increasingly valuable across therapeutic and diagnostic applications.
Cell detachment methodology significantly influences functional outcomes across downstream applications in therapy, regenerative medicine, and drug screening. Traditional enzymatic approaches present substantial limitations for advanced applications where preservation of surface markers, signaling capabilities, and differentiation potential is essential. Emerging technologies, particularly electrochemical detachment platforms, offer promising alternatives that maintain high viability and functionality while enabling scalability and reproducibility. As regenerative medicine and sophisticated in vitro models continue to advance, optimizing cell detachment processes will remain a critical component of successful technology translation. Researchers should select detachment methods based on comprehensive validation data specific to their application requirements, with particular attention to preserving those cellular attributes most relevant to their intended downstream use.
Cost-Benefit and Scalability Analysis for Research vs. Clinical Manufacturing
This technical guide provides a comprehensive analysis of adherent cell detachment methodologies, evaluating their cost-benefit and scalability from research-scale operations to clinical manufacturing. Adherent cell culture is a cornerstone of biomedical research and bioproduction, essential for applications ranging from basic cell biology to advanced cell therapies and cultured meat production [93]. The process of detaching cells from growth surfaces presents significant technical and economic challenges that vary considerably between small-scale research and large-scale industrial production. This analysis synthesizes current technologies, quantitative economic data, and implementation protocols to inform strategic decision-making for researchers, scientists, and drug development professionals working within the broader context of adherent cell detachment research.
Adherent cell culture involves the propagation of anchorage-dependent cell types that require physical attachment to a substrate for proliferation and survival [93]. This category includes many therapeutically relevant primary cells and continuous cell lines such as mesenchymal stem cells (MSCs), induced pluripotent stem cells (iPSCs), human embryonic kidney cells (HEK293), and Chinese hamster ovary (CHO) cells [93]. The fundamental challenge in scaling adherent cell processes lies in the inherent limitation of surface area, which necessitates sophisticated detachment protocols to maintain cell viability and functionality across different production scales [94].
The cell detachment process must balance efficiency with preservation of cellular integrity. As cells adhere to surfaces through complex interactions involving extracellular matrix (ECM) proteins and cell adhesion molecules, disrupting these connections requires methods that can effectively release cells while minimizing damage to surface proteins and cell membranes [13]. In the context of a broader thesis on adherent cell detachment research, understanding these fundamental biological interactions provides the foundation for evaluating the technical and economic trade-offs between different detachment methodologies at various scales.
Core Technologies for Adherent Cell Detachment
Chemical and Enzymatic Methods
Enzymatic detachment relies on proteolytic enzymes to cleave adhesion proteins. Trypsin, the most widely used enzyme, cleaves peptides after lysine or arginine residues but can damage critical surface proteins and affect cell functionality [13] [5]. Trypsin is often used with EDTA, a chelating agent that binds calcium ions essential for integrin-mediated adhesion [13]. Accutase, a milder enzymatic blend, is considered gentler on surface proteins but has been shown to specifically cleave certain surface markers like Fas ligands and Fas receptors, requiring approximately 20 hours for recovery of surface protein expression [5].
Non-enzymatic chemical methods utilize chelating agents like EDTA alone or in specialized buffer formulations. These work by sequestering divalent cations necessary for cell adhesion, potentially causing less damage to surface proteins than enzymatic methods [5]. However, they may be insufficient for strongly adherent cell types and often require mechanical assistance, which can introduce variability and reduce viability [5].
Physical and Emerging Methods
Physical methods include mechanical scraping, which preserves surface proteins effectively but risks cellular damage through shear forces [5]. Temperature-responsive surfaces enable detachment by changing the hydrophobicity of culture surfaces when temperature shifts, though this requires specialized culture vessels with precise thermal control [13].
Advanced emerging methods include electrochemical approaches, such as the application of alternating current on conductive polymer nanocomposite surfaces, which has demonstrated 95% detachment efficiency while maintaining over 90% cell viability without enzymatic treatment [4]. This method disrupts cell adhesion by modulating the ionic microenvironment and represents a promising approach for scalable, automated biomanufacturing with reduced consumable waste [4].
Quantitative Analysis of Detachment Methods
Table 1: Comparative Analysis of Cell Detachment Methods
| Method | Detachment Efficiency | Cell Viability | Impact on Surface Proteins | Recovery Time | Relative Cost |
|---|---|---|---|---|---|
| Trypsin-EDTA | High (90-95%) | Medium-High (85-90%) | High degradation | 4-8 hours | Low |
| Accutase | High (90-95%) | High (90-95%) | Selective cleavage (FasL/Fas) | ~20 hours | Medium |
| EDTA-Based | Medium (70-85%) | High (90-95%) | Minimal effect | 0-2 hours | Low |
| Scraping | Variable (60-90%) | Low-Medium (70-85%) | Minimal effect | 0-2 hours | Very Low |
| Electrochemical | High (95%) | High (>90%) | Theoretical minimal effect | Under investigation | High initial, lower operational |
Table 2: Scalability and Resource Requirements
| Method | Research Scale Suitability | Clinical Manufacturing Scalability | Automation Potential | Process Development Complexity | Regulatory Considerations |
|---|---|---|---|---|---|
| Trypsin-EDTA | Excellent | Limited (enzymatic residuals, animal origin) | Moderate | Low | Higher (safety, consistency) |
| Accutase | Excellent | Moderate (defined formulation) | Moderate | Low | Medium |
| EDTA-Based | Good for less adherent cells | Limited (efficiency constraints) | Moderate | Low | Lower |
| Scraping | Good for small scales | Poor (open system, variability) | Low | Low | Higher (validation challenges) |
| Electrochemical | Requires specialized equipment | Excellent (closed system, automation) | High | High | Promising (defined process) |
Scalability Analysis: Research to Clinical Manufacturing
Research-Scale Considerations
At the research scale, operational priorities favor flexibility, minimal capital investment, and technical accessibility. Standard tissue culture flasks, multiwell plates, and basic dissociation reagents dominate research environments due to their low upfront costs and ease of use [1] [95]. The research-scale workflow typically involves:
- Culture Monitoring: Visual assessment of confluency using microscopy [95]
- Medium Removal: Aspiration of spent culture medium [1]
- Rinsing: Application of balanced salt solution to remove serum residues [1]
- Detachment Reagent Application: Incubation with enzymatic or chemical detachment solutions [1]
- Reagent Neutralization: Dilution with serum-containing medium [95]
- Cell Collection: Centrifugation and resuspension for subsequent applications [1]
The economic considerations at this scale prioritize reagent costs over labor, with researchers typically managing multiple cell lines simultaneously in low-throughput formats.
Clinical Manufacturing Requirements
Transitioning to clinical manufacturing introduces stringent requirements for reproducibility, closed processing, regulatory compliance, and cost-effective scaling. The limitations of planar culture systems become prohibitive at commercial scales, where the surface area requirements for producing 10-100 kg of cultured meat, for example, would necessitate 10¹²-10¹³ cells [94]. Scale-up challenges for adherent cells include:
- Surface Area Limitations: Traditional multilayer vessels like cell factories and roller bottles require significant physical space and manual handling [96]
- Process Control Needs: Maintaining consistent temperature, pH, dissolved oxygen, and nutrient levels across large volumes [94]
- Shear Stress Management: Protecting cells from damage in agitated systems [94]
- Harvesting Efficiency: Effectively detaching cells from microcarriers or other substrates at scale [13]
Advanced technologies for manufacturing scale include fixed-bed bioreactors, microcarrier-based systems in stirred-tank reactors, and innovative approaches like the Tide Motion bioreactor with macroporous carriers that provide high surface-to-volume ratios and low shear stress [96].
Detailed Experimental Protocols
Standard Enzymatic Detachment Protocol
This protocol describes the widely used method for detaching adherent cells using trypsin-EDTA, suitable for research-scale applications with common cell lines [1] [95]:
Pre-harvest Assessment: Confirm cells are in log phase growth with >90% viability and approximately 80-90% confluency using microscopic examination [1] [95].
Media Removal: Aspirate and discard spent culture media completely from the culture vessel [1].
Rinsing Step: Wash cell layer with a balanced salt solution without calcium and magnesium (e.g., PBS) using approximately 2 mL per 10 cm² of culture surface. Gently rock the vessel to ensure complete coverage, then remove and discard the wash solution [1]. This critical step removes residual serum that would inhibit trypsin activity.
Enzyme Application: Add pre-warmed trypsin-EDTA solution (0.5 mL per 10 cm²) to cover the cell layer completely. Gently tilt the vessel to ensure even distribution [1].
Incubation: Allow the culture vessel to sit at room temperature for approximately 2 minutes (time varies by cell line). Observe cells under microscope periodically until ≥90% of cells appear rounded and partially detached (typically 5-15 minutes total) [1].
Neutralization: When detachment is sufficient, add complete growth medium with serum at approximately 2x the volume of trypsin used. Pipette medium over the cell layer surface several times to ensure complete cell detachment and enzyme neutralization [1].
Cell Collection: Transfer cell suspension to a centrifuge tube and pellet cells at 200 × g for 5-10 minutes. Resuspend in fresh growth medium for counting and subsequent use [1].
Electrochemical Detachment Protocol
This emerging protocol describes the enzyme-free electrochemical method based on recent research, suitable for sensitive cells and scalable manufacturing applications [4]:
Surface Preparation: Culture cells on conductive biocompatible polymer nanocomposite surfaces designed for electrochemical applications.
System Setup: Assemble the electrochemical cell system with appropriate electrodes and media circulation compatible with bioreactor configurations.
Parameter Application: Apply low-frequency alternating voltage at optimized parameters (specific frequency and voltage settings determined by cell type).
Process Monitoring: Monitor detachment progress microscopically, typically requiring several minutes for complete detachment.
Cell Collection: Transfer released cells to collection vessels without enzymatic neutralization requirements.
Surface Regeneration: Clean and recondition conductive surfaces for subsequent culture cycles.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Research Reagent Solutions for Adherent Cell Detachment
| Reagent/Material | Function | Application Context |
|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme cleaves adhesion proteins; EDTA chelates calcium | General purpose detachment for robust cell lines |
| Accutase | Blend of proteolytic and collagenolytic enzymes | Gentle detachment for sensitive cells and stem cells |
| Collagenase | Enzyme targeting collagenous adhesion proteins | Tissues and cells with strong collagen-mediated adhesion |
| Non-enzymatic dissociation buffers | Chelating agents without proteolytic activity | Surface protein preservation for flow cytometry |
| Temperature-responsive surfaces | Polymer surfaces that change properties with temperature | Non-invasive harvest for delicate primary cells |
| Conductive polymer nanocomposites | Electrochemical disruption of adhesion | Scalable, automated manufacturing processes |
Economic Analysis and Future Directions
The economic model for adherent cell detachment evolves significantly across scales. Research-scale economics focus primarily on reagent costs, with enzymatic detachment reagents representing minimal operational expenses. In clinical manufacturing, the economic considerations expand to include capital equipment, validation costs, labor, quality control, and the substantial impact on overall process efficiency [97] [94].
Current research focuses on reducing costs through innovation, particularly in the cultured meat industry where production costs have decreased from $2.3 million/kg for the first cultured beef burger to approximately $63/kg, with further reductions anticipated through optimized bioreactor systems that could lower costs to $1.95/kg [97]. Key innovation areas include:
- Serum-free media development to eliminate expensive and variable fetal bovine serum [97]
- Advanced bioreactor design with improved process control and scaling capabilities [97] [94]
- Non-enzymatic detachment methods that reduce reagent costs and improve cell quality [13] [4]
- Integrated continuous processing that minimizes downtime and manual intervention [94]
Future directions in adherent cell detachment research point toward increasingly automated, closed-system approaches that enable seamless scaling from research to clinical manufacturing while maintaining cell quality and process economics. The electrochemical detachment method represents a promising approach that addresses several limitations of current enzymatic methods, particularly for cell therapy applications where preserving native cell phenotypes is critical [4].
Visualizations
Adherent Cell Detachment Workflow
Technology Evolution Pathway
In adherent cell culture, the method chosen for detaching cells prior to analysis is a critical experimental variable. This case study examines the specific effects of two common detachment agents—the enzymatic solution Accutase and the non-enzymatic chelator EDTA—on the cell surface expression of Fas receptor (Fas) and Fas ligand (FasL). Findings demonstrate that while Accutase maintains high cell viability, it significantly and reversibly reduces the surface levels of Fas and FasL through proteolytic cleavage compared to EDTA-based methods. These results underscore the necessity of selecting a detachment protocol that is compatible with the specific surface markers under investigation to ensure experimental accuracy.
The study of cell surface proteins is a cornerstone of biomedical research, with implications for understanding immune function, cancer biology, and therapeutic development. For adherent cells, a necessary first step in many analytical workflows, such as flow cytometry, is the detachment of cells from their culture surface. This process, however, is not benign. Cell detachment strategies are essential for cell harvest and are usually facilitated by mechanical methods combined with a calcium chelator or enzymatic cleavage of adhesion proteins to achieve effective detachment while maintaining cell viability [5]. The choice of detachment method can profoundly influence the integrity of the cell surface proteome [13].
Trypsinization is frequently used for cellular dissociation but degrades most surface proteins and the extracellular matrix [5]. Consequently, milder alternatives like Accutase, a blend of proteolytic and collagenolytic enzymes, are often recommended to avoid cellular damage [5]. Non-enzymatic methods, such as the calcium chelator Ethylenediaminetetraacetic acid (EDTA), offer a different mechanism by disrupting integrin-mediated adhesion without proteolytic activity [5] [13]. The Fas/FasL pathway is a critical mediator of apoptosis and immune cytotoxicity. This case study, framed within broader adherent cell detachment research, presents a quantitative investigation into how Accutase and EDTA-based methods impact the surface expression of these functionally significant proteins, providing a methodological guide for researchers and drug development professionals.
Experimental Findings and Data Analysis
Quantitative Impact on Surface Protein Expression
The core experimental data comparing the effects of Accutase and an EDTA-based solution (Versene) on murine macrophage cell lines (RAW264.7 and J774A.1) revealed significant and specific alterations in surface marker expression. The following table summarizes the key quantitative findings from flow cytometry analysis:
Table 1: Summary of Flow Cytometry Results on Surface Marker Expression after Cell Detachment
| Surface Marker | Detachment Method | Effect on Mean Fluorescence Intensity (MFI) | Statistical Significance (p-value) | Implication |
|---|---|---|---|---|
| Fas Ligand (FasL) | Accutase | Significant Decrease | p < 0.001 | Marker is compromised |
| EDTA-based Solution | Minimal Change | Not Significant (vs. Scraping) | Marker is preserved | |
| Scraping (Control) | Highest Level Preserved | Baseline | Optimal preservation | |
| Fas Receptor (Fas) | Accutase | Significant Decrease | p < 0.001 | Marker is compromised |
| EDTA-based Solution | Minimal Change | Not Significant (vs. Scraping) | Marker is preserved | |
| F4/80 (Macrophage Marker) | Accutase | No Significant Change | Not Significant | Marker is unaffected |
| EDTA-based Solution | No Significant Change | Not Significant | Marker is unaffected |
The data show that the negative effect of Accutase was specific to Fas and FasL, as the surface levels of the murine macrophage-specific marker F4/80 remained unaltered [5]. Furthermore, the reduction in MFI was dependent on the duration of Accutase exposure, with a 10-minute incubation sufficient to cause a significant decrease compared to both scraping and EDTA-based methods [5].
Reversibility of Accutase-Induced Effects
A critical finding of this study was that the cleavage of surface proteins by Accutase is not permanent. The researchers treated macrophages with Accutase for 30 minutes and then allowed the cells to recover in a complete culture medium. The surface expression of both FasL and Fas was monitored over time.
Table 2: Recovery Timeline of Surface Protein Expression Post-Accutase Treatment
| Recovery Time | FasL Surface Expression | Fas Receptor Surface Expression | F4/80 Surface Expression |
|---|---|---|---|
| 0 hours (Post-detachment) | Significantly Decreased | Significantly Decreased | Unchanged |
| 2 hours | Low | Low | Unchanged |
| 20 hours | Restored to Normal Levels | Restored to Normal Levels | Unchanged |
The signal intensities for surface FasL and Fas increased over the recovery period, reaching normal levels after approximately 20 hours. This demonstrates that the effects are reversible and that cells require an adequate recovery period post-detachment to regenerate the cleaved surface proteins [5].
Detailed Experimental Protocols
To ensure reproducibility, the key methodologies from the cited research are outlined below.
Cell Culture and Detachment
- Cell Lines: The study used murine macrophage-like cell lines RAW264.7 and J774A.1 [5].
- Detachment Reagents:
- Accutase: A commercial preparation containing proteolytic and collagenolytic enzymes.
- EDTA-based Solution: Versene solution (from Thermo Fisher Scientific), a non-enzymatic, EDTA-based cell dissociation buffer.
- Mechanical Detachment: Cell scraping was used as a control method to maximize surface protein preservation.
- Detachment Procedure: Cells were incubated with the respective detachment solutions according to the manufacturers' instructions, typically for 10 to 30 minutes at 37°C. The reaction was stopped with complete culture medium, and the cells were centrifuged and prepared for subsequent analysis [5].
Flow Cytometry Analysis
- Staining: Detached cells were stained with fluorescently labeled antibodies specific for FasL, Fas receptor, and F4/80.
- Analysis: Cell staining was analyzed using a flow cytometer. The Mean Fluorescence Intensity (MFI) was quantified for each marker and compared across the different detachment method groups. Statistical analysis (ANOVA) was performed to determine significance [5].
Western Blot and Immunofluorescence
- Western Blot: Cell lysates and supernatants from Accutase- and EDTA-treated cells were collected. Proteins were separated by gel electrophoresis, transferred to a membrane, and probed with an antibody recognizing the extracellular portion of FasL. This technique was used to detect cleavage fragments [5].
- Immunofluorescence: Detached cells were adhered to slides, fixed, and stained with an anti-FasL antibody and a fluorescent marker (e.g., for F-actin). Cells were visualized by fluorescence microscopy to confirm the localization of FasL on the cell membrane [5].
Diagram 1: Experimental workflow for comparing detachment methods and their effects on FasL/Fas expression.
Mechanisms of Action and Functional Consequences
Proposed Mechanism of Accutase Action
The research provides evidence that Accutase does not merely cause the internalization of FasL but actively cleaves it from the cell surface. Western blot analysis revealed small FasL fragments (under 20 kD) in the supernatant of Accutase-treated cells, which were absent in the EDTA-treated group. Furthermore, immunofluorescence staining showed that FasL was no longer localized to the cell membrane after Accutase treatment [5]. This cleavage mechanism is analogous to the shedding of FasL by Matrix Metalloproteinases (MMPs) [5]. The diagram below illustrates this proposed mechanism and its consequences.
Diagram 2: Mechanisms of EDTA and Accutase action on cell adhesion and surface proteins.
Impact on Cell Viability and Function
A paradoxical yet important finding was that despite cleaving surface proteins, Accutase maintained superior cell viability compared to the EDTA-based solution and DPBS buffer, even after extended incubation periods (60-90 minutes) [5]. This highlights that cell viability, while a crucial metric, is not a comprehensive indicator of cellular integrity, as key surface receptors may be compromised even in viable cells.
The functional significance of cleaving FasL is profound. As a critical mediator of cytotoxic activity in the immune system, the loss of surface FasL would be expected to impair Fas/FasL-mediated apoptosis signaling and related immune functions [5]. This has direct implications for the accuracy of immunology and cell death assays.
The Scientist's Toolkit: Essential Research Reagents
Selecting the appropriate reagents is fundamental to designing experiments involving adherent cell detachment. The table below lists key solutions and their applications.
Table 3: Key Reagents for Adherent Cell Detachment Research
| Reagent / Solution | Type | Primary Function & Mechanism | Key Considerations |
|---|---|---|---|
| Accutase | Enzymatic Blend | Proteolytic and collagenolytic digestion of cell adhesion proteins. Considered gentler than trypsin. | Can cleave specific surface proteins (e.g., FasL, Fas); requires post-detachment recovery time. |
| EDTA-based Solutions (e.g., Versene) | Non-Enzymatic (Chelator) | Chelates calcium and magnesium ions, disrupting calcium-dependent cell adhesion. | Preserves many surface markers; may be less effective for strongly adherent cells. |
| Trypsin-EDTA | Enzymatic | Powerful protease (trypsin) cleaves adhesion proteins; EDTA enhances efficacy via chelation. | Known to degrade many surface proteins and can reduce cell viability; cost-effective. |
| Detachin | Enzymatic Blend | Proprietary formulation with protease/collagenase activities and EDTA for gentle detachment. | Marketed for gentle detachment of primary and delicate cells; maintains phenotype [98]. |
| Cell Scraper | Mechanical Tool | Physically dislodges cells from the culture surface by manual force. | Maximizes surface protein preservation but can cause cell damage and is not scalable. |
Discussion and Research Implications
This case study elucidates a critical methodological pitfall: a detachment reagent that preserves cell viability and most surface markers may still severely compromise specific proteins of interest. The finding that Accutase cleaves FasL and Fas, while leaving a common macrophage marker (F4/80) intact, underscores the need for marker-specific validation in experimental design.
These findings have several key implications:
- Assay Interference: Using Accutase for detachment will directly interfere with the accuracy of any assay measuring Fas/FasL surface expression or Fas/FasL-mediated functions, such as apoptosis assays.
- Recovery Period: If Accutase must be used, the data indicate that a recovery period of approximately 20 hours in culture is necessary for the surface expression of FasL and Fas to return to normal levels [5].
- Method Selection: For studies focusing on Fas, FasL, or other protease-sensitive markers, non-enzymatic methods like EDTA-based solutions or cold-shock detachment are recommended to preserve phenotypic accuracy [99]. Mechanical scraping, while not scalable, can serve as a useful control for maximum surface protein preservation.
Within the broader context of adherent cell detachment research, this case study highlights that the choice of dissociation method is not a one-size-fits-all decision. While Accutase is an excellent gentle enzymatic alternative to trypsin for general subculturing and for preserving many surface markers, it significantly compromises the Fas/FasL system. EDTA-based non-enzymatic methods offer a superior alternative for the study of these specific proteins. Therefore, researchers must align their cell detachment protocol with their specific analytical goals, validating the method for the target markers to ensure the biological relevance and reliability of their data.
Conclusion
Adherent cell detachment is far from a mere technical step; it is a decisive factor that influences experimental outcomes and therapeutic product quality. While enzymatic methods like trypsin remain widespread, they carry inherent risks of damaging surface proteins and altering cell physiology. Non-enzymatic and novel stimuli-responsive methods offer promising alternatives for preserving cell integrity, particularly for sensitive applications in cell therapy and regenerative medicine. The choice of detachment strategy must be validated against specific criteria—viability, phenotype, and functionality—to ensure biological relevance and manufacturing efficiency. Future progress will hinge on developing more robust, scalable, and gentle detachment technologies that integrate seamlessly with automated, closed-system biomanufacturing processes, ultimately advancing the capabilities of biomedical research and clinical applications.
References
- 1. Adherent Cell Culture Protocol [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/subculturing-adherent-cells.html]
- 2. Adherent culture [https://en.wikipedia.org/wiki/Adherent_culture]
- 3. Advances in cell culture: anchorage dependence - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4275909/]
- 4. An improved way to detach cells from culture surfaces [https://news.mit.edu/2025/improved-way-detach-cells-culture-surfaces-1118]
- 5. Different methods of detaching adherent cells and their ... [https://www.nature.com/articles/s41598-022-09605-y]
- 6. 100 mL Accutase® Cell Detachment Solution, Liquid [https://ecatalog.corning.com/life-sciences/b2c/US/en/Media%2C-Sera%2C-and-Reagents/Cell-Culture-Reagents/Enzymatic-Cell-Dissociation-Reagents/Accutase%C2%AE-Cell-Detachment-Solution/p/25-058-CI]
- 7. Three-Dimensional In Vitro Cell Culture Models for Efficient ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9412659/]
- 8. Comparative analysis of 3D-culture techniques for ... [https://www.nature.com/articles/s41598-025-13588-x]
- 9. Comparative analysis of 3D culture methodologies in ... [https://www.sciencedirect.com/science/article/pii/S0014482725002666]
- 10. AI technology automates tedious cell culture work and analyses [https://www.drugdiscoverytrends.com/ai-technology-automates-tedious-cell-culture-work-and-analyses/]
- 11. Extracellular Matrix Signaling Cues: Biological Functions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12271642/]
- 12. Targeting integrin pathways: mechanisms and advances in ... [https://www.nature.com/articles/s41392-022-01259-6]
- 13. Cell detachment: A review of techniques, challenges, and ... [https://www.sciencedirect.com/science/article/abs/pii/S0079610725000173]
- 14. Extracellular Matrix Components and Mechanosensing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11430160/]
- 15. Extracellular Matrix Proteins Confer Cell Adhesion ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2021.616580/full]
- 16. Mechanotransduction Pathways Linking the Extracellular ... [https://www.sciencedirect.com/science/article/abs/pii/B9780128001806000050]
- 17. Adhesion/ECM/Cytoskeleton [https://www.cellsignal.com/pathways/by-research-area/adhesion-ecm-cytoskeleton-pathways?srsltid=AfmBOoo968ipcqeJci4eC5rWpbLlSSdwQCwdIEverfkAoOtwgoIrj6wR]
- 18. Mammalian cells measure the extracellular matrix area and ... [https://www.nature.com/articles/s41467-025-62153-7]
- 19. A comparative analysis of detachment forces and energies ... [https://www.sciencedirect.com/science/article/pii/S0927776520301247]
- 20. Quantitative comparison of cell–cell detachment force in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10987375/]
- 21. Quantitative comparison of cell-cell detachment force in ... [https://arxiv.org/html/2312.13918v1]
- 22. Quantitative comparison of cell–cell detachment force in ... [https://institut-curie.org/publications/quantitative-comparison-cell-cell-detachment-force-different-experimental-setups]
- 23. Researchers present novel, enzyme-free strategy for ... [https://meche.mit.edu/news-media/researchers-present-novel-enzyme-free-strategy-detaching-cells-culture-surfaces]
- 24. MIT engineers solve the sticky-cell problem in bioreactors ... [https://news.mit.edu/2025/mit-engineers-solve-sticky-cell-problem-bioreactors-and-other-industries-1015]
- 25. A simple technique for quantifying apoptosis in 96-well plates [https://pmc.ncbi.nlm.nih.gov/articles/PMC1142306/]
- 26. Experimental protocol to study cell viability and apoptosis [https://www.ptglab.com/support/flow-cytometry-protocol/experimental-protocol-to-study-cell-viability-and-apoptosis/?srsltid=AfmBOorBSQ1IIB4aigiKTRnAeCxbu-GIim5VWePKp4ywPUzUepURNjNK]
- 27. BestProtocols: Cell Preparation for Flow Cytometry Protocols [https://www.thermofisher.com/us/en/home/references/protocols/cell-and-tissue-analysis/protocols/cell-preparation-flow-cytometery.html]
- 28. Cell Dissociation Market Demand 2025-2035 [https://www.futuremarketinsights.com/reports/cell-dissociation-market]
- 29. Different methods of detaching adherent cells and their ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8983651/]
- 30. Enzymatic and Non-Enzymatic Cell Dissociation Protocols [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 31. A Novel CTC-Binding Probe: Enzymatic vs. Shear Stress ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12345868/]
- 32. Accutase - The Perfect Solution for Dissociating Your Cells [https://captivatebio.com/advantages-of-dissociating-your-cells-with-accutase/]
- 33. Preliminary Study on Sensor-Based Detection of an ... [https://www.mdpi.com/1424-8220/25/9/2726]
- 34. EDTA & Trypsin in Single-Cell Suspension [https://purmabiologics.com/the-role-of-edta-and-trypsin-in-isolating-single-cell-suspensions/]
- 35. Ethylenediaminetetraacetic acid [https://en.wikipedia.org/wiki/Ethylenediaminetetraacetic_acid]
- 36. Ethylenediaminetetraacetic Acid (EDTA) - StatPearls - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK565883/]
- 37. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOoqWw0JGYbriOb7sl1Soc6m8wff_QQKj3nC7areqK2bzZBUlvrRQ]
- 38. Non-Enzymatic Cell Expansion and Harvesting Using a ... [https://www.mdpi.com/2310-2861/11/12/962]
- 39. Strategy for Nonenzymatic Harvesting of Cells via ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10614186/]
- 40. Adherent Cell Culture in Biopharmaceutical Applications [https://www.biopharminternational.com/view/adherent-cell-culture-biopharmaceutical-applications-cell-detachment-challenge]
- 41. Enzyme-free release of adhered cells from standard culture ... [https://www.nature.com/articles/s42003-019-0638-5]
- 42. Functional assessment of migration and adhesion to quantify ... [https://pubs.rsc.org/en/content/articlehtml/2025/sm/d4sm01351d]
- 43. Expansion of induced pluripotent stem cells under ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11802622/]
- 44. Adherent Cell Lines Protocol [https://www.axionbiosystems.com/resources/culture-protocol/adherent-cell-lines-protocol]
- 45. Responsive systems for cell sheet detachment - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3812292/]
- 46. Stimuli-responsive hydrogels for manipulation of cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9718481/]
- 47. Design and fabrication of pH-responsive charge- ... [https://pubmed.ncbi.nlm.nih.gov/40516771/]
- 48. Microcarrier Technology: Advancing Adherent Cell ... [https://ritaibioreactor.com/adherent-cell/]
- 49. Scale-Up Technologies for the Manufacture of Adherent Cells [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2020.575146/full]
- 50. A Comparative Analysis Microcarriers for the Expansion of ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324925002154]
- 51. Thermoresponsive BrushGel Microcarriers for Efficient Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12417781/]
- 52. Parameter Optimization and Capacitance-Based ... [https://www.mdpi.com/2227-9717/12/9/1887]
- 53. Process intensification of cultivated meat production ... [https://www.nature.com/articles/s41598-025-97813-7]
- 54. Monitoring cell detachment in microcarrier-based cultures [https://www.news-medical.net/whitepaper/20250903/Monitoring-cell-detachment-in-microcarrier-based-cultures.aspx]
- 55. Cell Dissociation and Trypsin for Cell Culture [https://www.thermofisher.com/us/en/home/life-science/cell-culture/mammalian-cell-culture/reagents/cell-dissociation.html]
- 56. Operation Tips of Cell Culture and Cell Detachment [https://www.g-biotec.com/operation-tips-of-cell-culture-and-cell-detachment.html]
- 57. Evaluation of enzymatic protocols to optimize efficiency ... [https://www.nature.com/articles/s41538-024-00313-7]
- 58. Mechanical harvesting of cell sheets: an efficient approach ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12175405/]
- 59. Apoptosis: A Comprehensive Overview of Signaling Pathways ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11592877/]
- 60. Targeting cell death signalling in cancer: minimising ' ... [https://www.nature.com/articles/bjc2016111]
- 61. Quantitative comparison of cell–cell detachment force in ... [https://link.springer.com/article/10.1140/epje/s10189-024-00416-9]
- 62. Quantitative comparison of cell-cell detachment force in ... [https://pubmed.ncbi.nlm.nih.gov/38563859/]
- 63. A high-throughput and low-waste viability assay for microbes [https://www.nature.com/articles/s41564-023-01513-9]
- 64. High-Throughput Processing to Preserve Viable Cells [https://pmc.ncbi.nlm.nih.gov/articles/PMC5582583/]
- 65. Flow Cytometry Protocols for Surface and Intracellular Antigen ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4396953/]
- 66. Flow Cytometry Protocol for Cell Surface Markers [https://www.rndsystems.com/resources/protocols/flow-cytometry-protocol-staining-membrane-associated-proteins-suspended-cells]
- 67. Flow Cytometry Protocol | Abcam [https://www.abcam.com/en-us/technical-resources/protocols/flow-cytometry-for-intracellular-and-extracellular-targets]
- 68. Flow Cytometry Sample Preparation: Best Practices [https://kcasbio.com/blogs/sample-preparation-for-flow-cytometry-best-practices/]
- 69. Comprehensive Microbial Control Strategy [https://www.bioprocessintl.com/qa-qc/beyond-traditional-microbiology-testing-developing-a-comprehensive-strategy-for-microbial-control-and-detection]
- 70. Contamination Control Strategies [https://www.ecolab.com/articles/2025/04/contamination-control-strategies]
- 71. What is Upstream and Downstream Bioprocessing? [https://www.tecnic.eu/what-is-upstream-and-downstream-bioprocess/]
- 72. Purification through Membrane Chromatography for ... [https://www.biopharminternational.com/view/purification-through-membrane-chromatography-for-advanced-therapies]
- 73. Reasons and Solutions for Adherent Cell Detachment [https://www.procellsystem.com/resources/cell-culture-academy/cell-culture-academy-reasons-and-solutions-for-adherent-cell-detachment-1980]
- 74. Basics of Culture Media | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-environment/culture-media.html]
- 75. Comparison of culture media supplements identifies serum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12382287/]
- 76. Bioreactor Design for Adherent Cell Culture [https://www.bioprocessintl.com/cell-culture-media/bioreactor-design-for-adherent-cell-culture-the-bolt-on-bioreactor-project-part-2-process-automation]
- 77. : A Review of Techniques, Challenges, and... | CoLab Cell Detachment [https://colab.ws/articles/10.1016/j.pbiomolbio.2025.02.004]
- 78. Enzymatic and Non-Enzymatic Cell ... | Thermo Fisher Scientific - KR [https://www.thermofisher.com/kr/ko/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 79. Gentle and rapid detachment of even delicate adherent ... cells [https://engineering-update.co.uk/2025/11/18/gentle-and-rapid-detachment-of-even-delicate-adherent-cells/]
- 80. A novel feature for monitoring the enzymatic harvesting ... [https://www.nature.com/articles/s41598-022-22561-x]
- 81. How To Passage Cells in Culture [https://bitesizebio.com/13680/how-to-passage-cells/]
- 82. Reasons and Solutions for Adherent Cell Detachment [https://www.linkedin.com/pulse/reasons-solutions-adherent-cell-detachment-pricella-np0me]
- 83. The application of cell surface markers to demarcate distinct ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6983944/]
- 84. Impact of cell harvesting methods on detection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7798138/]
- 85. Comparison of Enzymatic and Non- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3055293/]
- 86. Preserving the True Immune Landscape: Eliminating ... [https://blog.cellsonics.com/blog/preserving-immune-markers]
- 87. What is Cell Detachment Solution? Uses, How It Works & ... [https://www.linkedin.com/pulse/what-cell-detachment-solution-uses-how-works-top-companies-zvwpc/]
- 88. The regenerative journey: exploring stem cell roles from injury ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502613/]
- 89. Current Landscape of FDA Stem Cell Approvals and Trials ... [https://www.reprocell.com/blog/current-landscape-of-fda-stem-cell-approvals-and-trials-2023-2025]
- 90. Tissue Chip for Drug Screening [http://ncats.nih.gov/research/research-activities/tissue-chip]
- 91. Transforming Cancer Therapy: Insights from 2025 H1 FDA ... [https://blog.crownbio.com/transforming-cancer-therapy-insights-from-2025-h1-fda-approvals-and-preclinical-drug-discovery]
- 92. Medical embryology and regenerative medicine: research ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1619036/full]
- 93. The Pros and Cons of Adherent Versus Suspension Cell ... [https://www.biopharminternational.com/view/the-pros-and-cons-of-adherent-versus-suspension-cell-culture]
- 94. Scale-Up Technologies for the Manufacture of Adherent Cells [https://pmc.ncbi.nlm.nih.gov/articles/PMC7672005/]
- 95. Adherent Cell Culture Basics: Cell Seeding, Expanding, ... [https://www.corning.com/worldwide/en/products/life-sciences/resources/stories/at-the-bench/guide-to-adherent-cell-culture-basics-seeding-expanding-and-harvesting.html]
- 96. Adherent & Suspension Cell Culture in Bioprocessing [https://escovaccixcell.com/news/adherent-suspension-cell-culture-in-bioprocessing]
- 97. Scaling Cultured Meat: Challenges and Solutions for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12241508/]
- 98. Gentle and rapid detachment of even delicate adherent cells [https://onenucleus.com/gentle-and-rapid-detachment-even-delicate-adherent-cells]
- 99. Impact of Detachment Methods on M2 Macrophage ... [https://www.sciencedirect.com/science/article/abs/pii/S0022175915300284]