Enzymatic Cell Dissociation: Principles, Protocols, and Optimization for Modern Research
This article provides a comprehensive guide to enzymatic cell dissociation, detailing the fundamental principles and advanced methodologies essential for researchers, scientists, and drug development professionals.
Enzymatic Cell Dissociation: Principles, Protocols, and Optimization for Modern Research
Abstract
This article provides a comprehensive guide to enzymatic cell dissociation, detailing the fundamental principles and advanced methodologies essential for researchers, scientists, and drug development professionals. It covers the core mechanisms of enzymatic action on the extracellular matrix, tailored protocols for diverse tissue types, and systematic troubleshooting to balance cell yield and viability. Furthermore, it explores current limitations and evaluates emerging technologies and innovative solutions, offering a holistic resource for optimizing dissociation workflows in applications ranging from single-cell analysis to cell therapy manufacturing.
Understanding Enzymatic Dissociation: Core Principles and Cellular Targets
The Role of the Extracellular Matrix (ECM) in Tissue Integrity
The extracellular matrix (ECM) is a critical non-cellular, three-dimensional network of macromolecules that provides essential structural and biochemical support to surrounding cells [1]. It serves as the fundamental architectural framework of tissues, playing an indispensable role in maintaining tissue integrity through biomechanical strength, structural organization, and dynamic biochemical signaling [2] [3]. The ECM's composition varies significantly across different tissue types but typically includes water, proteins, proteoglycans, and glycosaminoglycans organized into a complex scaffold that directly influences cell behavior, including adhesion, proliferation, differentiation, and survival [1].
Beyond its passive structural role, the ECM actively orchestrates tissue development, maintenance, and repair through integrated biomechanical and biochemical cues [1] [2]. This dynamic reciprocity between cells and their ECM microenvironment is particularly relevant in the context of enzymatic cell dissociation research, where understanding ECM composition and organization is prerequisite to developing effective protocols for tissue dissociation into single-cell suspensions while preserving cellular integrity and viability [4] [5].
Molecular Composition and Structural Organization of the ECM
Core ECM Components
The ECM consists of a sophisticated interplay of structural proteins, specialized glycoproteins, proteoglycans, and glycosaminoglycans that collectively confer both mechanical stability and biochemical signaling capabilities [1].
Table 1: Major ECM Components and Their Primary Functions in Tissue Integrity
| ECM Component | Primary Function | Representative Tissues |
|---|---|---|
| Collagen Type I | Provides tensile strength, structural support; main fibrillar component [3] | Skin (80-85%), bone (>90%), tendon (60-80%) [3] |
| Elastin | Confers elasticity and resilience to repetitive deformation [1] | Blood vessels, skin, lung |
| Fibronectin | Mediates cell adhesion, migration; facilitates collagen assembly [2] | Provisional matrices, developing tissues |
| Laminin | Basement membrane component; cell adhesion, polarization [1] | Epithelial and endothelial basement membranes |
| Proteoglycans | Regulate hydration, compressibility; growth factor reservoir [1] | Cartilage, all connective tissues |
| Hyaluronan | Space-filling, lubrication; cell migration facilitation [1] | Cartilage, embryonic tissues |
Collagen represents the most abundant protein in the human ECM, with type I collagen constituting approximately 80% of total body collagen [3]. Its remarkable tensile strength derives from a unique hierarchical structure where three polypeptide α-chains form a right-handed triple helix (tropocollagen), which then assembles into microfibrils and larger fibrils with characteristic 67-nm banding patterns [3]. This elaborate supramolecular organization enables tissues to withstand substantial mechanical forces while maintaining structural integrity.
ECM-Bound Growth Factors and Matricryptic Sites
The ECM serves as a dynamic reservoir for various growth factors and signaling molecules, including fibroblast growth factor (FGF), epidermal growth factor (EGF), transforming growth factor-β (TGF-β), and vascular endothelial growth factor (VEGF) [1]. These factors are released in a spatiotemporally controlled manner to guide critical processes such as stem cell differentiation, angiogenesis, and tissue repair [1]. Additionally, proteolytic cleavage of ECM components can reveal cryptic bioactive sites (matricryptic sites) that influence cell behavior during development, homeostasis, and repair processes [2].
ECM Remodeling in Tissue Homeostasis and Repair
Dynamic ECM Turnover
ECM remodeling is a continuous, tightly regulated process essential for tissue homeostasis and wound healing [2]. This dynamic balance between synthesis and degradation ensures proper tissue architecture and function. During repair processes, the ECM undergoes sequential modifications:
- Provisional Matrix Formation: Following injury, a fibrin-rich provisional matrix forms, providing initial structural support and enabling cellular infiltration [2].
- Inflammatory Phase: The provisional matrix modulates inflammatory responses by recruiting fibroblasts and endothelial cells [2].
- Proliferative Phase: Fibroblasts deposit new ECM components, particularly collagen type III, which is later replaced by stronger collagen type I [2].
- Remodeling Phase: Mature collagen cross-linking and realignment enhance tissue tensile strength and restore structural integrity [2].
Enzymatic Mediators of ECM Remodeling
Matrix metalloproteinases (MMPs) represent the primary enzymatic system responsible for ECM degradation during remodeling processes [6] [2]. These zinc-dependent endopeptidases collectively degrade virtually all ECM components and are essential for facilitating cell migration, releasing growth factors, and resolving provisional matrices [6]. Dysregulated MMP activity is implicated in various pathological conditions, including excessive tissue scarring (fibrosis), chronic wounds, and cancer progression [6] [2].
Table 2: Key Matrix Metalloproteinases in ECM Remodeling and Their Substrates
| MMP Type | Primary Substrates | Biological Functions | Clinical Associations |
|---|---|---|---|
| MMP-1 (Collagenase-1) | Fibrillar collagens (I, II, III) [6] | Initiates collagen degradation; tissue repair [6] | Colorectal cancer inflammation [7] |
| MMP-2 (Gelatinase A) | Gelatin, collagen IV, V [6] | Basement membrane remodeling; angiogenesis [6] | Colitis-associated cancer biomarker [7] |
| MMP-9 (Gelatinase B) | Gelatin, collagen IV, V [6] | Inflammatory cell recruitment; vascular remodeling [6] | TNBC invasiveness (via LOXL4 induction) [7] |
| MMP-7 (Matrilysin) | Proteoglycans, fibronectin, laminin [6] | Epithelial repair; antimicrobial defense [6] | Diagnostic biomarker in skin cancer [6] |
| MMP-11 (Stromelysin-3) | Laminin, fibronectin, aggrecan [6] | Adipogenesis; tumor-stroma interactions [6] | Prognostic biomarker in skin cancer [6] |
| MMP-14 (MT1-MMP) | Collagen I, II, III; activates pro-MMP2 [6] | Pericellular proteolysis; cell invasion [6] | Central hub in protein interactions; diagnostic biomarker [6] |
ECM Mechanics and Cellular Mechanotransduction
ECM stiffness represents a crucial mechanical property that directly influences cell behavior through mechanotransduction pathways [1]. Cells sense and respond to ECM mechanical properties via integrin-mediated adhesion complexes, activating intracellular signaling cascades that regulate gene expression and cell fate decisions [1] [2].
The relationship between ECM stiffness and cell differentiation is particularly evident in stem cell biology, where soft matrices that mimic brain tissue (~0.1-1 kPa) promote neuronal differentiation, while stiffer matrices resembling bone (~25-40 kPa) favor osteogenesis [1]. This mechanosensitive regulation occurs through force-dependent changes in focal adhesion assembly and subsequent activation of FAK (focal adhesion kinase), MAPK/ERK, and PI3K/Akt signaling pathways [2].
Lysyl oxidases (LOX) and LOX-like proteins mediate collagen cross-linking, significantly contributing to ECM stiffening [1]. Excessive LOX activity leads to pathological ECM accumulation in fibrotic conditions and can promote cancer progression by enhancing tissue stiffness and activating pro-invasive signaling pathways [1] [7]. In triple-negative breast cancer, LOXL4 induces MMP-9 expression through NF-κB activation, increasing cancer cell invasiveness [7].
Diagram 1: ECM Mechanotransduction Signaling Pathway. ECM mechanical properties are sensed by integrin receptors that cluster into focal adhesions, activating FAK and downstream MAPK/ERK and PI3K/Akt pathways that ultimately regulate gene expression and cell fate decisions [1] [2].
Enzymatic Dissociation of Tissues: Targeting ECM Integrity
Principles of Enzymatic ECM Dissociation
Enzymatic tissue dissociation aims to disrupt the ECM and intercellular junctions to obtain viable single-cell suspensions for downstream applications [4] [5]. The approach requires careful selection of enzymes that target specific ECM components while preserving cell surface markers and viability [5]. The composition and density of ECM vary considerably across tissues, necessitating optimized dissociation protocols for each tissue type [4].
The ECM presents multiple enzymatic targets for dissociation protocols:
- Fibrillar collagens: Primarily targeted by collagenases
- Basement membrane collagens (type IV): Sensitive to dispase and certain collagenases
- Fibronectin and other glycoproteins: Cleaved by dispase and serine proteases
- Hyaluronic acid: Degraded by hyaluronidases
- Proteoglycan core proteins: Susceptible to various proteases
ECM-Targeting Enzymes in Dissociation Protocols
Table 3: Enzymes for ECM Disruption in Tissue Dissociation Protocols
| Enzyme Class | Specific Examples | ECM Targets | Applications | Considerations |
|---|---|---|---|---|
| Collagenases | Collagenase A, B, D, H, P [5] | Fibrillar collagens (I, II, III) [5] | Lung, heart, muscle, bone, liver, tumors [5] | Collagenase D preserves surface proteins [5] |
| Serine Proteases | Trypsin [5] | Broad-spectrum protease activity [5] | General tissue dissociation | Harsh; damages surface antigens [5] |
| Metalloproteases | Dispase [5] | Fibronectin, collagen IV [5] | Gentle dissociation protocols | Preserves membrane integrity [5] |
| Glycosidases | Hyaluronidase [5] | Hyaluronic acid [5] | Combined with other enzymes | Targets glycosaminoglycans [5] |
| Cold-Active Enzymes | Cold-adapted proteases [5] | ECM components at low temperatures [5] | Transcriptomic studies | Minimizes transcriptional changes [5] |
Advanced Dissociation Technologies
Recent technological innovations have significantly improved ECM dissociation efficiency while better preserving cell viability:
Microfluidic Dissociation Platforms: These systems integrate enzymatic and mechanical dissociation in controlled microenvironments, enabling rapid processing (20-60 minutes) with improved cell viability (60-95% depending on cell type) [4]. One mixed-modal platform demonstrated recovery of approximately 400,000 total cells/mg tissue from mouse kidney with 90% viability for epithelial cells [4].
Hypersonic Levitation and Spinning (HLS): This contact-free method utilizes a triple-acoustic resonator probe to generate microscale "liquid jets" that exert precise hydrodynamic forces on tissues, achieving 90% tissue utilization in 15 minutes with 92.3% cell viability while preserving rare cell populations [8].
Electrical Dissociation: Applied electric fields can dissociate bovine liver tissue with 95% efficiency in just 5 minutes while maintaining 90% cell viability [4].
Ultrasound-Assisted Dissociation: High-frequency sonication, particularly when combined with enzymatic methods, achieves 72% dissociation efficiency in bovine liver tissue [4].
Diagram 2: Experimental Workflow for Tissue Dissociation. Tissue processing involves mechanical mincing followed by either enzymatic digestion or advanced dissociation technologies to obtain single-cell suspensions for downstream applications [4] [8] [5].
Research Reagent Solutions for ECM Studies
Essential Reagents for ECM Manipulation and Analysis
Table 4: Key Research Reagents for ECM and Dissociation Studies
| Reagent Category | Specific Examples | Primary Function | Application Notes |
|---|---|---|---|
| Collagenases | Collagenase D from C. histolyticum [5] | Hydrolyzes native collagen helices [5] | Preferred for surface protein preservation [5] |
| Specialized Proteases | Dispase, Trypsin, Papain [5] | Targets specific ECM protein domains [5] | Dispase is gentler; trypsin is harsh but efficient [5] |
| Hyaluronidases | Bovine or recombinant hyaluronidase [5] | Degrades hyaluronic acid in ECM [5] | Typically used in enzyme cocktails [5] |
| Chelating Agents | EDTA, EGTA [4] | Disrupts calcium-dependent cell adhesions [4] | Used in combination with enzymes [4] |
| Decellularization Agents | SDS, Triton X-100, Sodium deoxycholate [1] | Removes cellular content while preserving ECM [1] | Ionic surfactants more effective but more disruptive [1] |
| MMP Inhibitors | Batimastat, Ilomastat [6] | Inhibits matrix metalloproteinase activity [6] | Research on ECM remodeling pathways [6] |
The extracellular matrix represents far more than a passive structural scaffold; it is a dynamic, information-rich microenvironment that actively regulates tissue integrity through biomechanical and biochemical signaling [1] [2]. Understanding ECM composition, organization, and remodeling mechanisms is fundamental to developing effective enzymatic dissociation protocols that balance dissociation efficiency with cellular viability and functionality [4] [5].
Recent advances in dissociation technologies, including microfluidic platforms, hypersonic levitation, and electrical dissociation methods, have significantly improved our ability to disrupt ECM integrity while preserving cellular characteristics [4] [8]. These innovations, coupled with a growing understanding of ECM biology, continue to enhance single-cell research applications and tissue engineering approaches.
The critical relationship between ECM integrity and successful tissue dissociation underscores the importance of continued research into ECM biology and the development of targeted reagents that specifically address the challenges posed by different tissue types and their unique ECM compositions. As our understanding of ECM heterogeneity and dynamics deepens, so too will our ability to precisely manipulate this complex microenvironment for research and therapeutic applications.
Within the framework of enzymatic cell dissociation research, the precise selection of enzymes based on their specific substrates is a fundamental determinant of success. The efficacy of tissue dissociation and subsequent cell viability hinges on a deep understanding of enzyme-substrate interactions. This whitepaper provides an in-depth technical guide to four pivotal enzymes—collagenase, trypsin, dispase, and hyaluronidase—detailing their specific substrates, mechanisms of action, and optimized applications. Aimed at researchers, scientists, and drug development professionals, this document synthesizes current biochemical knowledge to establish robust protocols and provide a curated toolkit for experimental design, thereby enhancing reproducibility and outcomes in primary cell isolation and tissue engineering.
Enzyme-Substrate Specificity and Biochemical Profiles
Table 1: Key Enzymes and Their Specific Substrates in Cell Dissociation
| Enzyme | Classification | Natural Substrate & Cleavage Specificity | Activators & Inhibitors | Primary Applications in Cell Dissociation |
|---|---|---|---|---|
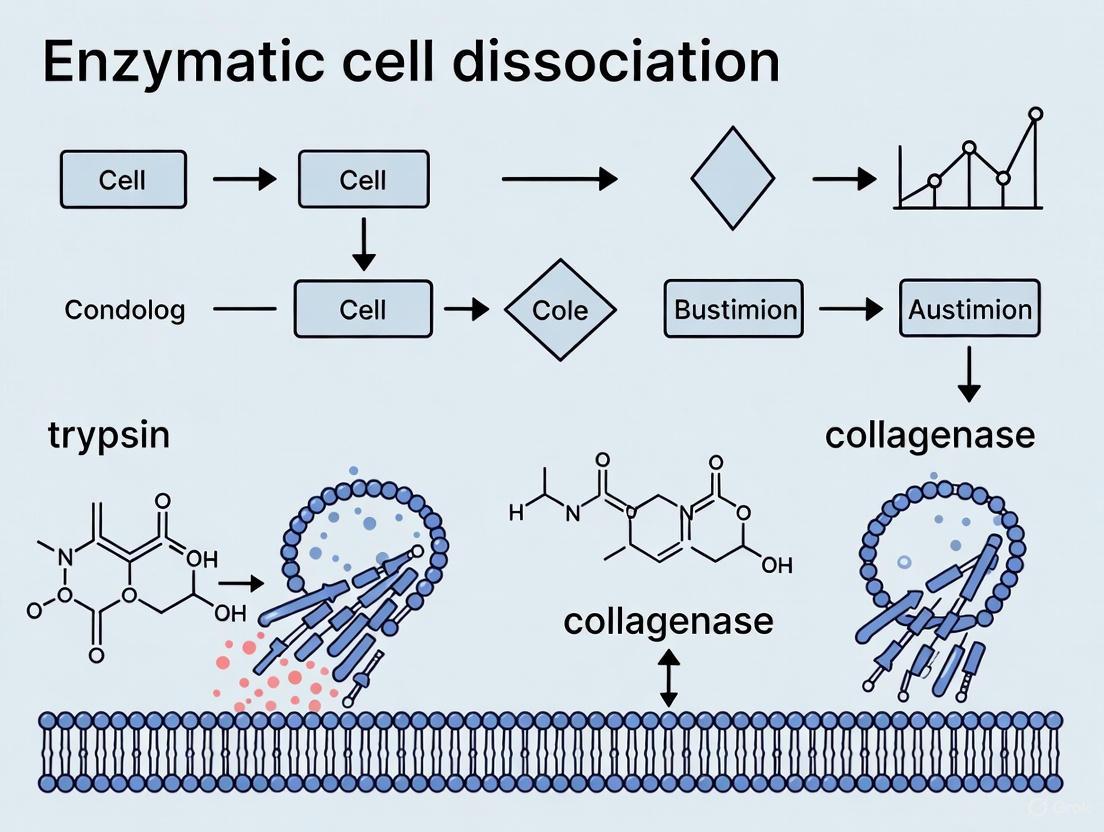
| Collagenase [9] | Metallo-peptidase (M9 family) | Native collagen (Triple-helical structure). Recognizes Pro-X-Gly-Pro sequence; cleaves between X and Gly [9]. | Activators: Zn²⁺, Ca²⁺ [9].Inhibitors: EDTA, 1,10-phenanthroline [10] [9]. | Digestion of dense connective tissues (epithelial, adipose, liver, bone) [9]; Treatment of fibrotic conditions and scar tissue [9]. |
| Trypsin [11] | Serine protease (S1 family) | Peptide bonds at the carboxyl side of Lysine and Arginine [11]. | Activator: Ca²⁺ (protects activity) [11].Inhibitors: Serine protease inhibitors (e.g., APMSF), some plant polyphenols [11]. | Dissociation of cell monolayers; Proteomics (protein digestion for mass spectrometry) [11]. |
| Dispase [10] | Neutral Protease (Metallo-peptidase) | Non-specific cleavage of peptide bonds containing Leucine and Phenylalanine [10]. | Activators: Divalent cations (Ca²⁺, Mg²⁺) [10].Inhibitors: EDTA, EGTA [10]. | Gentle separation of epithelial sheets; Hepatocyte and stem cell isolation [10]. |
| Hyaluronidase [12] [13] | Endo-β-N-acetylhexosaminidase | Hyaluronan (HA) and specific isoforms of Chondroitin Sulfate (CS) [12] [13]. Human HYAL4 is a CS-specific endo-β-N-acetylgalactosaminidase [13]. | Specific inhibitors are less commonly defined; activity is pH and ion-dependent. | Breakdown of hyaluronan in the extracellular matrix; used as a spreading agent to enhance diffusion of other enzymes [12]. |
Experimental Protocols for Enzymatic Activity Assessment
Collagenase Activity Assay
Principle: The assay measures the hydrolysis of a synthetic chromophore-substrate, which is specifically cleaved by collagenase between leucine and glycine residues, leading to the release of a colored fragment that can be quantified spectrophotometrically [14].
Protocol:
- Reagent Preparation: Reconstitute the collagenase chromophore-substrate (e.g., product #27667 from Sigma-Aldrich) according to manufacturer specifications. Prepare an appropriate buffer, typically containing Tris-HCl and calcium ions at pH 7.5, to maintain enzyme stability [14] [9].
- Reaction Setup: Mix the substrate solution with the collagenase enzyme sample. A standard reaction mixture might contain 0.5-1.0 mL of substrate solution and 0.1-0.5 mL of enzyme solution [14].
- Incubation and Measurement: Incubate the reaction mixture at 37°C for a fixed period (e.g., 10-30 minutes). Monitor the increase in absorbance at 320 nm in real-time or at the endpoint using a spectrophotometer [14].
- Calculation: One unit of collagenase activity can be defined as the amount of enzyme that catalyzes the release of 1 μmol of the chromogenic product per minute under the specified assay conditions. Use the molar extinction coefficient of the product to calculate enzyme activity.
Trypsin Activity Assay Using Fluorogenic Substrates
Principle: This highly sensitive assay utilizes a synthetic peptide (e.g., Nα-Benzoyl-L-arginine 7-amido-4-methylcoumarin hydrochloride) conjugated to a fluorescent group (AMC). Trypsin cleavage releases the AMC, resulting in a measurable increase in fluorescence [15] [11].
Protocol:
- Reagent Preparation: Prepare a stock solution of the fluorogenic substrate in DMSO or assay buffer. Dilute the substrate to the working concentration in an appropriate buffer (e.g., Tris-HCl, pH 8.0), ensuring the final concentration of organic solvent is ≤1% [11].
- Reaction Setup: In a quartz cuvette or a microplate well, combine the substrate solution with the trypsin sample. Include a negative control without the enzyme to account for background fluorescence.
- Kinetic Measurement: Immediately place the reaction mixture in a pre-warmed fluorometer (or spectrophotometer with a fluorescence detector) set at 37°C. Monitor the fluorescence intensity continuously for 5-10 minutes. Typical excitation and emission wavelengths for AMC are 380 nm and 460 nm, respectively [15].
- Data Analysis: Calculate the rate of fluorescence increase (ΔF/Δt) from the initial linear portion of the kinetic curve. Determine the enzyme activity by comparing this rate to a standard curve generated with known concentrations of free AMC.
Visualizing Enzymatic Cell Dissociation Workflows
Strategic Enzyme Selection for Tissue Dissociation
The following diagram illustrates the decision-making workflow for selecting enzymes based on target tissue composition, a core principle in enzymatic cell dissociation research.
Mechanism of Collagenase Action on Triple-Helical Collagen
This diagram details the unique mechanism by which collagenase recognizes and cleaves its native, triple-helical substrate, a key differentiator from other proteases.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents for Enzymatic Cell Dissociation
| Reagent / Product Name | Key Features & Specifications | Supplier Example | Primary Function in Research |
|---|---|---|---|
| Collagenase Chromophore-Substrate [14] | Powder/Crystals; for use at 320 nm; shipped on wet ice; store at -20°C. | Sigma-Aldrich | Quantitative spectrophotometric determination of collagenase activity [14]. |
| Nα-Benzoyl-L-arginine 7-amido-4-methylcoumarin HCl [15] | Fluorogenic substrate; CAS # 83701-04-6. | Santa Cruz Biotechnology | Sensitive, real-time fluorometric assay for trypsin and trypsin-like enzyme activity [15]. |
| Neutral Protease (Dispase), Purified [10] | From Bacillus polymyxa; Molecular Weight: ~36 kDa; pH optimum: 5.9-7.0; Animal-Free (AF). | Worthington Biochemical | Gentle dissociation of epithelial sheets and stem cells; used as a secondary enzyme in tissue dissociation protocols [10]. |
| L-Arginine 7-amido-4-methylcoumarin dihydrochloride [15] | Fluorogenic substrate; CAS # 113712-08-6. | Santa Cruz Biotechnology | A sensitive fluorogenic substrate for the assay of trypsin and cathepsin H [15]. |
| Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride [15] | Chromogenic substrate (pNA-based); CAS # 21653-40-7. | Santa Cruz Biotechnology | Colorimetric assay for trypsin, papain & other proteolytic enzymes; cleavage releases yellow nitroaniline [15]. |
| Collagenase from Clostridium histolyticum [9] | Available as Types I-V and NB with varying ratios of collagenase classes and secondary protease activities. | Various (e.g., Worthington) | Selective digestion of different tissues (e.g., Type I for epithelium, Type II for heart/bone) based on specific enzymatic profiles [9]. |
The strategic application of collagenase, trypsin, dispase, and hyaluronidase is foundational to advancing research in cell biology and drug development. The efficacy of these enzymes is inextricably linked to their precise substrate specificities: collagenase for the triple-helical structure of native collagen, trypsin for basic amino acid residues, dispase for bonds involving hydrophobic residues, and hyaluronidase for glycosaminoglycan networks [11] [10] [9].
Mastering the use of these tools—from selecting the appropriate collagenase type for a specific tissue [9] to employing fluorogenic substrates for kinetic analysis [15]—enables researchers to design highly effective and reproducible dissociation protocols. This knowledge empowers the development of more physiologically relevant cell cultures, improves the efficiency of primary cell isolations, and ultimately accelerates discovery in regenerative medicine and therapeutic development. As the field progresses, a continued deep understanding of enzyme-substrate interactions will remain a core principle driving innovation in enzymatic cell dissociation research.
The controlled dissociation of tissues into functional single cells is a cornerstone of modern biological research, enabling everything from single-cell transcriptomics to the development of cell-based therapies. At the heart of this process are proteolytic enzymes—specialized proteins that catalyze the breakdown of other proteins. This whitepaper examines the precise mechanisms by which these enzymes disrupt the critical adhesive structures that maintain tissue integrity: cell-cell and cell-matrix junctions. Understanding these mechanisms is not merely technical; it is fundamental to advancing research in cancer metastasis, inflammatory diseases, and regenerative medicine, where controlled and specific modulation of cell adhesion is paramount [16].
The architecture of tissues is maintained by a complex network of intercellular junctions and a scaffold of extracellular matrix (ECM). Cell-cell junctions, such as adherens junctions and tight junctions, are primarily composed of cadherins and claudins that form connections between adjacent cells. Cell-matrix junctions, notably focal adhesions, connect cells to the ECM via integrins and other receptors. These structures are cemented together by specific protein complexes, which become the direct targets for proteolytic enzymes used in dissociation protocols [17] [18]. A mechanistic understanding of how different classes of proteases recognize and cleave these targets allows researchers to select enzymes that maximize yield and viability while preserving the cellular phenotypes essential for their downstream applications.
Core Mechanisms of Junction Disruption
Proteolytic enzymes disrupt tissue integrity by employing a multi-faceted attack on the protein components of cell junctions and the extracellular matrix. The mechanism can be broken down into two primary, interconnected pathways.
Direct Cleavage of Junction Proteins
The most straightforward mechanism is the direct enzymatic cleavage of the core proteins that constitute cell-cell and cell-matrix adhesions.
Targeting Tight Junctions: Tight junctions, which form seals between epithelial and endothelial cells, are rich in proteins like occludin and claudin. Studies have shown that proteases from diverse biological sources, including allergenic pollen, directly degrade these proteins. For instance, diffusates from ragweed and birch pollen were found to cleave occludin, leading to a loss of barrier function, an effect that could be blocked by serine and cysteine protease inhibitors [19]. This demonstrates how proteases can directly compromise a critical cellular barrier.
Targeting Adherens Junctions: The adhesion between cells is largely mediated by E-cadherin, a key component of adherens junctions. Research on spontaneous bacterial peritonitis (SBP) has revealed a novel bacterial protease activity in pathogenic E. coli and Proteus mirabilis that is directly responsible for the cleavage of E-cadherin structures. This direct cleavage dissociates the intercellular adhesive contacts, facilitating bacterial translocation [20].
Targeting the Extracellular Matrix (ECM): The ECM provides the foundational scaffold for cells. Enzymes like collagenase hydrolyze collagens (the most abundant proteins in the body), while hyaluronidases cleave the glycosidic bonds in hyaluronic acid. Dispase is a gentler enzyme that specifically cleaves fibronectin and collagen IV, making it particularly useful for dissociating delicate tissues without disrupting cell membranes [21]. The summary of key enzyme targets is shown in Table 1 below.
Table 1: Proteolytic Enzymes and Their Primary Junctional Targets
| Enzyme Class | Primary Targets | Biological Consequence |
|---|---|---|
| Serine Proteases (e.g., Trypsin) | Broad-spectrum, including surface receptors and adhesion proteins | Efficient but harsh dissociation; can damage cell surfaces and reduce viability [21] |
| Collagenases | Interstitial collagens (I, II, III, etc.) | Breaks down the structural core of the ECM, facilitating tissue disintegration [21] |
| Dispases | Fibronectin and Collagen IV | Gentler dissociation of epithelial sheets; preserves cell membrane integrity [21] |
| Hyaluronidases | Hyaluronic acid | Degrades a major ECM carbohydrate component, often used in combination with other enzymes [21] |
| Bacterial/Cysteine Proteases | E-cadherin, Occludin | Directly cleaves cell-cell junction proteins, compromising epithelial integrity [20] [19] |
Indirect and Signaling-Mediated Disruption
Beyond direct cleavage, proteolytic activity can initiate downstream cellular processes that lead to the destabilization of junctions.
Activation of Host Proteolytic Systems: Research on SBP-inducing bacteria revealed that the degradation of the tight junction protein occludin is not directly caused by a bacterial enzyme, but is instead mediated by the host cell's own ubiquitin-proteasome system. The bacteria trigger increased ubiquitination of occludin, marking it for degradation by the host's intracellular proteasomes [20]. This represents a sophisticated indirect mechanism of junction disruption.
Dysregulation of Protease Suppression Pathways: Under normal conditions, E-cadherin-mediated cell-cell adhesion plays a role in suppressing the expression of specific proteinases, such as matrix metalloproteinase-9 (MMP-9). The adhesion signals through the PI3-kinase pathway to keep protease levels in check. When E-cadherin contact is lost—either through genetic downregulation or initial proteolytic cleavage—this suppression is lifted, leading to increased protease secretion and a feed-forward loop that enhances invasive behavior and further junction dissolution [22]. The following diagram illustrates the direct and indirect disruption pathways.
Diagram 1: Proteolytic disruption of cellular junctions occurs via direct cleavage of junctional and ECM proteins and through indirect signaling pathways that activate host degradation systems.
Quantitative Data in Enzymatic Dissociation
The efficacy of proteolytic enzymes is quantified through specific metrics that are critical for protocol optimization. Key parameters include viability (the percentage of live cells post-dissociation), yield (the absolute number of cells or nuclei recovered), and dissociation efficacy (the proportion of tissue successfully dissociated). These metrics are highly dependent on the enzyme, tissue type, and digestion conditions.
Recent advancements in dissociation technology have generated robust quantitative data comparing novel methods to traditional enzymatic approaches. For instance, Electric Field Facilitated Rapid Dissociation has demonstrated a significant increase in efficacy and speed compared to traditional methods when processing clinical glioblastoma samples [4]. The following table summarizes performance data from various contemporary dissociation strategies.
Table 2: Quantitative Performance of Tissue Dissociation Methods
| Technology / Approach | Tissue Type | Key Performance Metrics | Reported Viability | Source |
|---|---|---|---|---|
| Electric Field Dissociation | Bovine Liver / Human Glioblastoma | 95% ± 4% efficacy (liver); >5x higher yield vs. traditional (GBM) | 90% ± 8% (cell line); ~80% (GBM) | [4] |
| Ultrasound + Enzymatic | Bovine Liver | 72% ± 10% efficacy (with enzyme) | 91%-98% (cell line, sonication only) | [4] |
| Enzyme-Free Cold Ultrasound | Mouse Tissues (e.g., Lung, Brain) | 1.4 x 10⁴ live cells/mg (lung) | Not explicitly reported | [4] |
| Cryogenic Enzymatic Dissociation (CED) | FFPE Mouse Hippocampus | >10x higher nuclei yield vs. commercial kits | Preserved RNA integrity | [23] |
| Optimized Chemical-Mechanical | Bovine Liver / Breast Cancer Cells | 92% ± 8% efficacy (vs. 37%-42% enzymatic only) | >90% (cancer cells) | [4] |
Detailed Experimental Protocols
To ensure reproducibility and provide a practical reference, this section outlines detailed methodologies for key experiments that elucidate the mechanism of protease action.
Protocol: Assessing Protease-Induced Junction Disruption in Cell Monolayers
This protocol is adapted from studies investigating the effect of pollen proteases and bacterial pathogens on epithelial integrity [20] [19].
- Cell Culture: Use established epithelial cell lines such as Madin-Darby Canine Kidney (MDCK) or Calu-3. Culture cells in transwell inserts until they form a confluent monolayer with established transepithelial electrical resistance (TEER).
- Preparation of Protease Solution: For bacterial studies, co-culture live SBP-derived E. coli or P. mirabilis with the cells at a multiplicity of infection (MOI) of 0-10. For testing purified enzymes or diffusates (e.g., pollen), prepare solutions in serum-free medium.
- Treatment Application: Apply the protease-containing solution to the apical side of the monolayer. Include control wells with serum-free medium only. For inhibitor studies, pre-incubate the protease solution with broad-spectrum inhibitors (e.g., serine protease inhibitor AEBSF or cysteine protease inhibitor E-64) for 1 hour at 37°C.
- Incubation and Monitoring: Incubate cells for a defined period (e.g., 2-4 hours) at 37°C. Monitor integrity in real-time by measuring TEER if equipment is available.
- Post-Incubation Analysis:
- Immunofluorescence: Fix cells and stain for tight junction (occludin, claudin-1) and adherens junction (E-cadherin) proteins. Use fluorescently-labeled secondary antibodies and visualize via confocal microscopy. Loss of continuous, membrane-localized staining indicates junction disruption.
- Western Blotting: Lyse cells and subject proteins to SDS-PAGE. Probe for junction proteins like occludin and E-cadherin. Cleavage products will appear as lower molecular weight bands.
- Zymography: Use gelatin or casein zymography on protease-containing diffusates to directly visualize proteolytic activity.
Protocol: Cryogenic Enzymatic Dissociation (CED) for FFPE Tissues
This advanced protocol, snCED-seq, is designed for high-fidelity nuclei extraction from formalin-fixed paraffin-embedded (FFPE) tissues for single-nucleus RNA sequencing [23].
- Tissue Preparation: Cut 50 μm sections from an FFPE tissue block or use a small piece of the block itself.
- Deparaffinization and Rehydration: Immerse the sample in xylene (or a safe substitute) to remove paraffin, followed by a graded series of ethanol (100%, 95%, 70%) to rehydrate.
- CED Digestion Buffer: Prepare a digestion buffer containing the anionic surfactant sarcosyl and Proteinase K (PK). The optimal PK concentration must be determined empirically (e.g., >0.4 mg/mL for mouse brain).
- Digestion Process: Incubate the tissue in the CED buffer with constant agitation. The key differentiator is that the entire process is performed at low temperatures (e.g., 4°C) for a defined period (e.g., 2 hours). This protects nuclear membranes and retains intranuclear RNA.
- Nuclei Purification: Unlike traditional methods, the CED protocol eliminates the need for filtration or ultracentrifugation through a sucrose cushion. After digestion, centrifuge the suspension at low speed to pellet the nuclei. The use of sarcosyl minimizes cytoplasmic contamination.
- Quality Control: Resuspend the nuclei pellet and assess yield and integrity using automated cell counters and fluorescence microscopy (e.g., DAPI staining). The nuclei should be intact, well-dispersed, and free of large debris.
The Scientist's Toolkit: Essential Reagents & Materials
Successful experimentation in proteolytic dissociation requires a carefully selected suite of reagents and instruments. The following toolkit catalogs essential items, their functions, and considerations for use.
Table 3: Essential Research Reagent Solutions for Proteolytic Dissociation Studies
| Item | Function / Application | Key Considerations |
|---|---|---|
| Collagenase D | Enzymatic dissociation of dense tissues (lung, heart, tumors). | Preferred when functionality of cell-surface proteins must be preserved [21]. |
| Dispase | Gentle dissociation of epithelial sheets and tissues. | Cleaves fibronectin and collagen IV; less damaging to cell membranes than trypsin [21]. |
| Proteinase K | Digests proteins in FFPE tissue for nuclei isolation in CED protocol. | Effective at low temperatures; requires optimization of concentration for specific tissues [23]. |
| Broad-Spectrum MMP Inhibitor (e.g., BB-94) | Blocks activity of matrix metalloproteinases; used to validate protease-specific effects. | Useful as a proof-of-concept tool to prevent E-cadherin cleavage and stabilize junctions [20]. |
| Serine/Cysteine Protease Inhibitors | Blocks specific classes of proteases in mechanistic studies. | Can be used to identify the class of protease responsible for junction disruption [19]. |
| Sarcosyl | Anionic surfactant used in CED buffer for FFPE tissues. | More friendly to the nuclear membrane than SDS or Triton X-100, reducing RNA leakage [23]. |
| Refrigerated Incubated Shaker | Provides controlled temperature and agitation during enzymatic digestion. | Critical for protocols requiring low temperatures (e.g., CED). Agitation improves dissociation efficiency [21]. |
| Shaking Water Bath | Provides highly efficient heat transfer for enzymatic digestions at 37°C. | Preferred over air-incubated shakers for temperature consistency in traditional protocols [21]. |
The targeted disruption of cell-cell and cell-matrix junctions by proteolytic enzymes is a process of remarkable specificity, governed by the enzyme's target profile and the molecular composition of the tissue. As this whitepaper outlines, the mechanisms range from direct cleavage of structural proteins like E-cadherin and collagens to sophisticated indirect pathways that hijack the host's own degradation machinery. The quantitative data and detailed protocols provided herein underscore a critical evolution in the field: the move towards gentler, more specific, and highly controlled dissociation methods. Innovations such as cryogenic enzymatic dissociation and non-enzymatic physical methods are minimizing artifacts and preserving native cellular states, which is crucial for the next generation of single-cell analyses and cell-based therapeutics. For the researcher, a deep and mechanistic understanding of these principles is not an academic exercise—it is the foundation for designing robust, reproducible experiments and developing novel therapeutic strategies aimed at the fundamental processes of cell adhesion and migration.
Tissue dissociation into single-cell suspensions is a critical foundational technique for cell therapy manufacturing, single-cell analysis, and various downstream research applications [4]. This process involves breaking down the extracellular matrix (ECM) and cell-cell junctions that hold tissues together, liberating individual cells for further analysis or therapeutic use. However, this necessary disruption creates a fundamental tension: the very process of dissociation can itself compromise cell health and viability, potentially introducing artifacts that skew experimental results or reduce therapeutic efficacy [4]. Achieving the critical balance between sufficient dissociation for high cell yield and the preservation of cellular integrity represents a significant challenge in modern biological research and therapy development. This technical guide examines the current state of tissue dissociation methodologies, their impacts on cell health, and provides evidence-based protocols for optimizing this delicate balance.
The Fundamental Challenges in Tissue Dissociation
The process of creating single-cell suspensions from solid tissues presents multiple interconnected challenges that researchers must navigate. Tissue architecture, characterized by diverse extracellular matrix components and varied cell-cell adhesion complexes, requires tailored approaches for different tissue types [4]. The heterogeneous nature of biological samples means that no single protocol works optimally across all tissue types, leading to a proliferation of tissue-specific methods with little standardization [4].
Perhaps the most significant challenge lies in the potential for dissociation methods to create artifacts that distort downstream analyses. Enzymatic treatments, particularly those involving proteases, can damage cell surface proteins, compromising the viability of cells intended for therapeutic use and potentially destroying epitopes crucial for flow cytometry or other analytical techniques [4] [24]. Research has demonstrated that enzymes like dispase can cleave specific cell surface markers including CD4, CD8, CD69, and CD103 on immune cells, while also differentially affecting the recovery of various epithelial cell populations [24].
Additionally, extended processing times—some protocols require hours or even overnight digestion—increase the window for contamination, cellular stress, and the introduction of technical errors [4]. These temporal constraints can create difficult tradeoffs, where researchers must balance shorter processing times to maintain viability against longer digestion periods to improve cell yield [4].
Methodological Approaches: Comparative Analysis
Enzymatic Dissociation Methods
Enzymatic dissociation remains the most widely employed approach for tissue dissociation, utilizing various enzymes to break down ECM components and cell adhesions. The table below summarizes commonly used enzymes and their specific applications:
Table 1: Enzymatic Agents for Tissue Dissociation
| Enzyme | Primary Target | Applications | Considerations |
|---|---|---|---|
| Collagenase | Collagen triple helices | Various tissues including lung, liver, tumor | Type (P vs D) affects cell population recovery [24] |
| Trypsin | Proteins, cleaving peptide bonds | General tissue dissociation | Can damage cell surface proteins; often used with EDTA [25] |
| Dispase | Collagen IV, fibronectin | Epithelial cell isolation | Cleaves specific cell surface markers (CD4, CD8, CD69) [24] |
| Hyaluronidase | Hyaluronic acid | Combined with other enzymes for ECM degradation | Often used in enzyme cocktails [4] |
| DNase | Free DNA | Prevents cell aggregation from DNA released by dying cells | Commonly added to enzyme mixtures [24] |
The selection of specific enzymatic protocols significantly impacts experimental outcomes. Comparative studies have demonstrated that protocols high in dispase increase monocyte to macrophage yield while cleaving important immune markers, and collagenase type affects the recovery of specific cell populations—collagenase P protocols yielded increased AT1 and AT2 cells with decreased endothelial cells compared to collagenase D [24].
Mechanical and Non-Enzymatic Alternatives
Mechanical dissociation methods offer enzyme-free alternatives that can preserve cell surface epitopes and avoid enzymatic damage. These approaches range from simple mincing and agitation to automated systems like the Medimachine II, which processes tissue fragments through a specialized filter system [25]. Studies comparing mechanical and enzymatic approaches have found that mechanical dissociation can enrich for specific cell populations; in testicular tissue, mechanical dissociation yielded up to four times higher proportion of SALL4-positive spermatogonia compared to enzymatic methods [26].
Emerging non-enzymatic technologies show significant promise for specialized applications. Electrical dissociation methods can achieve rapid dissociation (as quick as 5 minutes) with high viability (90% ± 8% for MDA-MB-231 cells) [4]. Similarly, ultrasound-based approaches, particularly high-frequency sonication, can effectively dissociate tissues either alone or in combination with minimal enzymatic treatment [4].
Integrated and Microfluidic Platforms
Recent advancements have focused on integrating multiple dissociation modalities into streamlined platforms. Microfluidic systems enable more controlled processing of tissue samples with the potential for automation and reduced processing times [4]. These mixed-modal platforms combine mechanical and enzymatic approaches in optimized configurations, with studies demonstrating efficient dissociation of various mouse tissues (kidney, breast tumor, liver, heart) within 20-60 minutes while maintaining viability profiles between 50-95% depending on cell type [4].
Quantitative Comparison of Dissociation Methods
The table below summarizes performance metrics across various dissociation technologies, highlighting the tradeoffs between yield, viability, and processing time:
Table 2: Performance Metrics of Tissue Dissociation Methods
| Technology | Tissue Type | Cell Yield | Viability | Processing Time | Source |
|---|---|---|---|---|---|
| Optimized Chemical-Mechanical (Enzymatic + Mechanical) | Bovine Liver Tissue | 92% ± 8% | >90% | 15 min | [4] |
| Electrical Dissociation | Bovine Liver Tissue, MDA-MB-231 | 95% ± 4% | 90% ± 8% | 5 min | [4] |
| Ultrasound + Enzymatic | Bovine Liver Tissue | 72% ± 10% | 91%-98% | 30 min | [4] |
| Enzyme-Free Ultrasound | Mouse Heart Tissue | 3.6 × 10⁴ live cells/mg | 36.7% | Not specified | [4] |
| Optimized Protocol for Human Skin | Human Skin Biopsy | ~24,000 cells/4 mm punch | 92.75% | ~3 h | [4] [27] |
| Automated Mechanical (Medimachine) | Mouse Lung Tissue | 1-6 × 10⁵ cells | 60%-80% | ~1 h | [4] |
| Microfluidic Platform | Human Placental Tissue | 2,262 viable cells/mg | Not reported | 45 min to 2 h | [4] |
Optimized Experimental Protocols
Optimized Skin Dissociation Protocol for scRNA-seq
For single-cell RNA sequencing applications using human skin biopsies, researchers have developed an optimized protocol that balances yield with preservation of RNA integrity and cellular diversity [27]. The step-by-step methodology includes:
Tissue Collection and Preparation: Collect 4mm punch biopsies and place in complete RPMI medium with 10% FCS. Ship at 4°C and process within 2 hours of collection [27].
Mechanical Disruption: Using a scalpel, carefully cut the biopsy into small fragments of approximately 1-2mm³ to increase surface area for enzymatic action [27].
Enzymatic Digestion: Prepare an enzyme cocktail appropriate for skin tissue, typically containing collagenase and other proteases optimized for the dense extracellular matrix of dermal tissue. Incubation parameters must be carefully controlled for temperature and duration [27].
Termination and Filtration: Stop the enzymatic reaction by adding inhibition buffer containing serum. Pass the cell suspension through a 70μm strainer to remove undigested tissue fragments and cell aggregates [27].
Cell Washing and Resuspension: Centrifuge the filtrate and resuspend the cell pellet in appropriate buffer for counting and viability assessment [27].
This protocol successfully recapitulated main cell populations of existing single-cell skin atlases while identifying rare cell populations such as mast cells, demonstrating its comprehensive coverage of cellular diversity [27].
Balanced Protocol for Lung Tissue Dissociation
For pulmonary tissue, a systematic comparison of five common enzymatic protocols revealed significant differences in cell population recovery based on enzyme selection [24]:
Tissue Processing: Obtain human lung samples and weigh into 1.5g pieces. Perform mechanical dissociation using a gentleMACS Dissociator with appropriate program settings [24].
Enzymatic Digestion Conditions: Test multiple enzyme combinations including:
- Collagenase D-based protocol
- Collagenase P-based protocol
- High and low dispase concentration protocols Include appropriate controls with mechanical dissociation only [24].
Post-Digestion Processing: Strain samples through 70μm filters, centrifuge at 300G for 5 minutes at 4°C. Resuspend in ACK lysing buffer on ice for 5 minutes to remove red blood cells [24].
Analysis: Assess cell count and viability using automated cell counting systems, and perform phenotypic analysis via flow cytometry or prepare for single-cell RNA sequencing [24].
This comparative approach revealed that collagenase P protocols yielded increased AT1 and AT2 cells with decreased endothelial cells compared to collagenase D, informing protocol selection based on target cell populations [24].
Decision Framework for Method Selection
The following workflow diagram outlines a systematic approach for selecting appropriate dissociation methods based on research objectives, tissue characteristics, and downstream applications:
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents for Tissue Dissociation Research
| Reagent/Category | Specific Examples | Function & Application Notes |
|---|---|---|
| Proteolytic Enzymes | Collagenase (Type I, II, IV), Trypsin, Dispase, Papain | Target specific ECM components; collagenase types affect cell population recovery [24] |
| Support Enzymes | DNase I, Hyaluronidase | Prevent cell aggregation (DNase); degrade hyaluronic acid in ECM [24] |
| Chelating Agents | EDTA, EGTA | Bind calcium ions to disrupt cell-cell adhesions; often used with trypsin [4] |
| Mechanical Dissociation Systems | Medimachine, gentleMACS | Provide standardized mechanical disruption; can enrich specific populations [25] [26] |
| Viability Maintenance | BSA, FBS, Pluronic F-68 | Reduce mechanical shear stress and stabilize cells during processing [25] |
| Emerging Technologies | Electrical dissociation devices, Ultrasound systems | Offer rapid, minimal-enzyme alternatives; electrical methods achieve dissociation in 5 min [4] |
The critical balance between achieving high-quality single-cell suspensions and preserving cell health remains a dynamic area of methodological research. While significant advances have been made in understanding the tradeoffs between different dissociation approaches, the field continues to evolve toward more standardized, reproducible, and gentle methods. The integration of microfluidic technologies, the refinement of non-enzymatic alternatives, and the development of tissue-specific optimized protocols represent promising directions for the future [4]. As single-cell technologies continue to advance and therapeutic applications expand, the importance of mastering tissue dissociation fundamentals will only grow. Researchers must remain informed about methodological developments and carefully validate their dissociation approaches against their specific research goals and downstream applications to ensure both technical success and biological relevance.
From Theory to Practice: Developing and Implementing Effective Dissociation Protocols
Standardized Step-by-Step Enzymatic Dissociation Workflow
The process of enzymatic tissue dissociation serves as the foundational gateway for single-cell analysis, cell therapy manufacturing, and a vast array of downstream research applications. Within the broader thesis that rigorous, standardized methods are paramount for reproducible and reliable scientific discovery, this guide establishes a core, standardized enzymatic dissociation workflow. The overarching principle is that the quality of dissociation directly dictates the quality of all subsequent data; a poorly dissociated sample cannot be rescued by sophisticated downstream technology. The ideal dissociation method must effectively balance multiple criteria: it should be efficient in processing time, effective in yielding sufficient viable cells, and reliable across repeated experiments with minimal artificial manipulation of the isolated cells [28]. This guide synthesizes current literature and protocols to provide a standardized framework that meets these principles, enabling researchers to obtain high-quality single-cell suspensions that faithfully represent the original tissue's biology.
Core Principles and Comparative Method Analysis
The Rationale for Enzymatic Dissociation
Tissues are complex, three-dimensional structures where cells are embedded within an extracellular matrix (ECM) and connected by various cell-cell junctions. Enzymatic dissociation works by using specific proteins to digest these ECM components and intercellular connections, thereby releasing individual cells into suspension [29]. Unlike mechanical dissociation, which applies physical force that can damage cells, enzymatic methods offer a more controlled and efficient means of disaggregation, particularly for compact tissues [4] [29]. The selection of enzymes is critical, as different enzymes target distinct components of the tissue architecture. For instance, collagenase digests collagen, a major ECM protein, while trypsin cleaves peptide bonds, breaking down proteins that facilitate cell adhesion [30]. Papain, a cysteine protease, has proven particularly efficient for neural tissues, digesting substrates like laminins and proteoglycans with less cellular destruction than other proteases [28].
Quantitative Comparison of Dissociation Methods
The qualification of a dissociation method should be based on clear, quantitative criteria. A foundational study established the following benchmarks for a standard method: mononuclear cell viability post-processing ≥80%, absolute live mononuclear cell numbers ≥5 × 10^5 per sample, verified test-retest reliability, and a correlation between cell numbers and disease activity in model systems [28]. The table below summarizes performance data for various enzymatic and emerging methods, providing a basis for protocol selection.
Table 1: Performance Comparison of Tissue Dissociation Technologies
| Technology / Method | Tissue Type | Cell Viability | Cell Yield / Efficacy | Processing Time |
|---|---|---|---|---|
| Neural Tissue Dissociation Kit (Papain-based) [28] | Mouse Brain & Spinal Cord (EAE model) | >80% | High yield; correlation with disease score | ~60-90 min |
| Optimized Enzymatic (Collagenase/Dispase) [4] | Human Breast Cancer | 83.5% ± 4.4% | 2.4 × 10^6 viable cells | >1 hour |
| Trypsin-Based Primary Dissociation [30] | General Primary Tissue | >90% (recommended) | High (protocol-dependent) | 6-24 hours (incl. 4°C incubation) |
| Collagenase-Based Primary Dissociation [30] | General Primary Tissue | >90% (recommended) | High | 4-18 hours |
| Hypersonic Levitation (HLS) with Enzymes [8] | Human Renal Cancer | 92.3% | 90% tissue utilization | 15 minutes |
| Electric Field Dissociation [4] | Clinical Glioblastoma | ~80% | >5x higher than traditional enzymatic | 5 minutes |
Workflow Logic and Decision Pathway
The following diagram outlines the logical sequence and key decision points in a standardized enzymatic dissociation workflow, from tissue acquisition to quality control of the final single-cell suspension.
Standardized Experimental Protocol
Reagents and Materials
The following table details the essential reagents and materials required to execute the standardized enzymatic dissociation protocol.
Table 2: Research Reagent Solutions for Enzymatic Dissociation
| Item | Function / Description | Example / Specification |
|---|---|---|
| Enzymes | Digests extracellular matrix and cell-cell junctions. | Papain, Collagenase, Trypsin, TrypLE, Dispase [28] [30]. |
| Enzyme Buffer | Provides optimal ionic and pH environment for enzyme activity. | Hanks' Balanced Salt Solution (HBSS) or Dulbecco's PBS, with or without Ca²⁺/Mg²⁺ as required [30]. |
| Cell Culture Medium | Halts enzymatic activity; provides nutrients for cells. | Complete medium with serum (e.g., FBS) or serum-free with enzyme inhibitors [30]. |
| Wash Solution | Rinses tissue to remove blood, contaminants, and enzymes. | PBS without Ca²⁺/Mg²⁺ or a balanced salt solution [30]. |
| DNase I | Degrades free DNA released by damaged cells, reducing clumping. | Optional addition to the enzyme mix [4]. |
| Cell Strainer | Removes undissociated tissue clumps and debris. | Nylon or stainless steel, 70-100 μm pore size [30]. |
| Centrifuge Tubes | For washing, concentrating, and resuspending cells. | 15 mL or 50 mL conical tubes. |
Step-by-Step Workflow Protocol
This protocol is adapted from established methodologies for primary tissue dissociation [28] [30] and should be performed under sterile conditions.
Tissue Preparation:
- Place the freshly acquired tissue in a Petri dish containing cold wash solution (e.g., PBS without Ca²⁺/Mg²⁺).
- Using sterile forceps, scissors, and a scalpel, meticulously remove any unwanted fat, necrotic tissue, and connective tissue.
- Mince the remaining tissue into fine pieces of approximately 2-4 mm³.
- Transfer the minced tissue into a conical tube and wash it by adding cold wash solution, allowing the pieces to settle, and then removing the supernatant. Repeat this 2-3 times until the supernatant is clear.
Enzymatic Digestion:
- Prepare the appropriate enzymatic solution pre-warmed to 37°C. The volume should be sufficient to submerge the tissue (e.g., 1-5 mL per 100 mg of tissue).
- Enzyme Selection Guide:
- Neural Tissue: Use a commercial Neural Tissue Dissociation Kit (papain-based) or 20 U/mL papain [28].
- Fibrous/Tumor Tissue: Use collagenase (50-200 U/mL) or a blend with dispase (e.g., 60-100 U/mL collagenase + 0.3-0.6 U/mL dispase) in HBSS with calcium and magnesium [30].
- General/Epithelial Tissue: Use 0.25% trypsin or TrypLE in a buffer without calcium and magnesium [30].
- Add the enzyme solution to the tissue pieces and incubate at 37°C with gentle agitation (e.g., on a rocker). The incubation time must be determined empirically but typically ranges from 30 minutes to 2 hours.
Dissociation Arrest and Cell Recovery:
- Once the tissue appears digested (cloudy suspension, fragmented tissue), promptly add a volume of cold complete medium (with serum) or a specific enzyme inhibitor (e.g., soybean trypsin inhibitor) that is at least equal to the volume of the enzyme solution. This step is critical to prevent over-digestion and loss of cell surface markers.
- Gently triturate the solution 10-15 times using a serological pipette to aid in the dissociation of any remaining fragments.
- Pass the cell suspension through a 70-100 μm cell strainer into a new collection tube to remove any undigested tissue clumps.
Cell Washing and Concentration:
- Centrifuge the filtered cell suspension at 100-300 × g for 5-10 minutes at 4°C.
- Carefully decant the supernatant and resuspend the cell pellet in an appropriate buffer or culture medium.
Quality Control:
- Determine cell viability and concentration using an automated cell counter or hemocytometer with a dye like Trypan Blue. The viability should consistently be ≥90% for culture applications and ≥80% for analytical applications like flow cytometry [28] [30].
- Assess the success of dissociation by microscopy to confirm a predominantly single-cell suspension with minimal clusters.
Advanced Techniques and Future Directions
While standardized enzymatic protocols form the current backbone of tissue dissociation, the field is rapidly evolving to address key challenges such as processing time, cell type-specific damage, and the need for higher throughput. Advanced microfluidic platforms are being developed that integrate enzymatic and mechanical forces in a controlled manner, significantly reducing processing times to 20-60 minutes while improving cell yield for specific populations [4]. Furthermore, several non-enzymatic or enzyme-assisted physical methods are emerging as powerful alternatives.
Table 3: Emerging and Advanced Dissociation Technologies
| Technology | Mechanism | Key Advantages | Considerations |
|---|---|---|---|
| Microfluidic Platforms [4] | Enzymatic + controlled mechanical shear in microchannels. | Reduced processing time; improved consistency; integration with downstream steps. | Limited tissue capacity; potential for channel clogging. |
| Hypersonic Levitation (HLS) [8] | Acoustic waves generate "liquid jets" for non-contact dissociation. | High viability (>92%); preserves rare cells; rapid (15 min). | Early-stage technology; specialized equipment required. |
| Electrical Dissociation [4] | Electric fields permeabilize membranes and disrupt tissue. | Extremely rapid (5 min); high yield for tough tissues (e.g., glioblastoma). | Potential for inducing cellular stress responses. |
| Ultrasound Dissociation [4] | High-frequency sound waves apply mechanical force. | Enzyme-free or enzyme-assisted option; can be gentler than grinding. | Requires optimization of frequency and power. |
These advanced techniques align with the core principles of dissociation research by striving for greater efficiency, effectiveness, and reliability. The move towards automated, closed-system instruments is particularly promising for standardizing workflows across laboratories and for clinical cell manufacturing, where reproducibility and scalability are paramount [4] [8].
Tissue dissociation into single-cell suspensions represents a critical foundational technique for cell therapy manufacturing, single-cell analysis, and a vast array of downstream biomedical applications [4]. This process, which entails breaking down the complex architecture of the extracellular matrix (ECM) and cell–cell junctions, is the essential first step for modern techniques including single-cell sequencing, flow cytometry, establishing cell lines, cultivating organoids, and isolating specific cell types for therapeutic purposes [4]. The overarching challenge lies in the inherent heterogeneity of tissues—not only between different tissue types but also within populations of the same cell type—which demands carefully optimized, tissue-specific dissociation strategies [4].
The process is traditionally carried out via a combination of enzymatic and mechanical methods. However, conventional approaches face significant challenges concerning cell viability, yield, processing time, and the potential to create artifacts that can distort downstream analyses [4]. The enzymatic cocktail, digestion time, and mechanical force required must be precisely calibrated for each tissue type to balance dissociation efficiency against the preservation of cellular integrity and surface markers. This guide provides a detailed, technical framework for the dissociation of three particularly complex and biologically distinct tissues: solid tumors, neural tissue, and epithelial organs, framed within the core principles of enzymatic cell dissociation research.
The table below summarizes key performance metrics for various dissociation technologies applied to different tissue types, highlighting the trade-offs between efficacy, viability, and processing time.
Table 1: Comparative Performance of Tissue Dissociation Methods
| Technology | Tissue Type | Dissociation Efficacy (Live Cells/mg tissue) | Cell Viability (%) | Processing Time |
|---|---|---|---|---|
| Optimized Chemical-Mechanical Workflow [4] | Bovine Liver Tissue | 92% ± 8% (vs. enzymatic only at 37%-42%) | >90% | 15 min |
| Mixed Modal Microfluidic Platform [4] | Mouse Kidney | ~20,000 (epithelial), ~1,700 (leukocyte), ~900 (endothelial) | ~95% (epithelial), 60-90% (others) | 1-60 min |
| Mixed Modal Microfluidic Platform [4] | Mouse Breast Tumor | ~9,000 (epithelial), ~900 (leukocyte), ~300 (endothelial) | 70-80% (epithelial), 50-80% (others) | 1-60 min |
| Ultrasound + Enzymatic [4] | Bovine Liver Tissue | 72% ± 10% (with enzymes) | 91-98% | 30 min |
| Enzyme-Free Cold Ultrasound [4] | Mouse Heart | 3.6 × 10⁴ | 36.7% | Not Specified |
| Electric Field Dissociation [4] | Human Glioblastoma | >5x higher than traditional methods | ~80% | 5 min |
Tissue-Specific Dissociation Strategies
Solid Tumors
Solid tumors present a unique challenge due to their dense, fibrotic extracellular matrix and high degree of cellular heterogeneity. The primary goal is to efficiently break down the tough stromal components while preserving the viability of both cancerous and tumor-infiltrating immune cells for applications like single-cell RNA sequencing and cancer diagnostics [16].
- Recommended Enzymes: Collagenase is the most common class of enzymes used for dissociating dense tissues like solid tumors [31]. For tumors, Collagenase D is often recommended when the functionality and integrity of cell-surface proteins is important for downstream flow cytometry or functional assays [31]. This enzyme can be used in combination with hyaluronidase to target the hyaluronic acid component of the tumor ECM [4] [31].
- Mechanical Integration: Mechanical mincing of the tumor sample is a necessary first step [4]. Subsequent digestion is typically performed with agitation, often using an orbital shaker or shaking water bath at 37°C to enhance enzyme penetration and activity [31]. For a more standardized approach, semi-automated mechanical systems like paddle blenders (Stomachers) can be employed to reduce user-based variability [31].
- Performance Metrics: For triple-negative human breast cancer tissue, an optimized enzymatic/mechanical protocol can yield 2.4 × 10⁶ viable cells with a viability of 83.5% ± 4.4%, though processing can take over an hour [4]. Microfluidic platforms for breast tumors have reported yields of ~9,000 epithelial cells/mg tissue with viabilities between 70-80% [4].
Neural Tissue
Neural tissue is exceptionally delicate and susceptible to mechanical and enzymatic damage. The objective is to gently dissociate the tissue to obtain a viable cell suspension that accurately represents the brain's diverse cellular population, particularly neurons that are easily damaged and often underrepresented in single-cell datasets [31].
- Recommended Enzymes: Harsh enzymes like trypsin are unsuitable for neural tissue. Gentler alternatives such as papain are preferred [4] [31]. Dispase, which cleaves fibronectin and collagen IV without disrupting cell membranes, is another excellent option for a gentle digestion [31].
- Mechanical Integration: Aggressive mechanical methods like bead mill homogenizers must be used with extreme caution and at very low speeds to preserve cell viability [31]. The focus is on gentle trituration. For transcriptomic studies, consider using cold-active enzymes that function at lower temperatures (e.g., below 25°C) to minimize stress-induced changes to the transcriptional landscape during the prolonged dissociation process [31].
- Performance Metrics: An enzyme-free, cold-active acoustic (ultrasound) method for mouse brain tissue has been reported to yield 1.4 × 10⁴ live cells/mg of tissue [4]. Given the fragility of neural cells, viability is the paramount metric, and protocols must be prioritized to maximize it, even at the cost of yield.
Epithelial Organs (Intestinal Mucosa as a Model)
Epithelial organs like the intestine are characterized by crypt-villus structures held together by strong cell-cell junctions. The goal here is to selectively release intact crypts or glands for organoid culture, rather than a complete single-cell suspension, requiring a balance between structural dissociation and the preservation of stem cell viability.
- Recommended Enzymes: For establishing human intestinal organoids (HIOs), a non-enzymatic approach using the chelating agent EDTA is highly effective [32]. EDTA works by sequestering calcium ions, which are essential for cell-adhesion proteins like E-cadherin, thereby disrupting cell-cell contacts and freeing entire crypts from the surrounding mucosa [32].
- Mechanical Integration: Conventional protocols involve incubation in EDTA followed by manual pipetting to mechanically release the crypts, a step that introduces operator variability [32]. A advanced strategy employs a semi-automated mechanical dissociation system (e.g., Cytiva Via Extractor), which standardizes the mechanical agitation step. This system uses sealed pouches with EDTA, running at optimized conditions (e.g., 150 rpm for 7 minutes for fresh tissue at 4°C), leading to an improved success rate for organoid derivation from fresh biopsies [32].
- Performance Metrics: Studies show that semi-automated dissociation improves the success rate of organoid derivation from fresh intestinal mucosal biopsies compared to the conventional manual method [32]. This method produces organoids with growth kinetics, cell type proportions (LGR5+ stem cells, MUC2+ Goblet cells), and methylation profiles indistinguishable from those derived via conventional methods, proving its reliability and superior consistency [32].
The Scientist's Toolkit: Essential Reagents and Equipment
Table 2: Key Research Reagent Solutions for Tissue Dissociation
| Item | Function & Application |
|---|---|
| Collagenase D | Hydrolyzes collagen; recommended for dense tissues (tumors, liver) when surface protein integrity is critical [31]. |
| Papain | A gentle protease; ideal for sensitive tissues like neural tissue [4]. |
| Dispase | Gentle neutral protease; cleaves fibronectin and collagen IV; suitable for epithelial cells and neural tissue [4] [31]. |
| EDTA (Ethylenediaminetetraacetic acid) | A chelating agent; disrupts cell-cell adhesions by binding calcium; used for isolating intact crypts from intestinal epithelium for organoid culture [32]. |
| Hyaluronidase | Degrades hyaluronic acid in the ECM; typically used in combination with other enzymes like collagenase for more complete dissociation [4] [31]. |
| Semi-Automated Dissociator (e.g., Via Extractor) | Standardizes mechanical agitation, reducing user variability and improving yield and success rates for organoid establishment [32]. |
| Orbital Shaker / Shaking Water Bath | Provides consistent agitation and temperature control (usually 37°C) during enzymatic digestion, improving efficiency [31]. |
| Bead Mill Homogenizer (Low-Speed) | For mechanical dissociation; must be capable of very low speeds to preserve cell viability for subsequent culture or analysis [31]. |
The dissociation of complex tissues is a foundational and non-trivial step in modern biological research and therapeutic development. There is no universal protocol; success hinges on selecting a strategy tailored to the specific physical and biochemical properties of the target tissue. Solid tumors demand robust enzymatic cocktails to dismantle their dense stroma, neural tissue requires the gentlest possible handling to preserve fragile cells, and epithelial organs often benefit from non-enzymatic, mechanical methods to isolate functional tissue units. As the field advances, the integration of semi-automated platforms and novel non-enzymatic methods like electrical and ultrasound dissociation promises to deliver greater standardization, improved cell quality, and more reproducible results across the spectrum of precision medicine and basic research.
This technical guide provides a structured framework for selecting optimal enzymatic formulations for tissue dissociation, a critical first step in cell biology research and therapeutic development. Effective cell isolation hinges on understanding the complex interplay between specific enzyme activities, unique tissue compositions, and the stringent requirements of downstream applications. This document synthesizes current methodologies and empirical data to equip researchers with a systematic approach for developing robust, reproducible dissociation protocols that maximize cell yield, viability, and functional integrity.
Tissue dissociation into single-cell suspensions is a foundational technique for flow cytometry, single-cell transcriptomics, cell culture, and therapeutic cell manufacturing [16] [4]. The process involves breaking down the extracellular matrix (ECM) and cell-cell junctions that constitute the tissue's structural integrity. The ECM is a dynamic, heterogeneous network of proteins, glycoproteins, lipids, and glycolipids whose composition varies significantly by tissue type, species, and developmental age [33].
The core principle of enzymatic dissociation is the application of specific biocatalysts to digest these ECM components selectively. However, the choice of technique is often arbitrary rather than systematic [33]. The goal of any dissociation protocol is to maximize the yield of functionally viable cells, a outcome influenced by numerous variables including tissue type, enzyme(s) used, concentration, temperature, and incubation time [33]. This guide establishes a logical, experimental framework for enzyme selection based on tissue composition and application requirements, thereby moving beyond historical precedent to a principles-based approach.
Tissue Composition and Enzyme Mechanisms
A mechanistic understanding of tissue architecture and the enzymes that disrupt it is essential for rational protocol design. The table below catalogs common ECM components and the enzymes that target them.
Table 1: Key Extracellular Matrix Components and Corresponding Dissociation Enzymes
| ECM Component | Biological Function | Enzymes for Dissociation | Mechanism of Action |
|---|---|---|---|
| Collagen (Types I, II, III, etc.) | Provides tensile strength; the most abundant protein in the body [34]. | Collagenase (e.g., A, B, D, H, P) [30] [34] | Hydrolyzes native collagen triple helices at the Y-Gly bond in the -X-Gly-Pro-Y- sequence [34]. |
| Fibronectin & Collagen IV | Basement membrane component; mediates cell adhesion. | Dispase [30] [34] | A neutral protease that cleaves fibronectin and collagen IV without disrupting cell membranes [34]. |
| Hyaluronic Acid | Glycosaminoglycan that hydrates the ECM and provides cushioning. | Hyaluronidase [4] [34] | A glycosidase that cleaves glycosidic bonds, degrading hyaluronic acid [34]. |
| General Proteins & Peptides | Various structural and functional roles. | Serine Proteases (Trypsin, TrypLE) [30] | Cleaves peptide bonds on the C-terminal side of lysine and arginine residues [30]. |
| Cell-Cell Junctions | Proteins like cadherins that mediate cell-to-cell adhesion. | Chelating Agents (EDTA, EGTA) [4] [35] | Chelates cations (e.g., Ca²⁺, Mg²⁺) that are essential for the integrity of certain junctional complexes [35]. |
The following diagram illustrates the logical workflow for selecting a dissociation strategy based on tissue and application-specific factors.
Enzyme Formulations and Selection Guidelines
Enzymes for dissociation are available in various purities and formulations. Crude preparations, such as traditional trypsin or collagenase, contain multiple protease activities, polysaccharidases, nucleases, and lipases, which can enhance the digestion of complex tissues but may introduce variability and unintended cell damage [33]. Purified or defined formulations (e.g., TrypLE, specific collagenase blends) offer greater consistency and are tailored for specific applications, such as those requiring animal-origin-free reagents [30].
Selection must be guided by the target tissue and the needs of the cells. The table below provides a practical guide for matching enzyme types to common tissue types.
Table 2: Enzyme Selection Guide Based on Tissue Type and Application
| Tissue Category | Recommended Enzyme(s) | Typical Concentration | Key Considerations |
|---|---|---|---|
| Solid Tumors | Collagenase (D or P recommended) [34] [36], often with Hyaluronidase [4] | 50-200 U/mL Collagenase [30] | Dense ECM requires aggressive digestion. Collagenase D is preferred for surface protein integrity [34]. |
| Liver/Kidney | Collagenase [35] [34] | 50-200 U/mL [30] | High collagen content. Enzymes reduce fibrous connective tissue for higher yield [35]. |
| Lung/Heart | Collagenase [34] [36] | Protocol-dependent [34] | Optimize buffer volume and time to balance yield and viability [34]. |
| Spleen/Lymph Nodes | Gentle Mechanical [35] or Collagenase D [36] | N/A | Loosely associated tissues; mechanical can be sufficient. Enzymatic only for specific isolations [35] [36]. |
| Brain (CNS) | Papain [4] or specialized kits [36] | Protocol-dependent | Neurons are fragile and underrepresented; gentle protocols are critical [34] [36]. |
| Adherent Cell Cultures | Trypsin, TrypLE, or Dispase [30] | 0.05%-0.25% Trypsin [30] | Trypsin is efficient but harsh. TrypLE is a direct, gentler substitute. Dispase detaches cells as intact sheets [30]. |
| Skin/Epidermis | Dispase [30] | 0.6-2.4 U/mL [30] | Efficiently detaches epidermal sheets without dissociating intercellular junctions [30]. |
| Embryonic Tissues | Gentle Chemical (e.g., EGTA) [35] | Protocol-dependent | Preserves viability of rare/delicate cells; slower but gentler [35]. |
Experimental Protocols for Tissue Dissociation
This section outlines standard operating procedures for enzymatic dissociation of primary tissues and cultured cells. These are general templates that require empirical optimization for specific use cases [30].
General Enzymatic Dissociation of Adherent Cell Cultures
This protocol is suitable for trypsin, TrypLE, and other proteases [30].
- Aspiration: Remove and discard the spent cell culture media.
- Rinse: Wash the cell monolayer with a balanced salt solution without calcium and magnesium (e.g., DPBS). This removes residual divalent cations that inhibit trypsin.
- Digestion: Add the pre-warmed dissociation solution (e.g., 2-3 mL per 25 cm²). Ensure it covers the cell sheet completely. Incubate at 37°C.
- Monitoring: Gently rock the vessel and monitor under a microscope. Cells typically detach in 5-15 minutes. Tap the flask if necessary.
- Neutralization: When cells detach, add complete growth medium (containing serum, which inhibits trypsin) or a specific inhibitor.
- Collection: Pipette the suspension to disperse clumps. Transfer to a conical tube and centrifuge at 100 × g for 5-10 minutes.
- Resuspension: Discard supernatant and resuspend the cell pellet in fresh, pre-warmed complete medium.
- Counting: Determine viable cell density and percent viability (should be >90%) using an automated cell counter or hemocytometer [30].
Collagenase-Based Dissociation of Primary Tissue
This method is ideal for compact tissues like tumors, liver, and kidney [30].
- Mincing: With sterile instruments, mince the tissue into 3-4 mm pieces.
- Washing: Wash the tissue pieces several times with Hank's Balanced Salt Solution (HBSS) with calcium and magnesium, which are co-factors for collagenase activity.
- Digestion: Submerge tissue in sufficient HBSS with calcium and magnesium. Add collagenase to a final concentration of 50-200 U/mL. Supplementing with 3 mM CaCl₂ can increase efficiency.
- Incubation: Incubate at 37°C for 4-18 hours on a rocker platform for agitation.
- Dispersion & Filtration: Pass the digested mixture through a sterile 70-100 µm cell strainer to disperse cells and remove debris.
- Washing: Wash dispersed cells several times by centrifugation in HBSS without collagenase.
- Resuspension & Counting: Resuspend the final cell pellet in culture medium and determine viable cell density [30].
The following workflow diagram integrates these steps with the critical decision points for protocol optimization.
Advanced and Emerging Protocols
Recent innovations focus on improving viability and reducing artifacts. The Cryogenic Enzymatic Dissociation (CED) strategy for nuclei isolation from Formalin-Fixed Paraffin-Embedded (FFPE) tissues performs digestion at low temperatures. This protects nuclear membranes, maximizes RNA retention, and results in a tenfold increase in nuclei yield compared to conventional hyperthermic methods [23]. For single-cell transcriptomics, cold-active enzymes are also being explored to minimize alterations to the transcriptional landscape that can occur during prolonged incubation at 37°C [34].
The Scientist's Toolkit: Essential Reagents and Equipment
Successful tissue dissociation relies on a suite of specialized reagents and instruments. The following table details key solutions and their functions in a standard dissociation workflow.
Table 3: Essential Research Reagent Solutions for Tissue Dissociation
| Tool Category | Specific Examples | Function in Dissociation Workflow |
|---|---|---|
| Core Enzymes | Collagenase (C. histolyticum) [34], Trypsin [30], TrypLE Express [30], Dispase [30], Hyaluronidase [4] [34] | Target specific ECM components (see Table 1) to break down tissue structure and release individual cells. |
| Chelating Agents | EDTA, EGTA [4] [35] | Disrupts cell-cell junctions by chelating divalent cations (Ca²⁺, Mg²⁺) essential for adhesion proteins. |
| Buffers & Media | HBSS (with/without Ca²⁺/Mg²⁺) [30], DPBS [30], Cell Dissociation Buffer [30] | Provides a physiologically compatible environment for cells during dissociation. Salt-based non-enzymatic buffers offer a gentle alternative [30]. |
| Inactivation Reagents | Serum (FBS) [30], Soybean Trypsin Inhibitor [30] | Stops enzymatic activity post-detachment to prevent overtreatment and cell damage. |
| Specialized Kits | Commercial Nuclear Extraction Kits [23], Tumor Dissociation Kits [36] | Pre-optimized, standardized reagent blends for specific tissues or applications, ensuring reproducibility. |
| Mechanical Tools | GentleMACS Dissociator [4], Paddle Blenders (Stomachers) [34], Tissue Grinders [34], Bead Mill Homogenizers (Bullet Blender) [34] | Applies controlled physical force to mince and dissociate tissue, often used in combination with enzymes. |
| Incubation Equipment | Shaking Water Baths [34], Incubated Orbital Shakers [34] | Maintains precise, constant temperature with agitation to promote efficient and uniform enzymatic digestion. |
| Separation & Analysis | Cell Strainers (70-100 µm) [30], Centrifuges, Automated Cell Counters [30] | Removes undissociated debris, washes cells, and provides immediate feedback on yield and viability. |
Selecting the correct enzyme formulation is not a one-size-fits-all process but a critical, experiment-defining decision. A principled approach—one that considers the biochemical composition of the target tissue, the mechanism of action of available enzymes, and the stringent requirements of the downstream application—is fundamental to successful cell isolation. As the field advances, trends point toward greater standardization, automation, and the development of gentler, enzyme-free solutions like acoustic or electrical dissociation methods to better preserve cell surface markers and transcriptional fidelity [16] [4].
While this guide provides a structured framework, the dynamic nature of biological systems necessitates empirical optimization for each unique research context. By adhering to these principles, researchers can develop robust, reproducible dissociation protocols that ensure high cell viability, yield, and functional integrity, thereby laying a solid foundation for groundbreaking discoveries in basic biology and translational medicine.
Tissue dissociation into single-cell suspensions represents a critical initial step for a wide array of downstream applications in biomedical research and therapy development, including single-cell analysis, cell therapy manufacturing, and drug screening [4]. The process necessitates breaking down the complex architecture of the extracellular matrix (ECM) and disrupting cell-cell junctions that hold tissues together. While enzymatic (chemical) and mechanical approaches have traditionally been viewed as distinct methodologies, a growing body of evidence demonstrates that their strategic integration overcomes limitations inherent to either method when used alone [4] [37]. Enzymatic digestion alone often requires prolonged processing times—sometimes hours—which can compromise cell viability, activate stress responses, and potentially damage cell surface markers crucial for subsequent analysis [4] [38]. Conversely, exclusively mechanical methods can inflict substantial physical damage on cells, leading to reduced viability and viability, particularly in more delicate tissues [39].
The synergy between mechanical and chemical aids addresses these shortcomings. Mechanical disruption serves to increase the surface area of the tissue exposed to enzymatic action, thereby accelerating digestion kinetics and reducing the required incubation time [37]. Furthermore, gentle mechanical forces can help dislodge cells already loosened by enzymatic activity, minimizing the mechanical stress needed for final dissociation. This integrated approach is particularly vital for complex and dense tissues, such as solid tumors or fibrotic organs, where a single-method strategy frequently yields suboptimal results in terms of either cell yield, viability, or both [4]. Within the broader thesis on enzymatic cell dissociation principles, this whitepaper establishes that enzymatic action is most effective not as a standalone process, but as a core component of a meticulously designed hybrid workflow that leverages complementary mechanical forces.
Quantitative Comparison of Dissociation Technologies
Evaluating the performance of different dissociation methods requires a multi-faceted analysis of key metrics, including dissociation efficacy, cell viability, processing time, and tissue utilization rate. The following table synthesizes quantitative data from recent studies and advanced platforms to provide a comparative overview.
Table 1: Performance Metrics of Tissue Dissociation Methods
| Technology / Method | Tissue Type | Dissociation Efficacy | Cell Viability | Processing Time |
|---|---|---|---|---|
| Traditional Enzymatic-Mechanical [4] | Bovine Liver Tissue | 92% ± 8% | >90% | 15 min |
| Optimized Enzymatic Protocol [4] | Human Skin Biopsy | ~24,000 cells/4 mm punch | 92.75% | ~3 hours |
| Automated Mechanical Grinder [4] | Mouse Lung Tissue | 1-6×10^5 cells | 60%-80% | ~1 hour |
| Mixed Modal Microfluidic Platform [4] | Mouse Kidney Tissue | ~20,000 epithelial cells/mg | ~95% (epithelial) | 1-60 min |
| Electric Field Dissociation [4] | Human Glioblastoma | >5x higher than traditional | ~80% | 5 min |
| Ultrasound Sonication [4] | Bovine Liver Tissue | 53% ± 8% (sonication only) | 91%-98% | 30 min |
| Hypersonic Levitation & Spinning (HLS) [8] | Human Renal Cancer | High (90% tissue utilization) | 92.3% | 15 min |
The data reveals that advanced non-enzymatic or hybrid methods can achieve remarkable speed and preserve high viability. The Hypersonic Levitation and Spinning (HLS) method, for instance, demonstrates a standout performance with 90% tissue utilization in just 15 minutes while maintaining viability over 92% [8]. In contrast, traditional enzymatic protocols, while capable of high viability, often demand considerably longer processing times, which can be a critical bottleneck in time-sensitive applications [4]. Methods relying solely on mechanical force, such as automated grinders, frequently exhibit a trade-off, showing moderate efficacy but potentially lower and more variable viability [4] [39]. These quantitative comparisons underscore the necessity of selecting a method tailored to the specific tissue and downstream application, with combined methods frequently offering a superior balance of efficiency and cell health.
Experimental Protocols for Integrated Dissociation
This section provides detailed methodologies for implementing combined mechanical and chemical dissociation, as cited in recent literature.
Protocol: Optimization of a Clinically Relevant Chemical-Mechanical Workflow
This protocol, adapted from a study on bovine liver and breast cancer cells, outlines a rapid and efficient combined workflow [4].
- Objective: To achieve high-yield, high-viability single-cell suspensions from dense tissues with a short processing time.
- Materials:
- Tissue Sample: Fresh or freshly frozen tissue specimen.
- Enzymatic Cocktail: Typically a blend of collagenase (e.g., 1-2 mg/mL) and dispase (e.g., 1-2 mg/mL) in a buffered saline solution containing calcium and magnesium [4] [37].
- Mechanical Disruption Tool: Laboratory vortexer with tube attachment or a gentleMACS Dissociator (Miltenyi Biotec) or similar automated system.
- Incubation System: Temperature-controlled orbital shaker or water bath set to 37°C.
- Procedure:
- Tissue Preparation: Mince the tissue into approximately 1-2 mm³ fragments using sterile scalpels or razor blades in a small volume of dissociation buffer. This initial mechanical step is crucial for increasing surface area.
- Enzymatic Incubation: Transfer the minced tissue into a tube containing the pre-warmed enzymatic cocktail. Use a volume sufficient to submerge the tissue fragments (e.g., 4 mL per 100 mg of tissue) [37].
- Combined Digestion & Agitation: Incubate the tube at 37°C with constant, vigorous agitation. This can be achieved on an orbital shaker (e.g., 250 rpm) or by using a vortexer with a custom attachment that provides intermittent mixing (e.g., 15-second pulses every 5 minutes). The simultaneous application of mechanical agitation significantly enhances enzyme penetration and dispersal of dissociated cells.
- Reaction Termination: After 15 minutes of incubation, neutralize the enzymatic reaction by adding a buffer containing serum or a specific enzyme inhibitor (e.g., FBS for trypsin).
- Filtration and Washing: Pass the cell suspension through a sterile cell strainer (70-100 µm) to remove any remaining undissociated tissue aggregates. Centrifuge the filtrate to pellet the cells and wash twice with PBS or a cold culture medium.
- Assessment: Resuspend the final cell pellet and determine cell count and viability using a trypan blue exclusion assay or an automated cell counter.
Protocol: Microfluidic Mixed-Modal Platform for Complex Tissues
This methodology leverages a microfluidic device to integrate digestion and mechanical shear in a controlled environment, suitable for small tissue samples like mouse kidney and breast tumors [4].
- Objective: To dissociate complex tissues with high precision and preserve specific, sensitive cell populations (e.g., leukocytes, endothelial cells).
- Materials:
- Microfluidic Device: A mixed-modal microfluidic platform with integrated chambers and channels designed for tissue processing.
- Peristaltic or Syringe Pump: For controlled flow of reagents and tissue suspension.
- Enzymatic Solution: Collagenase-based solution, often at a lower concentration than used in bulk protocols.
- Procedure:
- Device Priming: Prime the microfluidic channels and chamber with dissociation buffer.
- Sample Loading: Load the pre-minced tissue fragments into the device's digestion chamber.
- Circulatory Digestion: Continuously circulate the enzymatic solution through the chamber containing the tissue fragments for a set period (20-60 minutes). The flow rate is carefully controlled to provide gentle mechanical shear without damaging cells.
- Continuous Harvesting: As cells are dissociated, they are carried away by the flow to an output reservoir, preventing over-digestion.
- Collection: Collect the output cell suspension from the device. The suspension is typically ready for filtration and washing without further processing.
The following diagram illustrates the logical decision-making process for selecting an appropriate combined dissociation strategy based on tissue properties and experimental goals.
Diagram 1: Strategy for selecting a combined dissociation method. This workflow guides researchers in choosing a protocol based on their specific tissue type and experimental requirements, emphasizing that no single method is universally optimal.
The Scientist's Toolkit: Essential Reagents and Equipment
Successful implementation of combined dissociation strategies relies on a core set of reagents, enzymes, and instruments. The selection of each component must be tailored to the specific tissue and the need to preserve cellular integrity and function.
Table 2: Research Reagent Solutions for Combined Dissociation
| Item Category | Specific Examples | Function & Rationale |
|---|---|---|
| Enzymes | Collagenase D [37] | Digests native collagen; preferred when preserving cell surface marker integrity is crucial for functional assays. |
| Trypsin [38] [37] | A harsh serine protease efficient for cell detachment; can damage surface antigens. Use for robust tissues. | |
| Dispase [37] | Gentler enzyme that cleaves fibronectin and collagen IV; suitable for delicate cells. | |
| Hyaluronidase [4] [37] | Degrades hyaluronic acid in the ECM; often used in enzyme cocktails for more complete digestion. | |
| Chemical Aids | EDTA/EGTA [38] | Chelating agents that bind divalent cations, disrupting cell adhesions. Often combined with enzymes. |
| Mechanical Instruments | Orbital Shakers [37] | Provides consistent agitation during enzymatic incubation, combining mechanical and chemical action. |
| Tissue Grinders [37] | Provides initial mechanical mincing or gentle homogenization of tissue pre- or post-enzymatic digestion. | |
| Paddle Blenders (Stomachers) [37] | Offers a standardized mechanical dissociation method with or without enzymes, minimizing user variability. | |
| Advanced Platforms | Microfluidic Dissociation Chips [4] | Integrates enzymatic and shear-based dissociation in a controlled, automated microenvironment. |
| Hypersonic Levitation (HLS) [8] | A non-contact method using acoustic streaming for "press-and-rotate" tissue dissociation, maximizing viability. |
Discussion and Implementation Guide
The integration of mechanical and chemical dissociation aids is not a mere sequential application but a synergistic process that requires careful optimization. Key parameters to consider include the type and concentration of enzymes, the duration and temperature of incubation, and the precise nature and intensity of the mechanical force applied [37]. For instance, increasing digestion buffer volume can improve cell viability by reducing local concentrations of harmful metabolites, while aggressive mechanical grinding may need to be traded for gentler, prolonged agitation to preserve rare cell populations [8] [37].
A critical, often overlooked aspect is the impact of the dissociation method on subsequent cellular behavior. Research demonstrates that the choice of dissociation reagent (e.g., trypsin-EDTA vs. enzyme-free buffer) can significantly alter how cells re-adhere to surfaces, potentially leading to misleading conclusions in adhesion studies or functional assays [38]. Therefore, the optimized protocol must be validated not just for yield and viability, but also for its suitability for the intended downstream application, whether it be single-cell RNA sequencing, cell therapy manufacturing, or drug screening [16] [37].
The following workflow diagram outlines the key steps and decision points in a generalized, optimized combined dissociation protocol.
Diagram 2: Generalized workflow for a combined mechanical-chemical dissociation. This protocol highlights the iterative and integrated nature of the process, where mechanical and chemical steps are interwoven for maximum efficiency.
The paradigm of tissue dissociation is decisively shifting from a choice between enzymatic or mechanical methods to a mandatory integration of both. As this whitepaper has detailed, combining mechanical and chemical aids is the most reliable pathway to achieving high-efficiency tissue dissociation that delivers high yields of viable, functional cells. This integrated approach directly addresses the pressing bottlenecks in manufacturing cell-based therapies and conducting robust single-cell analyses [4]. The future of this field lies in the development of standardized, automated, and closed-system platforms that seamlessly combine these modalities—exemplified by emerging technologies like microfluidic mixed-modal devices and hypersonic levitation systems [4] [8]. For researchers and drug development professionals, mastering these combined protocols is not merely an technical optimization but a fundamental requirement for generating reproducible, high-quality data and therapeutics in the evolving landscape of precision medicine and regenerative medicine.
Tissue dissociation into single-cell suspensions is a critical foundational technique in molecular cell biology, serving as the gateway to a plethora of downstream applications including single-cell RNA sequencing (scRNA-seq), flow cytometry, and cell culture [4] [8]. The process entails breaking down the extracellular matrix (ECM) and cell-cell junctions that hold tissues together, which is traditionally achieved through a combination of mechanical disruption and enzymatic digestion [4] [40]. However, the dissociation process itself represents one of the greatest sources of technical variability and potential artifacts in single-cell studies, directly impacting data quality, cell viability, and experimental reproducibility [4] [41].
Within the context of enzymatic cell dissociation research, a fundamental principle is that the method of dissociation must be carefully balanced against the requirements of the intended downstream application. Suboptimal dissociation can introduce significant experimental artifacts, including reduced cell viability, destruction of cell surface epitopes, altered transcriptional profiles, and loss of rare cell populations [4] [41] [42]. This technical guide examines the multifaceted impact of tissue dissociation methodologies on three critical downstream applications, providing researchers with evidence-based strategies to optimize protocol selection for their specific research objectives.
Principles and Challenges of Tissue Dissociation
Fundamental Approaches
The dissociation of solid tissues into single-cell suspensions employs three primary approaches, each with distinct mechanisms and applications:
Mechanical Dissociation: This simplest method utilizes physical force through cutting, crushing, or pulverizing tissue with instruments like scalpels, homogenizers, or through trituration [43] [40]. While fast and cost-effective, mechanical dissociation can inflict significant mechanical stress on cells, leading to membrane damage, reduced viability, and inconsistent results between operators [8] [43]. It is most appropriate for loosely associated tissues like bone marrow, spleen, and lymph nodes [43].
Enzymatic Dissociation: This approach employs specific proteolytic enzymes to digest ECM components and cell-cell junctions [40] [44]. Common enzymes include collagenase (cleaves collagen), dispase (targets fibronectin and collagen IV), trypsin (digestive protease), papain (cysteine protease), hyaluronidase (degrades hyaluronic acid), and DNase (cleaves DNA from lysed cells) [4] [40] [44]. Enzymatic methods generally provide higher cell viability and yield but require optimization of enzyme combinations, concentrations, and incubation times [43] [44].
Chemical Dissociation: This gentle approach uses chelating agents like EDTA or EGTA that bind cations maintaining intercellular bonds, causing tissue dissociation without enzymatic activity [43]. While preserving cell surface markers, chemical dissociation is slower and produces variable results depending on tissue type and chemical conditions [43].
Key Challenges and Limitations
Conventional dissociation methods face several significant challenges that can compromise downstream applications:
Cell Viability and Yield: Aggressive mechanical or enzymatic treatment can damage cell membranes, reducing viability and overall cell yield [4] [42]. Different cell types exhibit varying sensitivity to dissociation methods, potentially skewing cellular representation [41].
Molecular Integrity: Enzymatic digestion, particularly with broad-spectrum proteases, can damage cell surface proteins and epitopes, compromising flow cytometry and cell sorting applications [4] [43]. Extended digestion times can also activate cellular stress pathways, altering transcriptional profiles and introducing artifacts in scRNA-seq data [4] [44].
Temporal Considerations: Enzymatic digestion often requires substantial time—ranging from hours to overnight processing—which delays analyses and increases contamination risks [4]. Some protocols attempt to shorten processing times at the expense of reduced recovery or viability [4].
Standardization Issues: The heterogeneous nature of tissues has led to independently developed protocols for different tissue types, creating a lack of standardized approaches that challenges reproducibility and cross-study comparisons [4].
Impact on Specific Downstream Applications
Single-Cell RNA Sequencing
The quality of scRNA-seq data is profoundly influenced by the dissociation process, which must preserve RNA integrity while minimizing the introduction of transcriptional artifacts [41]. Different stress factors—including mechanical damage, digestion temperature, and prolonged enzymatic treatment—can skew cellular transcriptomes [41]. For instance, extended digestion times may increase cell yield but negatively impact viability and alter gene expression profiles [41].
Optimized protocols must balance cell release from difficult-to-digest tissues against cellular damage during processing. Research demonstrates that tailored dissociation methods significantly improve scRNA-seq outcomes across various tissue types:
Table 1: Optimized Dissociation Protocols for scRNA-seq Applications
| Tissue Type | Optimal Enzymes | Key Protocol Details | Performance Metrics | Reference |
|---|---|---|---|---|
| Human Skin | Dispase II + Collagenase IV + DNase I | Pre-treatment with Dispase II (10 U/mL, 2h, 37°C) followed by Collagenase IV (300 U/mL) + DNase I (5 U/mL) for 1h | ~24,000 cells/4mm biopsy; 92.75% viability; successful rare cell population identification | [41] |
| Brain Tumors | Neutral Protease (NP) | NP (0.11 DMC U/mL) for 2h at 37°C or overnight at ambient temperature | 93% viability in gliomas; significantly less cellular debris; preserved RNA integrity | [42] |
| FFPE Tissues | Cryogenic Enzymatic Dissociation (CED) | Proteinase K in sarcosyl solution at low temperature | 10x increase in nuclei yield; enhanced gene detection sensitivity; reduced mitochondrial contamination | [23] |
| Endocrine Tumors | Collagenase IV or MTDK | Tissue-specific timing: 20min for adrenal medullary, 7-15min for PitNETs | Optimized viability through tailored incubation times; preservation of cellular integrity | [44] |
The development of advanced technologies like microfluidic tissue dissociation platforms has demonstrated promising results, processing samples in 20-60 minutes with viability rates of 70-90% across various cell types [4]. Similarly, enzyme-free approaches utilizing electrical (5-minute processing with 90% viability) or ultrasound dissociation (30-minute processing with 91-98% viability) offer alternatives that minimize enzymatic-induced artifacts [4].
Flow Cytometry
Flow cytometry applications require high-quality single-cell suspensions with preserved cell surface epitopes and minimal cellular debris [42]. Enzymatic dissociation presents particular challenges for flow cytometry, as proteases can damage or remove the very surface markers used for cell identification and sorting [43]. Mechanical dissociation methods, while faster, can generate increased cellular debris and subcellular fragments that interfere with accurate analysis by causing background noise and clogging the flow cytometer's nozzle [43] [42].
A study on brain tumor dissociation demonstrated that the enzyme Neutral Protease (NP) produced cell mixtures with significantly less cellular debris and DNA release compared to collagenase, papain, dispase, and other common enzymes [42]. This improved dissociation quality directly enhances flow cytometry data quality by reducing background signal and improving detection sensitivity [42].
Post-dissociation purification steps, including debris removal and red blood cell lysis, are particularly important for flow cytometry applications [44]. Buoyancy-activated cell sorting (BACS) using microbubbles has emerged as a gentle alternative to magnetic or fluorescence-based sorting methods, preserving cell physiology while delivering highly enriched target populations [43].
Cell Culture
The success of primary cell culture and the establishment of cell lines depends critically on the dissociation method's ability to preserve cell viability, functionality, and proliferative capacity [40] [44]. Enzymatic dissociation must be optimized to ensure cells remain not only viable but also functionally competent for in vitro growth and experimentation [44].
Primary cells are especially sensitive to dissociation conditions due to their more fastidious growth requirements and limited lifespan compared to immortalized cell lines [40]. Research on endocrine tumors demonstrated that optimal dissociation parameters vary significantly by tissue type, with adrenal medullary tumors showing highest viability with 20-minute dissociation, while other tissues required different durations [44].
The choice of specific enzymes significantly impacts subsequent cell culture success. For example, papain has proven highly effective for neural tissue dissociation, resulting in greater efficiency and cell viability than other enzymes, making it ideal for primary neuronal cultures [40] [42]. Similarly, collagenase type I is recommended for liver, epithelial, adrenal tissue and adipocytes, while collagenase type II works better for muscle, thyroid, heart, bone, and cartilage [40].
Table 2: Enzyme Selection Guide for Cell Culture Applications
| Enzyme | Mechanism of Action | Optimal Tissue Applications | Considerations for Cell Culture | |
|---|---|---|---|---|
| Collagenase | Breaks down native triple-helical collagen | Type I: Liver, epithelial, adrenal, adipocytes; Type II: Muscle, heart, bone, cartilage | Preserves cell surface markers; different types show tissue specificity | [40] |
| Trypsin | Digestive protease that breaks down proteins mediating cell adhesion | Cell monolayers; often used with EDTA | Can damage surface epitopes; may be too harsh for sensitive primary cells | [40] [44] |
| Papain | Cysteine peptidase C1 protease that degrades myofibrillar proteins | Neural tissue; central nervous system | Higher efficiency and viability for neuronal cultures; gentle on sensitive cells | [40] [42] |
| Neutral Protease | Metalloprotease hydrolyzing peptide bonds of non-polar amino acids | Brain tumors; neural tissues | Preserves highest viability (93% in gliomas); minimal debris; clinically available | [42] |
| Dispase | Non-specific metalloprotease cleaving fibronectin and collagen | Epithelial cells; endocrine tumors | Suitable for isolating epithelial cells; less damaging than trypsin | [44] |
Emerging Technologies and Methodologies
Advanced Dissociation Platforms
Recent technological innovations have significantly advanced tissue dissociation capabilities:
Microfluidic Dissociation Platforms: These systems integrate enzymatic and mechanical dissociation within microfluidic channels, processing samples in 20-60 minutes with viability rates of 70-90% across various tissue types [4]. They offer improved consistency and reduced processing times but can face challenges with channel clogging from tissue fragments [4] [8].
Hypersonic Levitation and Spinning (HLS): This revolutionary contact-free approach utilizes a triple-acoustic resonator probe to levitate and spin tissue samples, generating microscale "liquid jets" that exert precise hydrodynamic forces [8]. HLS achieves 90% tissue utilization in just 15 minutes while maintaining 92.3% cell viability and better preserving rare cell populations compared to traditional methods [8].
Non-Enzymatic Alternatives: Electrical dissociation (95% efficacy in 5 minutes) and ultrasound dissociation (53-72% efficacy in 30 minutes) offer enzyme-free options that avoid protease-related damage to cell surface markers [4].
Specialized Applications
FFPE Tissue Dissociation: The development of Cryogenic Enzymatic Dissociation (CED) has revolutionized nuclei extraction from formalin-fixed, paraffin-embedded (FFPE) tissues, achieving a tenfold increase in nuclei yield with significantly reduced hands-on time [23]. This method minimizes secondary RNA degradation and preserves intranuclear transcripts, enabling superior gene detection sensitivity and reduced contamination in snRNA-seq applications [23].
Multi-Omics Integration: Foundation models like Nicheformer are now being trained on both dissociated single-cell and spatial transcriptomics data, highlighting the critical importance of dissociation quality for spatially-aware cellular analysis [45]. Models trained only on dissociated data fail to recover the complexity of spatial microenvironments, underscoring the need for dissociation methods that preserve biological context [45].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for Tissue Dissociation
| Reagent/Kit | Composition | Primary Applications | Function | |
|---|---|---|---|---|
| Multi Tissue Dissociation Kit (MTDK) | Enzyme D (undefined specific composition) | Adrenal gland neoplasms, thyroid carcinomas, pituitary neuroendocrine tumors | Optimized enzyme cocktail for multiple endocrine tissues | [44] |
| Neural Tissue Dissociation Kit (NTDK) | Enzyme A (undefined specific composition) | Adrenal gland neoplasms, neural tissues | Specialized formulation for neural and neuroendocrine tissues | [44] |
| Collagenase IV | Metalloprotease from Clostridium histolyticum | Brain tumors, endocrine tumors, various solid tissues | Cleaves native triple-helical collagen in ECM | [41] [44] |
| Dispase II | Non-specific metalloprotease from Bacillus polymyxa | Skin dissociation, epithelial cell isolation | Targets fibronectin and collagen IV in basement membrane | [41] |
| Neutral Protease | Metalloprotease from Clostridium histolyticum | Brain tumors, neural tissues | Hydrolyzes peptide bonds without collagenolytic activity | [42] |
| DNase I | Endonuclease | All tissue types requiring reduction of viscosity | Degrades DNA released from lysed cells to prevent clumping | [41] [44] |
| Proteinase K | Serine protease | FFPE tissue dissociation for snRNA-seq | Digests proteins in cross-linked FFPE samples | [23] |
Experimental Workflows and Methodologies
Standardized Dissociation Workflow
The following diagram illustrates a generalized optimized workflow for tissue dissociation applicable to multiple downstream applications:
Decision Framework for Dissociation Method Selection
The selection of an appropriate dissociation strategy requires consideration of multiple factors, as illustrated in the following decision framework:
Tissue dissociation methodology profoundly influences the success and interpretation of downstream applications including scRNA-seq, flow cytometry, and cell culture. The optimal dissociation strategy must be carefully tailored to both the tissue type and the specific requirements of the intended application, balancing cell yield against viability, molecular integrity, and functional preservation. Emerging technologies such as microfluidic platforms, hypersonic levitation, and cryogenic enzymatic dissociation offer promising avenues for overcoming limitations of conventional methods. As single-cell and spatial omics technologies continue to advance, the development of standardized, validated dissociation protocols will be essential for generating reproducible, biologically meaningful data across research and clinical applications.
Solving Common Challenges: A Guide to Troubleshooting and Optimizing Yield and Viability
Tissue dissociation into single-cell suspensions represents a critical foundational technique for cell therapy manufacturing, single-cell analysis, and numerous downstream applications in biomedical research [4] [46]. The process of preparing single-cell suspensions stands as one of the greatest sources of technical errors and variations in single-cell studies, making optimized dissociation protocols essential for experimental success [4]. Within the broader thesis on enzymatic cell dissociation principles, this guide addresses the fundamental challenge facing researchers: achieving high cell yield and viability while avoiding the dual pitfalls of under-digestion and over-digestion. These dissociation artifacts can significantly distort downstream analyses, compromising data integrity and potentially leading to erroneous biological conclusions [4] [44]. The complex morphological features of tissues—particularly tumors with their dense extracellular matrix networks, specialized structures, and secondary degenerative changes—present substantial challenges for enzymatic dissociation protocols [44]. This technical guide provides a systematic framework for diagnosing and resolving the most common dissociation problems, enabling researchers to produce high-quality single-cell suspensions that faithfully preserve biological truth.
Fundamental Principles of Tissue Dissociation
Mechanical and Enzymatic Basis of Dissociation
Tissue dissociation fundamentally involves disrupting two types of biological connections: cell-cell junctions and cell-extracellular matrix (ECM) adhesions [4] [47]. The extracellular matrix provides structural support through a three-dimensional fibrous network comprising proteins (particularly collagens), proteoglycans, glycosaminoglycans, and metalloproteinases [47]. Cell-cell adhesion occurs through various junctions including gap junctions, tight junctions, adherence junctions, and desmosomes, while cell-matrix adhesion primarily occurs through integrin-mediated focal adhesions, myotendinous junctions, and hemidesmosomes [47]. Effective dissociation protocols must successfully dismantle these connections while preserving cellular integrity, viability, and surface markers essential for downstream applications.
Enzymatic approaches target specific components of this complex architecture. Collagenases hydrolyze collagens, the most abundant proteins in the body that hold tissues together [48]. Serine proteases like trypsin cleave peptide bonds, while dispases target fibronectin and collagen IV, and hyaluronidases degrade hyaluronic acid in the ECM [4] [48]. These enzymatic methods are typically combined with mechanical approaches—including mincing, shaking, filtration, or more advanced techniques like acoustic streaming—to achieve complete tissue dissociation [4] [8] [48]. The optimal balance between enzymatic and mechanical forces varies significantly across tissue types and must be carefully determined through systematic optimization.
Critical Factors Influencing Dissociation Outcomes
The following workflow outlines the key decision points and optimization parameters in developing an effective tissue dissociation protocol:
Diagnostic Framework: Identifying and Resolving Dissociation Problems
Problem 1: Low Cell Yield
Low cell yield remains one of the most frequent challenges in tissue dissociation, resulting in insufficient material for downstream applications and potential loss of rare cell populations. This problem typically stems from inadequate disruption of the extracellular matrix or suboptimal processing conditions that fail to liberate cells effectively.
Root Causes and Diagnostic Indicators:
- Insufficient enzymatic activity: Collagen-rich tissues require adequate collagenase concentrations and activity to degrade predominant ECM components [44] [48].
- Inadequate mechanical disruption: Dense tissues often require complementary mechanical forces to enhance enzyme penetration and physical separation [4] [8].
- Suboptimal enzyme selection: Mismatch between enzyme specificity and tissue ECM composition (e.g., using trypsin for collagen-rich matrices) [44] [49].
- Excessive debris removal: Overly aggressive filtration or purification steps can discard viable cells along with debris [44].
Evidence-Based Solutions: Research demonstrates that enzymatic cocktails targeting multiple ECM components simultaneously can significantly improve yield. In colorectal cancer organoid generation, collagenase and hyaluronidase demonstrated superior tissue dissociation, yielding higher total cell counts compared to TrypLE and trypsin [49]. For endocrine tumors, optimal dissociation required tissue-specific protocols, with adrenal medullary tumors achieving best results with 20-minute incubation times [44].
Advanced non-enzymatic technologies also offer promising alternatives. The Hypersonic Levitation and Spinning (HLS) approach, which utilizes acoustic streaming to generate precise hydrodynamic forces, achieved 90% tissue utilization in just 15 minutes—significantly outperforming traditional methods [8]. Similarly, optimized microfluidic platforms can process tissue fragments with high efficiency, achieving yields of approximately 400,000 total cells per mg of mouse kidney tissue [4].
Table 1: Troubleshooting Low Cell Yield
| Root Cause | Diagnostic Indicators | Recommended Solutions |
|---|---|---|
| Inadequate Enzyme Selection | Low cell count across multiple batches; tissue fragments remain after digestion | Use collagenase for collagen-rich tissues (≥1 mg/mL); implement enzyme cocktails for complex tissues [44] [49] |
| Insufficient Mechanical Force | Visible tissue chunks in suspension; variability between operators | Combine enzymatic digestion with orbital shaking (200 rpm); consider bead mill homogenization at low speeds [4] [48] |
| Suboptimal Incubation Parameters | Inconsistent yields between experiments; time-dependent decline in viability | Optimize incubation time (20-35 min for most tissues); increase digestion buffer volume (4 mL per 100 mg tissue) [44] [48] |
| Excessive Purification Loss | High initial yield followed by significant loss after filtration | Use debris removal kits instead of filtration; implement gentle centrifugation (300g for 2 min) [44] |
Problem 2: Low Cell Viability
Low cell viability compromises virtually all downstream applications, particularly functional assays, cell culture, and therapeutic applications where metabolic activity and cellular integrity are paramount. Viability loss primarily occurs through apoptosis, necrosis, or mechanical damage during the dissociation process.
Root Causes and Diagnostic Indicators:
- Prolonged enzymatic exposure: Extended incubation times progressively damage cell membranes and intracellular components [44] [48].
- Enzyme-induced toxicity: Certain enzymes, particularly serine proteases like trypsin, demonstrate higher cytotoxicity compared to gentler alternatives [48] [47].
- Mechanical shear stress: Aggressive mechanical methods including high-speed homogenization or vigorous pipetting physically disrupt cell membranes [4] [8].
- Temperature stress: Extended processing at 37°C can activate cellular stress responses and apoptotic pathways [23].
Evidence-Based Solutions: Studies on endocrine tumors revealed that dissociation time significantly impacts viability. For adrenal medullary tumors, reducing incubation time to 20 minutes proved optimal, while different tumor types required specific enzyme combinations—Collagenase IV or MTDK enzyme kits with post-dissociation debris removal procedures significantly enhanced viability for adrenocortical tumors [44].
Comparative enzyme studies in colorectal cancer organoid generation demonstrated that TrypLE and trypsin-EDTA better preserved initial cell viability compared to collagenase and hyaluronidase, though the latter enzymes ultimately supported superior organoid formation [49]. This highlights the important distinction between immediate viability and functional preservation.
Advanced non-contact technologies show particular promise for viability preservation. The HLS method achieved 92.3% viability in human renal cancer tissue—significantly higher than conventional methods—by generating controlled hydrodynamic forces without direct physical contact [8]. Similarly, cryogenic enzymatic dissociation (CED) strategies better preserve nuclear integrity and RNA quality in FFPE tissues by maintaining low temperatures throughout processing [23].
Table 2: Troubleshooting Low Cell Viability
| Root Cause | Diagnostic Indicators | Recommended Solutions |
|---|---|---|
| Prolonged Enzymatic Exposure | Time-dependent viability loss; increased apoptotic markers | Reduce incubation time (7-15 min for delicate tissues); implement real-time monitoring [44] |
| Enzyme Cytotoxicity | Viability loss specific to enzyme type; surface marker degradation | Use gentler enzymes (Collagenase D, dispase); incorporate enzyme inhibitors with precise timing [48] |
| Mechanical Damage | Cellular debris; membrane damage visible in microscopy | Adopt non-contact methods (acoustic levitation); reduce homogenization speed; use larger bore pipettes [8] |
| Oxidative/Temperature Stress | ROS accumulation; heat shock protein activation | Use antioxidant supplements; lower processing temperatures; implement cold-active enzymes [23] |
Problem 3: Over-Digestion and Cellular Damage
Over-digestion represents a particularly insidious problem because it can preserve cell count and initial viability while compromising cellular function, surface markers, and transcriptional profiles. This creates artifacts that distort downstream analyses while providing false confidence in data quality.
Root Causes and Diagnostic Indicators:
- Excessive proteolytic activity: Uncontrolled enzyme action cleaves surface receptors, antigens, and critical membrane proteins [4] [47].
- Destruction of epitopes: Key antigenic determinants for flow cytometry or sorting are degraded, reducing detection sensitivity and resolution [48].
- Transcriptional artifacts: Extended processing at 37°C activates stress response pathways that alter mRNA expression profiles [4] [23].
- Functional impairment: Cellular signaling, metabolic activity, and secretory functions are compromised despite maintained membrane integrity [47].
Evidence-Based Solutions: Research highlights that over-digestion effects are highly cell-type dependent. Immune cells and neurons demonstrate particular sensitivity to prolonged enzymatic treatment, requiring optimized protocols with reduced exposure times [4] [48]. For flow cytometry applications where surface antigen preservation is critical, collagenase D is recommended due to its superior ability to maintain protein integrity compared to other collagenase variants or trypsin [48].
Novel approaches to minimize over-digestion include cryogenic enzymatic dissociation, which maintains processing at low temperatures to better preserve nuclear membranes and RNA integrity while still achieving effective dissociation [23]. Similarly, enzyme-free methods such as acoustic dissociation or mechanical approaches using paddle blenders can eliminate proteolytic damage entirely, though they require careful optimization to avoid alternative sources of cellular stress [8] [48].
The following decision tree provides a systematic approach to diagnosing dissociation problems based on experimental outcomes:
Advanced Methodologies: Protocols and Technical Approaches
Optimized Dissociation Protocol for Complex Tissues
Based on current research, the following integrated protocol represents best practices for dissociating complex tissues while minimizing artifacts:
Step 1: Tissue Preparation
- Collect fresh tissue in cold preservation solution (e.g., Miltenyi Biotec Tissue Storage Solution or Advanced DMEM/F12 with HEPES and antibiotics) [44] [49].
- Process within 1-2 hours of collection, minimizing ischemic time.
- Remove necrotic, hemorrhagic, and fatty tissues when possible.
- Rinse with ice-cold DPBS containing antibiotics (penicillin/streptomycin, amphotericin B) until supernatant is clear [49].
Step 2: Mechanical Size Reduction
- Mince tissue into 0.5-1 mm fragments using sterile scalpels or razor blades on a chilled surface.
- For microfluidic platforms, additional mincing to <0.5 mm may be necessary [4].
- Consider initial gentle mechanical disruption using paddle blenders or tissue grinders for particularly dense tissues [48].
Step 3: Enzymatic Dissociation
- Prepare enzyme solution tailored to tissue type:
- Use sufficient buffer volume (recommended 4 mL per 100 mg tissue) [48].
- Incubate at 37°C with continuous agitation (200 rpm) for tissue-specific duration:
Step 4: Reaction Termination and Cell Collection
- Neutralize enzymes with excess cold wash buffer containing 10% FBS [49].
- Pass cell suspension through 70-100 μm filters to remove undigested fragments.
- Centrifuge at 300g for 2-5 minutes at 4°C [49].
- Resuspend pellet in appropriate buffer for downstream application.
Step 5: Purification and Quality Assessment
- Implement debris removal steps using commercial kits (e.g., Miltenyi Biotec Debris Removal Solution) [44].
- Perform red blood cell lysis if necessary using ammonium chloride or commercial kits.
- Assess viability via trypan blue exclusion or fluorescent methods (7-AAD) [49].
- Quantify yield and viability using automated cell counters or flow cytometry.
Experimental Validation Methods
Rigorous validation of dissociation outcomes requires multiple complementary approaches:
Viability Assessment:
- Trypan Blue Exclusion: Simple, rapid assessment of membrane integrity; count >15,000 cells for statistical reliability [49].
- 7-AAD Flow Cytometry: Fluorescent-based method providing quantitative viability data across heterogeneous populations [49].
- Functional Assays: Mitochondrial activity (MTT), apoptosis markers (Annexin V), or metabolic dyes for functional validation.
Yield and Purity Quantification:
- Hemocytometer Counts: Manual counting with Neubauer chambers for direct visualization.
- Automated Cell Counters: Cedex, Countess, or similar instruments for rapid, reproducible quantification.
- Flow Cytometry: Bead-based absolute counting methods for highest precision.
Functional Integrity Assessment:
- Surface Marker Preservation: Flow cytometry analysis of critical antigens (e.g., CD133, LGR5 for stem cells) [49].
- Transcriptional Integrity: RNA quality assessment (RIN) and stress gene expression profiling [4] [23].
- Functional Capacity: Organoid formation efficiency, colony formation assays, or stimulus response tests [49].
The Scientist's Toolkit: Essential Reagents and Technologies
Table 3: Research Reagent Solutions for Tissue Dissociation
| Reagent/Technology | Function | Application Notes |
|---|---|---|
| Collagenase Type IV | Degrades native collagen while preserving cell surface markers | Optimal for endocrine tumors; use at 2 mg/mL in HBSS with FBS [44] |
| Multi Tissue Dissociation Kit (MTDK) | Enzyme cocktail for simultaneous targeting of multiple ECM components | Effective for adrenal, thyroid tissues; 25 μL per sample [44] |
| TrypLE Express | Recombinant fungal trypsin replacement with gentler activity | Superior viability preservation for colorectal cancer; less damaging than animal trypsin [49] |
| Hyaluronidase Type IV-S | Targets hyaluronic acid in extracellular matrix | Enhances penetration of other enzymes; effective in CRC at 1 mg/mL [49] |
| Debris Removal Solution | Density-based separation of viable cells from debris | Critical for viability improvement in adrenocortical tumors [44] |
| Hypersonic Levitation (HLS) | Non-contact dissociation via acoustic streaming | 92.3% viability; 90% tissue utilization in 15 minutes [8] |
| Cryogenic Enzymatic Dissociation | Low-temperature nucleus isolation for FFPE tissues | Prevents RNA degradation; 10x higher yield vs. mechanical methods [23] |
| Microfluidic Platforms | Integrated dissociation and filtration with precise control | ~400,000 cells/mg kidney tissue; 60-90% viability across cell types [4] |
Tissue dissociation remains both an art and a science, requiring careful balancing of competing priorities to achieve high yields, excellent viability, and minimal artifacts. The principles outlined in this diagnostic guide provide a systematic framework for addressing the most common challenges in single-cell suspension preparation. As research advances, several emerging technologies show particular promise for overcoming current limitations. Non-contact methods like Hypersonic Levitation and Spinning demonstrate that precise hydrodynamic forces can achieve rapid dissociation while preserving delicate cellular structures [8]. Cryogenic approaches address the critical challenge of transcriptional artifact formation during processing [23]. Additionally, integrated microfluidic platforms continue to evolve toward higher throughput capabilities while maintaining precise control over dissociation parameters [4].
The future of tissue dissociation lies in the development of standardized, validated systems that enable reproducible dissociation across laboratories and tissue types. Such standardization is particularly crucial for clinical applications where cell products must meet rigorous regulatory standards. By understanding the fundamental principles of dissociation biology and applying the diagnostic strategies presented here, researchers can significantly improve the quality and reliability of their single-cell data, ultimately advancing our understanding of cellular heterogeneity and function in health and disease.
Tissue dissociation into single-cell suspensions is a critical foundational technique for cell therapy manufacturing, single-cell analysis, and downstream processing in modern biological research [4]. The process is traditionally performed via enzymatic and mechanical dissociation of tissue, yet conventional methods face significant challenges regarding cell viability, yield, processing time, and potential for introducing artifacts that distort downstream analyses [4]. The current bottleneck in manufacturing tissue-engineered and cell-based regenerative medicine therapies is the lack of rigorous, standardized, and validated systems that enable reproducible dissociation of tissues into optimal cell populations [4].
Enzymatic dissociation, particularly using collagenase-based mixtures, presents unique challenges due to the inherent variability of traditional enzyme products. The most common approach involves pre-qualifying available "off the shelf" traditional crude or enriched collagenase products by evaluating several lots to select a "good lot" that effectively isolates cells of interest [50]. Once identified, researchers typically purchase enough of the good lot to last a year or more, creating consistency challenges for long-term research projects [50]. This variability stems from the minimal lot consistency of traditional collagenase products, where each lot is unique in its composition and performance characteristics [50].
Theoretical Foundation of the RDMTDE Framework
Core Principles of Enzyme-Mediated Cell Isolation
The Rational Design Method for Tissue Dissociation Enzyme (RDMTDE) optimization represents a systematic approach to formulating enzyme mixtures based on principles from a hypothetical model for enzyme-mediated cell isolation [50]. This model simplifies optimization of collagenase-protease enzyme mixtures through four key assumptions that provide the theoretical foundation for the RDMTDE framework.
The RDMTDE method operates on the principle that under conditions of excess Collagen Degradation Activity (CDA), neutral protease activity becomes the controlling factor for digestion speed, cell yield, viability, and detection of cell membrane markers used to characterize cell populations [50]. This conceptual framework allows researchers to systematically optimize enzyme formulations rather than relying on empirical testing of random enzyme lots.
Comparative Advantages of Defined Enzyme Formulations
The transition from traditional collagenase products to purified-defined enzymes represents a fundamental shift in tissue dissociation methodology. The table below summarizes the key differences between these approaches:
| Characteristic | Traditional Collagenase | Purified-Defined Collagenase |
|---|---|---|
| Lot Consistency | Minimal: Each lot unique | Maximal: Use of purified enzymes ensures consistency |
| Collagenase Purity | 4-8% (w/w) for crude; 15-25% (w/w) for enriched | >95% (w/w) |
| Need to Pre-Qualify New Lots | Necessary to optimize isolation procedure | No need after initial optimization step performed |
| Replication of Results in Other Labs | Less likely if good lots hard to find | No problem, enzyme composition defined |
| Additional Modification of Enzyme Formulation | Not possible | Always an option |
| Shelf Stability | Variable | 4 years |
Table 1: Comparison between traditional and purified-defined collagenase products based on VitaCyte's documentation [50].
The higher purity of defined enzymes (>95% w/w compared to 4-25% for traditional products) explains why most end-users must pre-qualify new lots of traditional collagenase [50]. The potential for other biochemical components in traditional products to influence cell viability or function necessitates this rigorous qualification process [50].
The RDMTDE Methodology: A Step-by-Step Experimental Framework
Determining the Reference Collagenase Dose
The RDMTDE method begins with estimating the milligrams of collagenase and the neutral protease activity required for the enzyme solution, referred to as the Reference Collagenase Dose (RCD) [50]. If the specific neutral protease activity is unknown, the method recommends assuming that 100% of the Worthington Collagenase Type 1 activity is sufficient for cell isolation as a starting point [50].
A critical step involves preparing several enzyme solutions with decreasing neutral protease activities to assess the effect on functional cell yield and viability [50]. This systematic approach allows researchers to establish a dose-response relationship for protease activity rather than relying on fixed formulations.
Enzyme Formulation Calculations and Adjustments
The methodology utilizes a specialized TDE Excel Calculator to determine the precise amounts of PD Collagenase 800 and Collagenase Gold needed to prepare enzyme solutions with specific neutral protease activities [50]. Researchers input the milligrams of collagenase, milliliters of enzyme solution, and percentage of Worthington Collagenase Type 1 neutral protease activity required to prepare each enzyme solution [50].
The relationship between these components follows a logical progression that can be visualized as follows:
Figure 1: The experimental workflow for implementing the RDMTDE method shows the iterative process of enzyme optimization.
This systematic approach requires researchers to determine if VitaCyte's products provide comparable results to those obtained using their current collagenase enzyme [50]. The methodology acknowledges that slower digestion times may occur as neutral protease activity decreases, consistent with the hypothetical model of enzyme-mediated cell isolation [50]. Researchers must then determine if extending the digestion time provides benefits to the recovered cell population that justify the modified protocol [50].
Practical Implementation and Protocol Development
Experimental Parameters for Assessment
When implementing the RDMTDE framework, researchers should establish clear assessment criteria for evaluating enzyme performance. Based on recent advancements in tissue dissociation technologies, the following parameters should be quantified:
- Dissociation efficacy: Measured as percentage of tissue dissociated or cells recovered per milligram of tissue [4]
- Cell viability: Typically should exceed 90% for most applications [30]
- Processing time: Documented from tissue mincing to single-cell suspension [4]
- Cell surface marker integrity: Particularly important for flow cytometry applications [51]
- Downstream functionality: Assessment of cellular function after isolation [52]
Recent studies have demonstrated the importance of optimizing multiple parameters simultaneously. For example, in murine lung tissue studies, extending Collagenase D digestion from one to two hours improved efficiency but compromised cell viability, while increasing digestion buffer volume from 1mL to 4mL per 100mg of tissue improved both viability and yield [51].
Integration with Mechanical Dissociation Methods
The RDMTDE enzymatic approach is frequently combined with mechanical dissociation methods to enhance efficiency. Recent advancements have documented the effectiveness of combined approaches:
| Technology | Tissue Type | Dissociation Efficacy | Viability | Time |
|---|---|---|---|---|
| Enzymatic + Mechanical | Bovine Liver Tissue | 92% ± 8% | >90% | 15 min |
| Automated Mechanical + Enzymatic | Mouse Lung Tissue | 1-6×10^5 cells | 60%-80% | ~1 h |
| Mixed Modal Microfluidic | Mouse Kidney | ~20,000 cells/mg | ~95% (epithelial) | 1-60 min |
Table 2: Performance metrics of combined enzymatic and mechanical dissociation methods across different tissue types, adapted from recent literature [4].
The integration of enzymatic and mechanical methods demonstrates the importance of a holistic approach to tissue dissociation that considers both biochemical and physical parameters.
Research Reagent Solutions for RDMTDE Implementation
Successful implementation of the RDMTDE method requires access to specific reagent systems and tools. The following table outlines essential materials and their functions within the optimization framework:
| Reagent/Tool | Function in RDMTDE | Application Specifics |
|---|---|---|
| PD Collagenase 800 | Provides collagenase with neutral protease activity | Contains neutral protease activity close to Worthington Collagenase Type 1 [50] |
| Collagenase Gold | Provides purified collagenase with minimal protease | Used in combination with PD Collagenase 800 to adjust neutral protease levels [50] |
| TDE Excel Calculator | Calculates enzyme mixture ratios | Determines amounts of PD Collagenase 800 and Collagenase Gold for target protease activity [50] |
| Clostripain | Supplemental protease for adjustment | Trypsin-like enzyme with complementary activity to BP Protease [50] |
| Collagenase Type I, II, IV | Tissue-specific dissociation | Type I for epithelial, adrenal; Type II for heart, liver; Type IV for islet cells [53] |
| Dispase | Gentle metalloenzyme for tissue dissociation | Cleaves fibronectin and collagen IV without disrupting cell membranes [51] |
| Hyaluronidase | Glycosidase for ECM degradation | Cleaves glycosidic bonds in hyaluronic acid; used in combination with other enzymes [51] |
Table 3: Essential research reagents and tools for implementing the RDMTDE optimization framework.
The RDMTDE method explicitly uses PD Collagenase 800 and Collagenase Gold as core components because PD Collagenase 800 contains neutral protease activity that is close to the activity found in Worthington Collagenase Type 1, while Collagenase Gold contains the same amount of collagenase but without this protease activity [50]. This specific formulation allows precise control over protease levels while maintaining consistent collagenase activity.
Results Interpretation and Troubleshooting
Assessment of Experimental Outcomes
The RDMTDE framework provides clear guidance for interpreting optimization results. If comparable results to current collagenase lots are obtained, researchers have successfully defined the enzyme formulation required for their specific cell type [50]. This knowledge transfer represents a significant advantage over traditional methods where successful formulation remains a "black box."
When results are not equivalent with initial enzyme mixtures, the method recommends supplementing with additional proteases such as Clostripain, which provides complementary enzyme activity compared to BP Protease [50]. Clostripain's trypsin-like enzyme activity cuts at different regions of proteins than BP Protease, potentially enabling more effective tissue dissociation for challenging applications [50].
Comparative Effort Analysis
The investment required to implement the RDMTDE method must be evaluated against current practices:
| Parameter | Traditional Lot Qualification | RDMTDE Optimization |
|---|---|---|
| Effort Required | Equivalent | Equivalent |
| Knowledge Gained | None, no knowledge of enzyme composition | Essential: Enzyme composition defined |
| Lot Pre-Qualification | No change, must prequalify new lots | Once defined, no need to prequalify future lots |
| Ability to Modify Formulation | None | Yes |
| Long-Term Consistency | Variable | 4 years shelf stability |
Table 4: Comparison of effort and outcomes between traditional qualification and RDMTDE optimization approaches [50].
This comparison demonstrates that while the initial effort is equivalent between approaches, the RDMTDE method provides significant long-term benefits through defined enzyme compositions and elimination of lot pre-qualification requirements.
Implications for Enzymatic Cell Dissociation Research
The RDMTDE framework represents a shift toward rational, principles-based experimental design in tissue dissociation that aligns with semi-rational approaches in enzyme engineering, which combine benefits of directed evolution and rational design [54]. By creating defined libraries based on structural and functional knowledge, these approaches efficiently sample mutations likely to affect enzyme function, resulting in remarkable improvements in substrate selectivity and specificity [54].
The principles underlying RDMTDE contribute to addressing fundamental challenges in enzymatic cell dissociation research, including:
- Standardization: Defined enzyme compositions enable replication of results across different laboratories [50]
- Mechanistic Understanding: The model relating CDA and protease activities provides testable hypotheses for optimization
- Customization: The ability to modify formulations supports tissue-specific and application-specific optimization
As the field advances, integration of the RDMTDE framework with emerging technologies such as microfluidic dissociation platforms [4] and non-enzymatic approaches [4] will further enhance capabilities for producing high-quality single-cell suspensions for research and therapeutic applications.
The adoption of defined enzyme systems has demonstrated significant benefits in specific applications such as hepatocyte isolation, where studies show comparable viable yields (56 ± 9.41 million cells/g liver) and viability (78.4% ± 1.1%) to traditional enzymes, but with dramatically lower endotoxin contamination (3.6 EU/mg for collagenase vs. 59-256 EU/mg in traditional products) [52]. This improvement in product quality highlights the tangible benefits of the rational design approach for sensitive cell types and therapeutic applications.
Enzymatic cell dissociation is a critical gateway step in modern life science research, enabling the progression from tissue to single-cell suspensions essential for cell therapy manufacturing, single-cell analysis, and downstream processing [4]. The fundamental principle governing this process is the precise balance between efficient tissue disruption and cellular integrity preservation. Within this framework, three parameters emerge as critically interdependent: enzyme concentration, incubation time, and temperature. Optimization is not a standalone goal but a necessary precondition for reproducible research outcomes, as suboptimal dissociation introduces significant technical artifacts that compromise downstream applications including transcriptional profiling, cell surface marker detection, and viability [4] [44]. This guide establishes the mechanistic relationship between these parameters and provides a structured approach for their systematic optimization within the broader context of robust experimental design.
The Parameter Triad: Mechanisms and Interdependencies
The enzymatic dissociation process relies on proteolytic enzymes to degrade the extracellular matrix (ECM) and cell-cell junctions. The efficacy of this process is governed by the interaction of the following core parameters:
Enzyme Concentration: The concentration of proteolytic enzymes (e.g., collagenase, trypsin, Liberase) directly determines the rate of ECM degradation. Excessive concentration accelerates structural breakdown but risks cell surface protein damage and reduced viability, while insufficient concentration leads to low cell yield and extended processing times [55] [30].
Incubation Time: The duration of enzyme exposure must be sufficient to release cells from the tissue matrix. Prolonged incubation, particularly in combination with high enzyme concentrations, activates cellular stress pathways, compromises membrane integrity, and introduces artifacts in subsequent analyses [4] [44]. Shorter incubation periods are generally preferable but must be balanced with yield requirements.
Temperature: As enzyme activity is inherently temperature-dependent, the dissociation process is typically conducted at 37°C to maximize catalytic efficiency [30]. However, elevated temperature also accelerates enzyme-induced cytotoxicity. Managing this trade-off is essential, with some protocols incorporating a preliminary cold incubation phase to allow enzyme penetration with minimal activity [30].
The following diagram illustrates the logical workflow for balancing these critical parameters to achieve optimal dissociation outcomes.
Quantitative Data: Evidence-Based Parameter Ranges
Empirical data is essential for defining effective parameter ranges. The following tables consolidate quantitative findings from recent research, providing a reference for protocol development.
Table 1: Optimized Enzymatic Dissociation Parameters from Recent Studies
| Tissue Type | Enzyme | Optimal Concentration | Optimal Incubation Time | Temperature | Key Outcome | Source |
|---|---|---|---|---|---|---|
| Bovine Adipose Tissue | Liberase | 0.1% | 3 hours | 37°C | Highest cell yield, low population doubling time [55] | |
| Bovine Adipose Tissue | Collagenase Type I | 0.1% | 6 hours | 37°C | Successful isolation, lower yield than Liberase [55] | |
| Human Adrenal Medullary Tumors | Collagenase IV / MTDK Kit | 2 mg/mL / Per kit | 20 minutes | 37°C | Critical factor for viability was short dissociation time [44] | |
| Human PitNETs | Collagenase IV / MTDK Kit | 2 mg/mL / Per kit | 7-15 minutes | 37°C | Very short incubation sufficient for delicate tissue [44] | |
| General Primary Tissue | Collagenase | 50-200 U/mL | 4-18 hours | 37°C | General guideline for a wide range of tissues [30] | |
| General Primary Tissue | Trypsin | 0.25% | 6-18h (4°C) + 20-30min (37°C) | 4°C → 37°C | Cold penetration phase minimizes initial trypsin activity [30] |
Table 2: Impact of Suboptimal Parameters on Dissociation Outcomes
| Parameter | Effect of Excessive Value | Effect of Insufficient Value |
|---|---|---|
| Enzyme Concentration | Damaged cell surface proteins; reduced cell viability and functionality [4] [44] | Low cell yield; incomplete tissue dissociation; extended processing time [55] |
| Incubation Time | Activation of stress pathways; introduction of transcriptional artifacts; reduced viability [4] [44] | Low cell yield; failure to release specific cell populations [55] |
| Temperature | Increased enzymatic cytotoxicity; accelerated degradation of sensitive epitopes | Greatly reduced enzymatic efficiency; dramatically longer dissociation times [30] |
Detailed Experimental Protocols for Parameter Optimization
Protocol: Systematic Titration of Enzyme and Time Parameters
This protocol, adapted from a study on bovine adipose-derived mesenchymal stromal cells, provides a method for simultaneously evaluating enzyme concentration and incubation time [55].
Step 1: Tissue Preparation
- Obtain fresh subcutaneous adipose tissue and transport in cold storage solution (e.g., MACS Tissue Storage Solution).
- Wash tissue thoroughly with a balanced salt solution (e.g., HBSS) containing antibiotics.
- Mince tissue into uniform 3–4 mm fragments using sterile scissors or a scalpel.
Step 2: Enzymatic Titration Setup
- Prepare four different enzyme solutions: Collagenase Type I (0.04% and 0.1%), Liberase (0.1%), and a combination of Collagenase Type I + Trypsin.
- Distribute minced tissue equally into pre-labeled tubes, each containing one of the enzyme solutions.
- Incubate samples at 37°C with gentle agitation. For each enzyme condition, set up multiple tubes to be terminated at different time points (e.g., 3 h, 6 h, overnight).
Step 3: Termination and Cell Harvesting
- At each designated time point, add a complete medium (containing FBS) to the corresponding tube to neutralize enzyme activity.
- Filter the cell suspension through a sterile 70–100 μm cell strainer to remove undigested tissue and debris.
- Centrifuge the filtrate at 100–400 × g for 5–10 minutes. Discard the supernatant and resuspend the cell pellet in an appropriate washing buffer (e.g., DPBS with 0.1% BSA).
Step 4: Outcome Assessment
- Cell Yield: Count the total number of viable cells using an automated cell counter or hemocytometer. Normalize the yield to the initial tissue mass (e.g., cells per gram of tissue).
- Viability: Determine viability via Trypan Blue exclusion or fluorescent dyes (e.g., propidium iodide).
- Functionality (Optional): For stem/progenitor cells, perform a colony-forming unit (CFU) assay. For other cell types, assess specific functions like attachment efficiency or differentiation potential.
Protocol: Viability-Optimized Dissociation for Delicate Tissues
This protocol, informed by studies on human endocrine tumors, prioritizes cell viability through shortened incubation and careful post-processing [44].
Step 1: Sample Handling and Mincing
- Use a minimal amount of delicate tissue (e.g., 7-10 mg for pituitary tumors).
- Perform all preliminary washing and mincing steps on ice to pre-cool the tissue and reduce metabolic activity.
- Mince tissue as finely as possible using fine scissors or a scalpel to maximize the surface area for enzyme action.
Step 2: Time-Critical Enzymatic Digestion
- Submerge the minced tissue in a pre-warmed dissociation solution containing a defined enzyme (e.g., 2 mg/mL Collagenase IV or a commercial kit like Miltenyi's MTDK).
- Incubate at 37°C for a short, defined period (e.g., 7-20 minutes, depending on tissue consistency).
- Agitate the tube gently every 5 minutes via manual pipetting or gentle rocking—do not use vigorous vortexing.
Step 3: Post-Dissociation Purification
- Immediately after incubation, place the tube on ice and add cold wash buffer.
- Perform debris removal using a density gradient solution or a commercial debris removal kit.
- If the sample is bloody, perform red blood cell lysis using a hypotonic buffer.
- Centrifuge and resuspend the final cell pellet in a cold, protein-containing buffer (e.g., 0.1% BSA-PBS).
The Scientist's Toolkit: Essential Reagents and Solutions
Table 3: Key Research Reagent Solutions for Enzymatic Dissociation
| Item | Function / Application | Example in Context |
|---|---|---|
| Liberase | A purified enzyme blend (Collagenase + Neutral Protease) known for high purity and lot-to-lot consistency. | Optimal for isolating MSCs from bovine adipose tissue, providing high yield and viability [55]. |
| Collagenase (Type I, IV) | Crude or purified preparations that degrade native collagen. Type I is common; Type IV is gentler for delicate cells. | Used for dissociation of various human endocrine tumors; choice of type impacts viability [44]. |
| Trypsin-EDTA | Serine protease effective at dissociating adherent cell monolayers and tissues. EDTA chelates calcium to disrupt cell adhesion. | Often combined with collagenase for dissociating tough tissues or high-density cultures [30]. |
| Dispase | Neutral protease that cleaves fibronectin and collagen IV. Gentle on cell surfaces; suitable for epithelial cell isolation. | Used for detaching cells as intact sheets; can be mixed with collagenase for efficient tissue dissociation [30]. |
| TrypLE Express | A recombinant fungal protease that functions like trypsin. Animal-origin-free; stable at room temperature. | A direct, consistent substitute for trypsin in standard cell detachment protocols [30]. |
| Hanks' Balanced Salt Solution (HBSS) | A balanced salt solution used as a wash and dilution buffer. Can be with or without Ca2+/Mg2+. | Used as a base for creating enzyme working solutions and for washing tissue pieces pre-digestion [44] [30]. |
| Debris Removal Solution | A density-based solution that separates viable, intact cells from dead cells and tissue debris during centrifugation. | Critical post-dissociation step for improving sample purity and downstream analysis quality [44]. |
Fine-tuning enzymatic dissociation is not merely a technical exercise but a fundamental application of biochemical kinetics to cellular systems. The interdependent relationship between enzyme concentration, incubation time, and temperature dictates that optimization must be holistic, not sequential. As research advances toward greater standardization and reproducibility, the principles outlined here—empirical validation, tissue-specific adaptation, and rigorous outcome assessment—will form the foundation of robust cell isolation protocols. Future developments in enzyme engineering, microfluidic dissociation technologies, and non-enzymatic methods will build upon this foundational understanding of parameter control [4]. The ultimate goal remains the consistent production of high-quality single-cell suspensions that faithfully represent the in vivo state, thereby ensuring the reliability of all subsequent scientific discoveries.
Strategies to Minimize Cellular Stress and Preserve Surface Marker Integrity
The process of creating single-cell suspensions from solid tissues is a critical first step in cell therapy manufacturing, single-cell analysis, and downstream drug development pipelines. However, conventional enzymatic dissociation methods introduce significant technical artifacts that compromise data integrity and therapeutic cell quality. This whitepaper details the documented pitfalls of enzymatic digestion and provides evidence-based strategies and protocols to minimize cellular stress and preserve surface marker integrity, ensuring that experimental results and cell-based products truly reflect in-situ biology.
The Critical Bottleneck in Single-Cell Research
Tissue dissociation into single-cell suspensions is the foundational step for single-cell RNA sequencing, flow cytometry, and the manufacturing of cell-based regenerative medicine therapies [4]. The current bottleneck in these fields is the lack of rigorous, standardized systems for the reproducible dissociation of tissues into highly purified cell populations prior to initiating subsequent processes [4]. Traditional methods, which rely on a combination of mechanical mincing and enzymatic digestion using cocktails of collagenase, dispase, trypsin, and other proteases, face considerable challenges. These include variable cell viability and yield, long processing times, and the potential to create artifacts that distort downstream analyses [4]. The integrity of this initial step is paramount, as any introduced bias propagates through all subsequent data and products.
Documented Pitfalls of Enzymatic Dissociation
A growing body of literature systematically characterizes the specific artifacts introduced by enzymatic digestion, which can be broadly categorized into impacts on surface markers and induction of cellular stress.
Surface Marker Degradation and Alteration
Enzymatic digestion can cleave or mask critical cell surface epitopes, leading to inaccurate phenotyping and false-negative results in flow cytometry. The impact is highly dependent on the specific enzymes used.
Table 1: Impact of Enzymatic Digestion on Key Immune Markers
| Marker | Cell Type | Impact of Enzymatic Digestion | Key Enzymes Implicated | Experimental Evidence |
|---|---|---|---|---|
| CD4, CD8, CD69, CD103 | T Cells | Cleavage and reduced detection | Dispase, Collagenase/Dispase cocktails | Flow cytometry showed significant reduction in marker detection [24]. |
| CD19 | B Cells | Reduced median fluorescence intensity (MFI) | Standard enzymatic cocktails | Acoustic dissociation preserved CD19 MFI significantly better than enzymatic methods [56]. |
| CD138 | Plasma Cells | Marked reduction in detection | Proteolytic Enzymes | Acoustic dissociation preserved the highest CD138 signal, indicating enzymatic degradation of proteoglycan-rich epitopes [56]. |
| CD45 | Pan-Leukocyte | Reduced signal intensity | Standard enzymatic cocktails | Enzymatic dissociation caused notable reduction in CD45 MFI, potentially biasing immune cell quantification [56]. |
| B220, CXCR5 | B Cells | Reduced expression levels | Standard enzymatic cocktails | Altered detection of B cell and chemokine receptor markers [56]. |
Furthermore, enzyme selection can skew the apparent cellular composition of a tissue. For example, protocols high in dispase not only cleave immune markers but also contribute to an increased monocyte-to-macrophage yield and a differential epithelial cell profile [24]. Similarly, collagenase P protocols have been shown to yield increased AT1 and AT2 cells and decreased endothelial cells from human lung tissue compared to collagenase D protocols [24].
Induction of Transcriptional and Proteotype Bias
Beyond surface markers, enzymatic dissociation induces profound and consistent alterations in the cellular transcriptome and proteome. A systematic investigation of brain tissue dissociation demonstrated that standard enzymatic digestion at 37°C (ED37°) induces significant transcriptional changes compared to mechanical dissociation at 4°C (MD4°) [57].
- Transcriptional Artifacts: Single-cell RNA-sequencing revealed that ED37° caused significant deregulation of genes in microglia (226 genes), astrocytes (290 genes), and neurons (771 genes) [57]. A hallmark of this enzymatic-induced stress is the increased expression of immediate early genes (IEGs) such as Fos and Jun, which mimic cellular activation across multiple cell types [57]. This also includes deregulated expression of genes encoding for ribosomal and mitochondrial proteins [57].
- Proteotype Artifacts: Mass spectrometry proteotype analysis revealed marked differences in microglia and astrocytes isolated via ED37° versus MD4°. In microglia, 1,619 proteins were significantly different, and in astrocytes, 1,984 proteins were altered following enzymatic digestion, confirming that the artifacts extend beyond the transcriptome to the functional protein level [57].
The following diagram illustrates the cascade of cellular stress and artifacts triggered by suboptimal dissociation conditions.
Strategic Approaches for Improved Dissociation
Optimization of Traditional Enzymatic Methods
For situations where enzymatic digestion remains necessary, protocol optimization can significantly mitigate its negative impacts.
- Enzyme Selection and Cocktail Formulation: Choose enzymes with specificity for the target extracellular matrix while minimizing activity against cell surface proteins. Avoid or limit the use of dispase when T cell markers like CD4, CD8, and CD69 are of interest [24]. Consider using purified collagenase blends instead of crude preparations to reduce non-specific proteolysis.
- Temperature and Time Control: Minimize both incubation time and temperature. Where possible, perform digestions at lower temperatures (e.g., room temperature or 4°C) to reduce cellular metabolic activity and the induction of stress genes [57]. Using cold-active proteases can facilitate this. Establish the minimal time required for effective dissociation to limit exposure [58].
- Inhibition and Monitoring: Have quenching agents (e.g., serum, inhibitors) ready to immediately halt enzymatic activity once dissociation is complete. Implement objective monitoring systems, such as lens-free imaging, to determine the optimal inhibition time-point based on detachment metrics rather than subjective estimation, thus preventing over-digestion [58].
Adoption of Non-Enzymatic and Novel Dissociation Technologies
Emerging technologies offer pathways to circumvent the inherent problems of enzymatic methods altogether.
- Mechanical Dissociation at 4°C (MD4°): An optimized mechanical dissociation protocol performed entirely at cold temperatures has been shown to keep cells in a quiescent state, preserving native transcriptional and proteotype profiles [57]. This method is cost-effective and technically straightforward, providing a robust alternative for sensitive applications like single-cell sequencing and mass spectrometry.
- Acoustic Dissociation: Systems like the Cellsonics SimpleFlow use controlled acoustic energy to dissociate tissues without enzymes or elevated temperature [56]. This method gently agitates tissues to liberate cells while preserving epitope integrity (e.g., CD19, CD45, CD138) and minimizing the induction of artificial stress responses, providing a superior platform for accurate immunophenotyping [56].
- Electrical and Ultrasound Methods: Novel microfluidic and physical dissociation methods are under active development. Electrical dissociation has been shown to achieve high yields (~95%) and viability (~90%) from bovine liver tissue in as little as 5 minutes [4]. Ultrasound dissociation, either alone or in combination with minimal enzymes, also presents a rapid, enzyme-free or enzyme-reduced alternative [4].
Table 2: Comparison of Tissue Dissociation Method Efficacies
| Technology | Dissociation Type | Tissue Type | Cell Viability | Key Advantages | Source |
|---|---|---|---|---|---|
| Optimized Mechanical (MD4°) | Mechanical (4°C) | Mouse Brain | Preserved | Minimal transcriptomic/proteotype bias; cost-effective. | [57] |
| Acoustic Dissociation | Physical (Acoustic) | Murine Spleen, Kidney | High | Excellent surface marker preservation; no stress gene induction. | [56] |
| Electrical Dissociation | Electrical | Bovine Liver, Glioblastoma | 80-90% | Rapid (5 min); high yield. | [4] |
| Ultrasound Dissociation | Ultrasound ± Enzyme | Bovine Liver, Mouse Tissues | >90% | Rapid; can be enzyme-free. | [4] |
| Microfluidic Platform | Microfluidic/Enzymatic | Human Placental, Rat Liver | Not Reported | Integrated, controlled workflow. | [4] |
The following workflow provides a strategic decision-making process for selecting and applying a dissociation method that prioritizes cellular integrity.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Research Reagent Solutions for Cell Dissociation
| Reagent / Material | Function / Purpose | Key Considerations |
|---|---|---|
| Collagenase (P & D) | Degrades collagen in the extracellular matrix. | Collagenase P yields different cell populations (e.g., more AT2 cells) than Collagenase D in lung tissue [24]. |
| Dispase | Neutral protease that cleaves fibronectin and collagen IV. | Known to cleave CD4, CD8, CD69, CD103; use with caution or avoid in immune phenotyping studies [24]. |
| DNase I | Degrades free DNA released by dying cells, preventing cell clumping. | Essential additive to prevent aggregation and loss of yield in challenging digestions. |
| Accutase | Proprietary mixture of proteolytic and collagenolytic enzymes. | Can be used for milder dissociation; effective for adherent cell cultures [28] [58]. |
| Papain | Cysteine protease effective for neural tissues. | Efficient and less destructive for isolating viable neurons compared to other proteases [28]. |
| TrypLE | Recombinant fungal trypsin substitute. | Gentler alternative to animal-derived trypsin for harvesting adherent cells [58]. |
| EDTA | Chelating agent that binds Ca2+, disrupting cell adhesions. | Often used in combination with enzymes to improve efficiency. |
| Neural Tissue Dissociation Kit (Miltenyi) | Standardized pre-packaged enzymatic kit for CNS tissue. | Provides a reliable, qualified method for brain and spinal cord dissociation [28]. |
The choice of tissue dissociation strategy is not merely a technical preliminary but a decisive factor determining the validity of experimental data and the quality of cellular products. Enzymatic methods, while widely used, introduce significant and documented biases including surface marker degradation, transcriptional stress artifacts, and proteotype alterations. Adherence to the principles outlined in this whitepaper—including the rigorous optimization of enzymatic protocols with attention to enzyme selection, temperature, and time, and the adoption of advanced non-enzymatic methods like mechanical dissociation at 4°C and acoustic dissociation—is essential for any research or development program where biological fidelity is paramount. By implementing these strategies, scientists and drug developers can minimize technical artifacts, preserve the true immune and cellular landscape, and ensure that their findings and therapies are built upon a foundation of authentic biology.
Enzymatic cell dissociation is a cornerstone technique for obtaining viable single cells from tissues, a critical first step for downstream applications such as single-cell RNA sequencing, flow cytometry, and drug screening [59] [60]. However, the process itself inherently inflicts stress on cells, potentially compromising cell viability, yield, and the integrity of molecular data. Traditional dissociation protocols using proteases like trypsin and collagenase at elevated temperatures (28–37°C) are known to induce transcriptional cell stress and alter the expression of native genes, particularly those involved in extracellular matrix (ECM) production [61].
Within this context, the strategic incorporation of specific supplements—Bovine Serum Albumin (BSA), DNase, and enzyme inhibitors—is not merely an optional refinement but a fundamental requirement for advanced protocol optimization. These components work synergistically to protect cell integrity, minimize artifactual stress responses, and ensure that the isolated cells truly reflect their in vivo state. This guide frames their use within the broader thesis that successful enzymatic dissociation research demands a holistic understanding of both disruptive enzymes and protective agents to preserve biological fidelity.
Functions of Key Supplements in Dissociation Protocols
The following table summarizes the primary roles and considerations for each key supplement discussed in this guide.
Table 1: Key Supplements for Optimized Cell Dissociation
| Supplement | Primary Function | Mechanism of Action | Key Considerations |
|---|---|---|---|
| Bovine Serum Albumin (BSA) | Protein carrier and inhibitor of contaminant nucleases [62] [63] [64]. | Acts as a non-specific protein carrier; competitively inhibits acid DNases, likely by binding to the enzyme or inducing non-Michaelis-Menten kinetics [63]. | - Use in DPBS for a FACS sorting buffer [59].- Its inhibitory effect is pH-dependent [63]. |
| DNase I | Prevents cell clumping [59] [61] [40]. | Hydrolyzes phosphodiester bonds in free DNA released from damaged cells, breaking down the sticky mesh that traps cells [40]. | - Requires divalent metal ions (Ca²⁺, Mg²⁺/Mn²⁺) for activity [40].- Included in cold protease working solutions [61]. |
| Enzyme Inhibitors (e.g., Soybean Trypsin Inhibitor - STI) | Halts proteolytic activity post-digestion [59]. | Irreversibly inhibits trypsin, preventing over-digestion which promotes cell aggregation and reduces viability [59]. | - A crucial "stop" step to preserve cell surface proteins and viability.- Dissolved in DPBS and stored at 4°C [59]. |
Advanced Optimization Strategies
Counteracting Dissociation-Induced Artifacts
A primary challenge in dissociation is managing the cellular stress response. Studies comparing traditional warm dissociation with cold-active protease methods have demonstrated that high-temperature dissociation specifically downregulates hallmark genes involved in cell specification and ECM production [61]. For example, tenocytes (tendon fibroblasts) dissociated with collagenase at 37°C showed suppressed expression of key tenocyte markers compared to those dissociated with subtilisin A at 4°C [61]. This underscores the importance of not only choosing the right primary enzyme but also implementing strategies to mitigate its side effects.
The use of DNase is a direct response to a key artifact of dissociation: cell clumping. As cells are damaged during the process, they release genomic DNA, which forms a viscous, sticky network that entraps individual cells, drastically reducing yield and viability [40]. By digesting this free DNA, DNase I is indispensable for maintaining a workable single-cell suspension.
The Synergy of Combined Supplementation
The most effective protocols employ these supplements in concert. A cold-active protease protocol for sensitive zebrafish tendons detailed a working solution containing both DNase I to handle released DNA and BSA in a separate DPBS-based solution to act as a carrier and protectant during subsequent cell handling and sorting [61]. Furthermore, the sequential application of a protease (like trypsin) followed by its specific inhibitor (like STI) provides precise temporal control over the dissociation process, ensuring the reaction is halted before it damages the cells of interest [59].
Detailed Experimental Protocols
Protocol: Supplemented Cold-Active Protease Dissociation for Transcriptomic Preservation
This protocol, adapted from a peer-reviewed method for tendon dissociation, is designed to minimize transcriptional artifacts [61].
Objective: To dissociate tissue into a single-cell suspension while preserving native gene expression profiles, ideal for bulk or single-cell RNA sequencing.
Materials and Reagents:
- Biological Material: Tissue of interest (e.g., dissected zebrafish tendons, though the protocol is not species-specific).
- Cold Protease Stock Solution: 100 mg/mL Protease from Bacillus licheniformis (Subtilisin A) in PBS, aliquoted and stored at -80°C.
- DNase Stock Solution: 20 U/μL DNase I in PBS, aliquoted and stored at -20°C.
- Cold Protease Working Solution: Prepare fresh on ice (See Table 2 for formulation).
- DPBS-BSA Solution: 0.01% BSA in DPBS, stored on ice.
- Other Reagents: Ringer's solution, CaCl₂, EDTA.
Table 2: Cold Protease Working Solution Formulation
| Reagent | Final Concentration | Volume for 1 mL |
|---|---|---|
| 100 mg/mL Cold Protease Stock | 10 mg/mL | 100 μL |
| 1 M CaCl₂ | 5 mM | 5 μL |
| 0.5 M EDTA | 0.5 mM | 1 μL |
| 20 U/μL DNase Stock | 100 U/mL | 5 μL |
| 1x DPBS (no Ca²⁺, no Mg²⁺) | To final volume | 889 μL |
Methodology:
- Tissue Preparation: Dissect tissue into small pieces in ice-cold Ringer's solution. Minimize mechanical stress.
- Enzymatic Digestion: Transfer tissue to the cold protease working solution. Incubate on an ice bath (4°C) with gentle agitation for the required duration (e.g., 45-90 minutes, tissue-dependent).
- Reaction Termination: Add a 5-10x volume of ice-cold DPBS-BSA solution. Gently invert to mix. The BSA acts as a carrier and helps neutralize any residual protease activity.
- Cell Collection: Centrifuge the cell suspension at 300-400 x g for 5 minutes at 4°C.
- Washing and Resuspension: Carefully aspirate the supernatant. Gently resuspend the cell pellet in fresh DPBS-BSA solution.
- Filtration and Analysis: Filter the cell suspension through a sterile strainer (e.g., 35 μm) to remove any remaining aggregates. Count cells and assess viability using trypan blue exclusion before proceeding to downstream applications.
Protocol: Standard Trypsin Dissociation with Aggregation Prevention
This protocol highlights the critical steps for preventing cell aggregation during a standard warm dissociation, common for patient-derived organoids [59].
Objective: To dissociate 3D organoids into a viable single-cell suspension for passaging or drug screening.
Materials and Reagents:
- Biological Material: Patient-derived organoids (e.g., head and neck squamous cell carcinoma PDOs).
- Enzymes: 0.05% trypsin.
- Inhibitor: Soybean Trypsin Inhibitor (STI), dissolved in DPBS at a defined concentration and stored at 4°C.
- Other Reagents: Advanced DMEM/F12, Y-27632 (ROCK inhibitor).
Methodology:
- Washing: Wash organoids with DPBS to remove residual culture medium.
- Enzymatic Digestion: Incubate organoids with 0.05% trypsin at 37°C for a defined, optimized period (e.g., 5-15 minutes). Avoid higher trypsin concentrations and extended incubation times, as these promote aggregation [59].
- Mechanical Dissociation: During incubation, gently pipette the organoids up and down every few minutes to aid disaggregation.
- Reaction Termination: Add a pre-chilled volume of STI solution that provides a 1:1 (v/v) ratio or is otherwise calibrated to neutralize the trypsin. Mix immediately.
- Cell Collection: Centrifuge the cell suspension and aspirate the supernatant.
- Resuspension and Analysis: Resuspend the cell pellet in a complete culture medium, often supplemented with a ROCK inhibitor (Y-27632) to further enhance single-cell survival. Count cells and assess viability.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Reagents for an Optimized Dissociation Workflow
| Reagent | Function | Example Application |
|---|---|---|
| Soybean Trypsin Inhibitor (STI) | Halts tryptic digestion to prevent over-digestion and cell aggregation [59]. | Quenching trypsin activity after organoid dissociation. |
| Protease (Subtilisin A) | Cold-active protease for digesting tissue with minimal transcriptional stress [61]. | Dissociating sensitive tissues for RNA-seq. |
| DNase I | Cleaves extracellular DNA to disrupt the viscous network causing cell clumping [61] [40]. | Added to any dissociation mix where cell damage is expected. |
| Bovine Serum Albumin (BSA) | Non-specific protein carrier; stabilizes cells and inhibits certain nucleases [59] [63]. | Component of FACS buffer and cell resuspension buffers. |
| Y-27632 (ROCK Inhibitor) | Enhances survival of dissociated single cells, particularly in stem cells and organoids [59]. | Added to culture media after passaging. |
| EDTA | Chelates divalent cations (Ca²⁺, Mg²⁺), disrupting cell adhesions and acting as a DNase co-factor [40]. | Component of trypsin-EDTA solutions; included in cold-protease buffer. |
Workflow and Pathway Visualization
The following diagram synthesizes the core principles of an optimized dissociation strategy, integrating the key supplements and their functional relationships to achieve high-quality single-cell outputs.
The advanced optimization of enzymatic cell dissociation protocols moves beyond the singular focus on tissue-disrupting enzymes. A deep understanding of the protective and corrective roles of BSA, DNase, and specific enzyme inhibitors is paramount. As the field advances towards applications demanding the highest biological fidelity—such as single-cell omics and personalized drug screening—the principles outlined here provide a framework for developing robust, reliable, and reproducible dissociation methods. The integration of these supplements is not an ancillary step but a core pillar of modern enzymatic cell dissociation research, ensuring that the cells we study are accurate representations of their native state.
Beyond the Basics: Validating Protocols and Comparing Enzymatic with Emerging Technologies
Enzymatic cell dissociation is a foundational step in single-cell RNA sequencing (scRNA-seq) and other single-cell analyses, where the goal is to disaggregate solid tissues into viable, single-cell suspensions while preserving native cellular states. The quality of the starting cell suspension directly dictates the reliability and interpretability of all downstream data. Quality control (QC) metrics thus serve as the essential bridge between tissue processing and biological discovery, ensuring that the isolated cells accurately represent the original tissue's composition and viability. Within the broader thesis on enzymatic dissociation principles, this document establishes the core QC pillars—cell viability, cell yield, and population representation—that researchers must rigorously quantify to validate their dissociation protocol and avoid analytical artefacts.
Failure to implement comprehensive QC can lead to significant and costly experimental failures. Overly aggressive dissociation can induce widespread cell death and stress-related transcriptional responses that compromise data integrity, while insufficient dissociation results in cell clumps that are misread as biologically relevant multiplets or novel cell types [65]. Therefore, a meticulously optimized and QC-driven dissociation protocol is not merely a preparatory step but a critical determinant of experimental success.
Essential QC Metrics and Their Assessment
This section details the three core quality control metrics, providing the methodologies required for their precise quantification.
Cell Viability
Cell viability determines the percentage of live cells in a suspension post-dissociation. This metric is a primary indicator of dissociation-induced stress and protocol harshness [65].
Assessment Protocols:
- Dye Exclusion Methods: These methods rely on membrane integrity. Viable cells with intact membranes exclude certain dyes, while non-viable cells take them up.
- Trypan Blue: A classic azo dye that stains membrane-compromised cells blue. It is simple to use but can also stain debris, which may compromise accurate counting [65].
- Propidium Iodide (PI) / SYTO9: A fluorescent dye combination used with fluorescence microscopy or flow cytometry. PI is membrane-impermeable and stains dead cells' DNA red, while SYTO9 is a green, cell-permeable nucleic acid stain. The ratio of green to red fluorescence provides a quantitative measure of viability [65].
- Acridine Orange (AO): A cell-permeable dye that emits green fluorescence when bound to DNA and orange when bound to RNA, which can help distinguish cell cycle phases and identify viable cells [65] [41].
- Metabolic Assays: These measure a marker activity associated with viable cell metabolism.
- MTT Tetrazolium Reduction Assay: Metabolically active cells convert the yellow MTT substrate into a purple, insoluble formazan product. The amount of formazan, measured by absorbance after solubilization, is proportional to the number of viable cells. This is considered an endpoint assay due to MTT's cytotoxicity [66].
Table 1: Cell Viability Assessment Methods
| Method | Principle | Readout | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Trypan Blue | Membrane integrity | Brightfield microscopy | Low cost, simple protocol | Stains debris, less quantitative |
| PI/SYTO9 | Membrane integrity | Fluorescence microscopy/Flow Cytometry | High accuracy, quantitative | Requires fluorescent equipment |
| Acridine Orange | Nucleic acid binding | Fluorescence microscopy | Can provide cell cycle information | Requires careful interpretation |
| MTT Assay | Metabolic activity | Absorbance (570 nm) | Amenable to higher throughput | Endpoint assay; cytotoxic reagent |
Cell Yield
Cell yield refers to the total number of live cells recovered from a known starting mass of tissue. It is a direct measure of dissociation efficiency and is crucial for planning downstream applications like scRNA-seq, which often require a minimum cell number.
Assessment Protocol:
- Cell Counting: Use an automated cell counter (e.g., Luna Automated Cell Counter) or a hemocytometer [41].
- Viability Staining: Combine counting with a viability dye (e.g., AO/PI) to count only viable cells [41].
- Calculation: The yield is typically reported as the number of viable cells per milligram of starting tissue. For example, an optimized skin dissociation protocol can enable the isolation of a high number of viable cells from small 4mm punch biopsies for scRNA-seq [41].
Population Representation
Population representation assesses whether the dissociated cell suspension reflects the true cellular diversity of the original tissue. This is the most complex metric but is critical for avoiding biased biological conclusions.
Assessment Protocols:
- Microscopic Examination for Cell Clumping: Use brightfield or confocal microscopy to visually inspect the single-cell suspension. Cell clumps indicate incomplete dissociation, which can lead to multiplets in scRNA-seq data that are misinterpreted as novel cell types or hybrid transcriptional profiles [65].
- Downstream Computational Analysis:
- scRNA-seq Data Integration: Tools like single-cell Population Level Integration (scPoli) can integrate multiple samples and are benchmarked using metrics that assess how well batch effects are corrected and biological cell identities are conserved across samples [67].
- Sample-Level Representation: Methods like GloScope represent each sample as a probability distribution of its cells in a low-dimensional space. Comparing these distributions helps quantify and visualize whether sample-level differences (e.g., due to phenotype) are preserved while technical batch effects are minimized [68].
- Multiplet Detection: Software packages like DoubletDecon and GMM-Demux are specifically designed to detect multiplets in scRNA-seq data, which often arise from cell clumps [65].
The Impact of Dissociation Parameters on QC Metrics
Dissociation outcomes are highly sensitive to protocol parameters. A one-size-fits-all approach is ineffective, and conditions must be tailored to the specific tissue type.
Key Findings from Systematic Studies:
Research on human endocrine tumors has demonstrated that optimal dissociation conditions are tissue-specific [44]. The table below summarizes findings on how different parameters affect viability and yield.
Table 2: Optimizing Dissociation Parameters for Different Tissues
| Tissue Type | Optimal Enzyme | Optimal Time | Post-Dissociation Step | Impact on Viability/Yield |
|---|---|---|---|---|
| Adrenal Medullary Tumors | Not Specified | 20 minutes | Not Specified | Critical factor for viability; longer times reduce viability [44] |
| Adrenocortical Tumors | Collagenase IV or MTDK Kit | Not Specified | Debris Removal System (DRS) | Significantly higher cell viability [44] |
| Pituitary Neuroendocrine Tumors (PitNETs) | MTDK Kit or Collagenase IV | 7-15 minutes | Not Specified | Effective for delicate tissues [44] |
| Skin | Dispase II + Collagenase IV + DNase I | ~2 hours (fresh), ~1 hour (cultured) | Filtration (40-70μm strainer) | High yield of highly viable cells; recapitulates known and rare cell populations [41] |
Other Critical Parameters:
- Enzyme Type: Collagenases (I, IV) degrade collagen; Dispase is effective for epithelial cells; DNase I prevents cell clumping by degrading released DNA [41] [44].
- Time: Prolonged enzymatic incubation damages cell integrity, activates stress pathways, and alters transcriptional profiles [44].
- Temperature & Mechanics: Consistent, gentle pipetting and controlled temperatures (typically 37°C) are standard but must be optimized to minimize mechanical and thermal stress.
A Standardized QC Workflow
The following diagram maps the logical sequence of a comprehensive QC workflow, from tissue receipt to sample-level analysis, integrating the metrics and methods described above.
The Scientist's Toolkit: Essential Reagents and Kits
Successful cell dissociation and QC rely on a suite of specialized reagents and tools.
Table 3: Essential Research Reagent Solutions
| Reagent / Kit | Function / Application | Specific Examples |
|---|---|---|
| Proteolytic Enzymes | Digest extracellular matrix (ECM) to release single cells. | Collagenase I, IV [44]; Dispase II [41]; Trypsin [44] |
| Enzyme Cocktail Kits | Pre-optimized mixtures for specific tissue types. | Multi Tissue Dissociation Kit (MTDK); Neural Tissue Dissociation Kit (NTDK) [44] |
| Nucleases | Prevent cell clumping by degrading free DNA from lysed cells. | DNase I [41] [44] |
| Viability Stains | Distinguish live and dead cells for counting and sorting. | Propidium Iodide (PI); Acridine Orange (AO); SYTO9; Trypan Blue [65] [41] |
| Cell Separation Reagents | Isolate specific cell types from a heterogeneous mixture. | Magnetic-activated cell sorting (MACS) reagents; Antibodies for FACS [69] |
| Debris Removal Kits | Purify suspension by removing dead cells and ECM debris. | Density-based separation systems [44] |
Rigorous quality control is the cornerstone of any robust enzymatic cell dissociation protocol. By systematically quantifying cell viability, yield, and population representation, researchers can move beyond qualitative assessments and establish a quantitative framework for protocol optimization and validation. The integration of traditional bench techniques (dye exclusion, counting) with modern computational biology tools (scPoli, GloScope) provides a multi-scale view of dissociation quality, from individual cell membranes to global sample phenotypes. Adherence to the metrics and methodologies outlined in this guide ensures that the foundational step of single-cell isolation supports, rather than compromises, the integrity of subsequent scientific discovery.
Tissue dissociation into single-cell suspensions is a critical foundational technique in biomedical research and therapeutic development, serving as the essential first step for downstream applications such as single-cell analysis, cell therapy manufacturing, and drug screening [4]. The process involves breaking down the extracellular matrix and cell-cell junctions that hold tissues together, enabling researchers to isolate individual cells for further study [70]. Within the broader context of enzymatic cell dissociation research principles, understanding the comparative advantages and limitations of different dissociation approaches becomes paramount for experimental success.
The current cell dissociation landscape encompasses three primary methodologies: enzymatic, mechanical, and chemical dissociation, each with distinct mechanisms and applications [70]. enzymatic dissociation employs proteolytic enzymes to digest tissue components, mechanical dissociation utilizes physical forces to disaggregate tissues, and chemical dissociation relies on chelating agents to disrupt intercellular bonds [70]. The selection of an appropriate dissociation method directly impacts cell viability, yield, phenotype preservation, and ultimately, the validity of experimental outcomes across various research and clinical applications.
This technical analysis provides a comprehensive comparison of these three fundamental dissociation approaches, examining their underlying mechanisms, performance parameters, and suitability for specific research contexts. By synthesizing current technical specifications and emerging methodologies, this review aims to equip researchers with the evidence-based insights necessary to optimize tissue dissociation protocols within their specific experimental frameworks.
Methodological Fundamentals and Comparative Analysis
Core Mechanisms and Technical Specifications
The three primary dissociation methods operate through distinct biochemical and physical mechanisms to disaggregate tissues into single-cell suspensions. enzymatic dissociation utilizes proteolytic enzymes including collagenase, trypsin, papain, hyaluronidase, and elastase to selectively digest extracellular matrix proteins and cell adhesion molecules [4] [70]. This targeted enzymatic action breaks down the structural integrity of tissues, releasing individual cells while generally preserving membrane integrity. The specificity of enzymatic cleavage can be both an advantage and limitation, as different enzyme combinations must be optimized for particular tissue types based on their extracellular matrix composition.
Mechanical dissociation employs physical forces through methods such as cutting, crushing, grinding, or agitation to physically disrupt tissue architecture [70]. Unlike enzymatic approaches, mechanical techniques apply non-selective force to tear apart cell-cell and cell-matrix connections. While this avoids introducing foreign biochemical agents, the uncontrolled nature of these forces can compromise cell membrane integrity, particularly for delicate cell types. Mechanical methods often utilize simple tools like mortar and pestle or more sophisticated automated systems with rotor-stator mechanisms to apply controlled shear forces [8].
Chemical dissociation employs chelating agents such as ethylene diamine tetra-acetic acid (EDTA) or egtazic acid (EGTA) to bind cations (particularly calcium) that are essential for cell adhesion molecules like cadherins [4] [70]. By sequestering these ions, chemical dissociation disrupts intercellular connections without enzymatic proteolysis or significant mechanical force. This approach is generally considered gentler on cell surface markers but may require extended processing times for effective tissue disaggregation [70].
Table 1: Comprehensive Comparison of Cell Dissociation Methods
| Parameter | Enzymatic Dissociation | Mechanical Dissociation | Chemical Dissociation |
|---|---|---|---|
| Mechanism of Action | Proteolytic digestion of ECM and cell junctions | Physical disruption via shear forces | Chelation of divalent cations essential for cell adhesion |
| Primary Reagents/ Tools | Collagenase, trypsin, dispase, hyaluronidase [4] | Mortar and pestle, automated grinders, microfluidic devices [70] [8] | EDTA, EGTA [4] [70] |
| Processing Time | Moderate to long (30 minutes to several hours) [4] | Fast (minutes to 1 hour) [70] | Long (often several hours) [70] |
| Cell Viability | Variable (can damage sensitive cells) [4] | Lower for fragile cells [70] | Generally high [70] |
| Cell Yield | High for most tissues [70] | Variable between users and tissue types [70] | High viability but potentially lower yield [70] |
| Impact on Cell Surface Markers | Can alter or destroy surface epitopes [4] | Minimal effect on epitopes | Preserves surface proteins well [70] |
| Tissue Specificity | High (enzyme selection must match tissue type) | Works better for loosely associated tissues [70] | Suitable for gentle dissociation needs [70] |
| Reproducibility | Moderate to high with optimization | Low to moderate (operator-dependent) [70] | Moderate (affected by concentration, environment) [70] |
| Cost Considerations | Moderate to high (enzyme costs) | Low (simple tools) to high (automated systems) | Low to moderate |
| Downstream Applications | Cell culture, flow cytometry, single-cell sequencing [4] | Applications where surface marker integrity is critical | Sensitive cell types, embryonic cells [70] |
| Key Limitations | Potential cell damage, time consumption, cost [4] | Inconsistent results, lower viability for delicate cells [70] | Slow process, environmental sensitivity [70] |
Performance Metrics and Efficacy Assessment
The efficacy of dissociation methods is quantitatively assessed through multiple parameters including cell viability, yield, processing time, and preservation of cellular characteristics. Recent comparative data illustrates the performance disparities between methods across different tissue types. enzymatic dissociation typically achieves viability rates of 70%-90% depending on tissue type and protocol optimization, with yields ranging from 2.4×10^6 viable cells from triple-negative human breast cancer tissue (83.5% viability) to approximately 24,000 cells from 4mm human skin biopsy punches (92.75% viability) [4].
Mechanical dissociation demonstrates more variable performance, with viability rates spanning 50%-90% depending on tissue robustness and operator skill [70]. Automated mechanical systems have improved consistency, producing yields of 1×10^5 to 6×10^5 cells from mouse lung tissue with 60%-80% viability [4]. The method remains particularly suitable for loosely associated tissues like spleen, lymph nodes, and bone marrow where minimal mechanical force enables sufficient disaggregation [70].
Chemical dissociation typically delivers high viability rates (often exceeding 85%) due to its gentle mechanism of action, though complete dissociation may require extended processing times [70]. The approach demonstrates particular value for sensitive applications involving embryonic cells or primary cell cultures where surface marker preservation is critical [70].
Table 2: Advanced Dissociation Technologies and Performance Metrics
| Technology | Dissociation Type | Tissue Type | Cell Yield | Viability | Time |
|---|---|---|---|---|---|
| Optimized Chemical-Mechanical Workflow [4] | Enzymatic/Mechanical | Bovine Liver Tissue | 92% ± 8% (with both methods) | >90% | 15 min |
| Automated Mechanical Device [4] | Mechanical | Mouse Lung/Kidney/Heart | 1×10^5 to 1.5×10^6 cells (depending on tissue) | 50%-80% (depending on tissue) | ~1 h |
| Mixed Modal Microfluidic Platform [4] | Microfluidic/Mechanical/Enzymatic | Mouse Kidney/Breast Tumor/Liver/Heart | 900-20,000 cells/mg tissue (varies by cell type) | 50%-95% (depending on cell type) | 1-60 min |
| Electric Field Dissociation [4] | Electrical | Bovine Liver/GBM | 95% ± 4% (bovine liver); >5× higher than traditional (GBM) | 80%-90% | 5 min |
| Ultrasound Sonication [4] | Ultrasound/Enzymatic | Bovine Liver | 53% ± 8% (sonication alone); 72% ± 10% (with enzyme) | 91%-98% | 30 min |
| Hypersonic Levitation and Spinning (HLS) [8] | Acoustic/Mechanical | Human Renal Cancer | 90% tissue utilization | 92.3% | 15 min |
Technological Innovations and Emerging Methodologies
Advanced Dissociation Platforms
Recent technological innovations have significantly expanded the toolbox available for tissue dissociation, addressing limitations of conventional methods. Microfluidic platforms represent a promising advancement, enabling precise control over mechanical forces while reducing reagent consumption and processing times [4]. These systems facilitate both enzymatic and non-enzymatic dissociation through constricted channels that apply controlled shear stress to tissue fragments. Mixed modal microfluidic platforms have demonstrated efficacy across multiple tissue types, achieving epithelial cell yields of approximately 20,000 cells/mg from mouse kidney tissue with 95% viability, and 9,000 cells/mg from mouse breast tumor tissue with 70%-80% viability [4]. However, these systems face challenges with channel clogging and limited processing capacity for larger tissue samples [8].
Acoustic-based technologies represent another innovative approach, with Hypersonic Levitation and Spinning (HLS) demonstrating particularly promising results. This contact-free method utilizes a triple-acoustic resonator probe to levitate and spin tissue samples within a confined flow field, generating microscale "liquid jets" that exert precise hydrodynamic forces [8]. The technology achieves 90% tissue utilization in just 15 minutes while maintaining 92.3% cell viability and better preserving rare cell populations compared to conventional methods [8]. The non-contact nature of HLS minimizes mechanical damage while enhancing enzyme penetration when used in combination approaches.
Electrical dissociation methods have also emerged as rapid alternatives, using electric fields to disrupt tissue integrity without enzymatic or significant mechanical stress. This approach has demonstrated impressive efficiency, achieving 95% ± 4% dissociation of bovine liver tissue in just 5 minutes with 90% ± 8% viability [4]. Similarly, ultrasound-assisted dissociation techniques combine high-frequency sonication with minimal enzymatic treatment, achieving 72% ± 10% dissociation efficiency with bovine liver tissue while maintaining 91%-98% viability [4].
Market Trends and Implementation Landscape
The cell dissociation market reflects these technological shifts, with enzymatic dissociation currently dominating at 47.9% market share due to its established efficacy and broad applicability [71]. However, non-enzymatic dissociation represents the fastest-growing segment as researchers seek to avoid enzyme-induced alterations to cell surfaces [72]. The global cell dissociation market size reached USD 620 million in 2024 and is projected to grow at a compound annual growth rate (CAGR) of 13.63% from 2025 to 2034, reaching approximately USD 2,224 million [72].
North America constitutes the largest regional market (over 39% revenue share in 2024), driven by substantial research infrastructure, presence of key market players, and significant investments in cell-based therapies [72]. The Asia-Pacific region represents the fastest-growing market, fueled by expanding healthcare infrastructure, increasing biopharmaceutical investment, and growing research capacity [72].
The pharmaceutical and biotechnology sector constitutes the dominant end-user segment (71.60% share), with accelerating adoption driven by escalating investment in cell-based therapies and drug discovery pipelines [71]. Academic and research institutes represent the fastest-growing end-user segment as fundamental research into cellular mechanisms expands globally [72].
Practical Implementation and Workflow Integration
Method Selection Framework and Experimental Design
Optimal dissociation method selection requires systematic consideration of multiple experimental parameters including tissue characteristics, target cell population, and downstream applications. The following decision framework provides guidance for method selection based on primary experimental requirements:
Diagram 1: Dissociation Method Selection
For tissues with robust cellular architecture and time-sensitive applications, combined enzymatic-mechanical approaches frequently optimize both yield and processing efficiency. Recent research demonstrates that optimized workflows integrating both modalities achieve 92% ± 8% dissociation efficiency for bovine liver tissue in just 15 minutes with viability exceeding 90% [4]. Such integrated approaches leverage enzymatic digestion to weaken extracellular matrix integrity followed by gentle mechanical agitation to complete dissociation, balancing efficiency with cell preservation.
Research Reagent Solutions and Essential Materials
Successful implementation of dissociation protocols requires appropriate selection of reagents and equipment tailored to specific experimental needs. The following table catalogues essential research solutions across the three methodological approaches:
Table 3: Essential Research Reagent Solutions for Cell Dissociation
| Reagent/Tool | Function | Application Context |
|---|---|---|
| Collagenase [4] | Digests collagen in extracellular matrix | Enzymatic dissociation of connective tissues |
| Trypsin [4] [70] | Proteolytic enzyme targeting cell adhesion proteins | Enzymatic dissociation for cell culture |
| Hyaluronidase [4] | Degrades hyaluronic acid in extracellular matrix | Enzymatic dissociation, often used in combination |
| Dispase [4] | Protease that cleaves fibronectin and collagen | Gentle enzymatic dissociation preserving surface markers |
| Papain [4] | Cysteine protease for tissue dissociation | Specialized enzymatic protocols |
| EDTA/EGTA [4] [70] | Chelating agents that bind calcium ions | Chemical dissociation preserving surface proteins |
| Automated Mechanical Dissociator [4] | Standardized mechanical disruption | Reproducible mechanical dissociation |
| Microfluidic Dissociation Chips [4] | Controlled shear stress in microchannels | Gentle, efficient dissociation for small samples |
| Acoustic Levitation Devices [8] | Contactless dissociation via hypersonic waves | Preservation of rare and fragile cells |
| Enzyme-Free Dissociation Buffers | Chemically defined non-enzymatic solutions | Surface marker-intensive applications |
Integrated Workflow for Optimal Cell Harvesting
Implementing a structured dissociation workflow ensures consistent results and maximizes cell viability and function. The following workflow diagram illustrates a comprehensive approach integrating method selection with subsequent processing steps:
Diagram 2: Integrated Dissociation Workflow
This integrated workflow emphasizes quality assessment points that enable researchers to troubleshoot suboptimal dissociation before proceeding to resource-intensive downstream applications. Critical assessment parameters include viability thresholds (typically >80% for most applications), yield adequacy for planned experiments, and functionality validation through membrane integrity tests or surface marker expression analysis.
The comparative analysis of enzymatic, mechanical, and chemical dissociation methods reveals a complex landscape with no universally superior approach. Each methodology demonstrates distinctive strengths and limitations that must be aligned with specific research objectives, tissue characteristics, and downstream applications. enzymatic dissociation remains the workhorse for most applications due to its efficacy across diverse tissue types, while mechanical approaches offer rapid processing for robust tissues, and chemical methods provide gentle action for sensitive cells.
Emerging technologies including microfluidic systems, acoustic levitation, and electrical dissociation present promising avenues for overcoming limitations of conventional methods. These advanced platforms offer enhanced precision, reduced processing times, and improved preservation of delicate cell populations, potentially enabling previously challenging experimental approaches. The escalating integration of automation and artificial intelligence in dissociation workflows further promises enhanced reproducibility and optimization through real-time parameter adjustment [73] [71].
Future methodological development will likely focus on several key areas: First, increasing standardization to reduce protocol variability between laboratories and improve experimental reproducibility. Second, enhancing compatibility with complex downstream applications including single-cell multi-omics and functional assays requiring maximal cell viability and functional preservation. Third, developing integrated systems that seamlessly transition from tissue dissociation to subsequent processing steps, minimizing manual handling and potential sample degradation.
As the field advances, the optimal dissociation strategy will continue to balance multiple competing parameters: efficiency versus preservation, specificity versus universality, and technical simplicity versus experimental sophistication. By thoughtfully applying the principles outlined in this analysis, researchers can make evidence-based decisions that maximize experimental outcomes while acknowledging the inherent methodological tradeoffs in tissue dissociation.
The dissociation of tissue into viable single-cell suspensions represents a critical foundational step in modern biological research, particularly for single-cell sequencing, drug discovery, and precision medicine applications. Traditional dissociation methods, primarily utilizing enzymatic or mechanical approaches, present significant limitations including reduced cell viability, loss of rare cell populations, and alteration of cell surface markers [74] [8] [25]. Within the context of enzymatic dissociation research, these limitations have driven the development of novel technologies that seek to minimize cellular damage while improving efficiency and yield. The inherent heterogeneity of individual cells within populations plays a significant role in disease development and progression, making the gentle and accurate dissociation of tissues into representative single-cell suspensions crucial for meaningful experimental outcomes [74]. Emerging technologies—particularly microfluidics, acoustic levitation, and automated systems—address these challenges through innovative physical principles that minimize direct contact with cells while enabling precise control over the dissociation environment. This technical guide provides an in-depth evaluation of these emerging dissociation technologies, their operational principles, experimental protocols, and performance metrics relative to traditional methods, framed within the ongoing evolution of enzymatic dissociation research.
Microfluidics-Based Dissociation
Microfluidics, often described as "lab-on-a-chip" technology, provides a novel tool for single-cell analysis in an efficient manner by precisely controlling fluids in microscale channels typically ranging from 10 to 100 micrometers wide [74]. This technology enables the miniaturization of dissociation processes, working with picoliter to nanoliter volumes of solution, which reduces sample loss and increases sensitivity while making high-throughput single-cell analysis possible [74]. Microfluidic platforms for single-cell isolation include microwells, dielectrophoresis, droplets, paper-based microfluidics, valve-based systems, and hydrodynamic devices [75]. These systems facilitate the gentle separation of viable single cells while preserving original phenotypic characteristics, in contrast to conventional enzymatic dissociation that often degrades cell surface proteins and affects viability [75]. The most advanced microfluidic isolation methods utilize microdroplets to encapsulate single cells in an inert carrier oil, creating an enclosed space that reduces the risk of sample contamination while enabling high-throughput processing of thousands of cells in very short timeframes [74].
Acoustic Levitation Technologies
Acoustic levitation represents a contact-free approach to tissue manipulation and dissociation that leverages precisely controlled sound waves to generate hydrodynamic forces. The Hypersonic Levitation and Spinning (HLS) method, a recent innovation, utilizes a triple-acoustic resonator probe that enables target tissue samples to levitate and execute a 'press-and-rotate' operation within a confined flow field [8]. This mechanism generates microscale 'liquid jets' that exert precise hydrodynamic forces in a non-contact manner, enhancing shear forces on tissue surfaces to facilitate rapid and efficient dissociation while safeguarding cell integrity [8]. The underlying principle involves hypersonic streaming phenomena, where high-frequency acoustic waves (at GHz frequencies) interact with fluid molecules to generate a steady flow. At these frequencies, fluid particles move with high velocities due to short decay length, allowing for precise microscale manipulation of the fluid flow [8]. The acoustic radiation forces employed in these systems are particularly suited for label-free manipulation of living cells, organoids, and tissues, enabling their transportation and organization into deterministic complex patterns within seconds [76].
Automated Mechanical Dissociation Systems
Automated mechanical systems standardize the dissociation process through controlled mechanical forces, reducing operator-dependent variability. The Medimachine II system represents one such approach, employing a disposable Medicon capsule containing a steel mesh with approximately 100 hexagonal holes framed by six microblades [25]. The instrument spins tissue fragments within this cartridge at a constant speed (typically 100 rpm) for time periods ranging from 15 to 55 seconds, resulting in efficient cutting of tissues into single-cell suspensions [25]. These systems are designed to minimize the bias arising from operator skill and provide a simple, fast, standardized method for tissue processing. Another approach involves automated tissue grinders that employ a motorized rotor and stator insert with microstructures to compress and break down tissue [8]. However, these systems face challenges including potential cell damage from rigid contact between components and uneven force distribution due to inability to adapt to varying tissue shapes and sizes [8].
Table 1: Comparison of Emerging Dissociation Technology Principles
| Technology | Operating Principle | Force Mechanism | Throughput | Key Advantages |
|---|---|---|---|---|
| Microfluidics | Precise fluid control in microchannels | Hydrodynamic forces, droplet encapsulation | High (1000-10,000 cells/run) [74] | Low sample volume, high sensitivity, integration capability [74] |
| Acoustic Levitation | Hypersonic streaming with resonator probes | Non-contact hydrodynamic shear forces | Moderate (varies with system size) | Maintains cell integrity, preserves rare populations [8] |
| Automated Mechanical | Rotating blades or meshes with controlled motion | Mechanical cutting and shearing | High (>1000 cells/run) [74] | Standardization, minimal operator dependency [25] |
Experimental Protocols and Methodologies
Microfluidics Workflow for Single-Cell Analysis
The application of microfluidics for single-cell sequencing involves a multi-step process that begins with tissue dissociation and progresses through sequencing preparation. The complete workflow includes: (1) single cell separation, (2) single cell lysis, (3) nucleic acid amplification, (4) high-throughput sequencing, (5) data processing, and (6) data analysis [74]. For the dissociation phase specifically, microfluidic devices utilize various principles including hydrodynamic cell traps, pneumatic membrane valves, and droplet-in-oil-based isolation [74]. The droplet-based method, particularly prominent in high-throughput applications, involves:
- Preparing a cell suspension from minimally processed tissue
- Loading the suspension into a microfluidic device designed for droplet generation
- Combining the aqueous cell suspension with an inert carrier oil to form monodisperse droplets
- Encapsulating individual cells within separate droplets at rates of thousands per second
- Collecting the droplets for downstream processing or analysis
This approach creates an enclosed microenvironment for each cell, preventing cross-contamination and maintaining cellular integrity [74]. The flexibility in microfluidic channel design enables customization according to specific tissue types and experimental requirements, with different chip architectures optimized for various cellular dimensions and characteristics [75].
Hypersonic Levitation and Spinning Protocol
The HLS methodology employs an automated tissue dissociation apparatus that integrates dissociation, fluid replacement, filtration, and output functions in a single system [8]. The specific experimental protocol involves:
- Apparatus Setup: The system features four vertical tubes (inlet, sample, outlet, and waste) and two chambers (digestion and single-cell collection) [8].
- Sample Loading: Tissue samples are placed within the dissociation chamber containing appropriate digestion media.
- Acoustic Activation: The triple-acoustic resonator probe is activated, generating localized acoustic fields that cause the tissue sample to levitate and execute a 'press-and-rotate' operation.
- Dissociation Monitoring: The process continues for a predetermined duration (approximately 15 minutes for human renal cancer tissue), during which the hypersonic streaming generates microscale liquid jets that apply hydrodynamic shear forces to dissociate the tissue.
- Cell Collection: Dissociated cells are transported to the collection chamber while the original tissue remnant remains levitated, enabling separation of intact cells from tissue debris.
- Fluid Exchange: The integrated system automatically performs fluid replacement to remove enzymes or debris and filters the resulting single-cell suspension.
This contactless approach enhances chemical dissociation processes by enabling enzyme solutions to permeate deeper into tissue layers while applying precisely controlled mechanical forces [8].
Automated Mechanical Dissociation Procedure
The automated mechanical dissociation protocol using systems like the Medimachine II follows a standardized approach:
- Tissue Preparation: Fresh tissues are dissected into Petri dishes containing cold phosphate-buffered saline (PBS) and cut into approximately 1 mm³ pieces using a scalpel [25].
- Device Assembly: Disposable Medicon capsules are filled with 1 mL of appropriate medium (such as RPMI 1640) before adding the minced tissue pieces.
- Mechanical Processing: The filled Medicons are inserted into the Medimachine, and the instrument is run for a tissue-dependent duration ranging from 15 to 55 seconds at a constant speed of 100 rpm.
- Cell Collection: The cell suspension is aspirated with a syringe from the bottom part of the Medicon capsule.
- Optional Repetition: After the first run, additional medium can be added to the processed tissue remnant, and the dissociation repeated with cell fractions subsequently pooled to increase yield.
This automated–mechanical procedure provides a standardized, operator-independent method that minimizes variability while maintaining cell function for downstream applications [25].
Figure 1: Experimental Workflow for Emerging Dissociation Technologies
Performance Comparison and Quantitative Analysis
Cell Viability and Yield Metrics
Comprehensive evaluation of dissociation technologies requires assessment of multiple performance parameters, with cell viability and yield representing fundamental metrics. Research comparing automated mechanical and enzymatic dissociation has demonstrated that mechanical procedures can preserve specific cell functions, with better maintenance of lysosome and mitochondria labeling compared to enzymatic approaches [25]. Conversely, enzymatic dissociation appears to induce lower amounts of intracellular reactive oxygen species (ROS) in some tissue types [25]. In studies focused on adipose-derived stromal vascular fraction (SVF) isolation, both enzymatic and mechanical procedures yielded comparable results in terms of cell viability (70%-99% for enzymatic vs. 46%-97.5% for mechanical) and cell yield (2.3-18.0 × 10⁵ cells/mL for enzymatic vs. 0.03-26.7 × 10⁵ cells/mL for mechanical) [77]. The Hypersonic Levitation and Spinning technology demonstrates exceptional performance, achieving 92.3% cell viability with 90% tissue utilization in just 15 minutes, compared to approximately 70% utilization in 60 minutes with traditional methods [8]. This represents a significant advancement in both efficiency and preservation of cellular integrity.
Preservation of Rare Cell Populations and Surface Markers
A critical advantage of emerging dissociation technologies is their enhanced ability to preserve rare cell populations and maintain native cell surface marker expression. The gentle, non-contact nature of acoustic levitation specifically excels in preserving rare cell populations, including fragile cell types that would be lost or damaged using conventional methods [8]. This has profound implications for cancer research where rare tumor cells drive metastasis or exhibit drug resistance. Surface marker preservation varies significantly across dissociation methods. Studies investigating cell detachment methods have demonstrated that accutase, often considered a mild enzymatic approach, can compromise certain surface proteins including Fas ligands and Fas receptors, requiring up to 20 hours for recovery after treatment [78]. EDTA-based nonenzymatic cell dissociation buffers better preserve these markers, while mechanical detachment methods (such as scraping) maintain the highest surface levels of sensitive markers like FasL [78]. Microfluidic approaches offer advantages in surface marker preservation by minimizing enzymatic exposure and reducing mechanical shear compared to traditional methods [75].
Table 2: Quantitative Performance Comparison of Dissociation Methods
| Method | Cell Viability (%) | Processing Time | Tissue Utilization | Rare Cell Preservation | Surface Marker Integrity |
|---|---|---|---|---|---|
| Hypersonic Levitation | 92.3% [8] | 15 minutes [8] | 90% [8] | Excellent [8] | High (non-contact) |
| Microfluidics | >90% (varies by design) [74] | Seconds to minutes (high throughput) [74] | Varies by tissue type | Good (gentle separation) [75] | High (minimized enzyme use) [75] |
| Automated Mechanical | 46%-97.5% [77] | 15-55 seconds [25] | Moderate to high | Moderate | Variable (mechanically induced) |
| Traditional Enzymatic | 70%-99% [77] | 50-210 minutes [77] | ~70% in 60 minutes [8] | Poor to moderate | Low (enzyme degradation) [78] |
| Traditional Mechanical | Variable, often lower | Minutes to hours | Moderate | Poor | Moderate |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of emerging dissociation technologies requires specific reagents and materials optimized for each platform. The following table details key solutions and their functions:
Table 3: Essential Research Reagent Solutions for Tissue Dissociation
| Reagent/Material | Composition/Characteristics | Primary Function | Technology Compatibility |
|---|---|---|---|
| Accutase | Proteolytic and collagenolytic enzymes in PBS | Gentle enzymatic dissociation while maintaining cell surface proteins [78] | Microfluidics, general cell culture |
| EDTA-Based Dissociation Buffer | Enzyme-free, isotonic, aqueous formulation of salts and chelating agents | Chelates calcium and magnesium ions to disrupt cell adhesion [78] | All technologies (gentle alternative) |
| Collagenase Preparations | Varying purity levels (including recombinant forms) | Digestion of collagen in extracellular matrix [79] | Traditional and emerging enzymatic methods |
| PEGDA-Gelatine Hydrogel | Polyethylene glycol diacrylate-gelatine composite | Sample embedding and spatial organization during acoustic manipulation [76] | Acoustic levitation platforms |
| Carrier Oils (HFE-7500, etc.) | Fluorinated, inert, biocompatible oils | Forms immiscible phase for droplet generation [74] | Microfluidic droplet systems |
| Medicon Capsules | Disposable dissociators with steel mesh (100 hexagonal holes) | Tissue containment and mechanical disaggregation [25] | Automated mechanical systems |
Integration with Downstream Applications
The ultimate value of dissociation technologies lies in their compatibility with downstream analytical applications, particularly single-cell RNA sequencing, flow cytometry, and advanced cell culture models. Microfluidics has established particularly strong integration with single-cell sequencing workflows, with various microfluidic technologies emerging as frontiers in single-cell research over the past decade [74]. The ability to efficiently process thousands of cells in short timeframes makes these systems ideal for preparing representative single-cell suspensions for genomic analysis. Acoustic levitation technologies demonstrate exceptional compatibility with primary cell culture and single-cell sequencing, as evidenced by comprehensive experiments on human renal cancer tissue that included flow cytometry, primary cell culture, immunofluorescence, and single-cell RNA sequencing [8]. Automated mechanical systems have shown excellent performance in maintaining cellular function for flow cytometric analysis and confocal microscopy, providing high-quality single-cell suspensions while preserving organelles and metabolic functions [25]. When selecting dissociation technologies for specific downstream applications, researchers must consider the sensitivity of target cells to mechanical or enzymatic stress, the importance of rare cell populations, and the required throughput for the intended experimental workflow.
Figure 2: Technology Performance Comparison Across Key Parameters
The evolution of tissue dissociation technologies from traditional enzymatic and mechanical approaches toward microfluidics, acoustic levitation, and automated systems represents significant progress in single-cell research capabilities. While enzymatic dissociation remains valuable for specific applications, particularly with ongoing improvements in enzyme purity and specificity [79], emerging technologies address fundamental limitations including cell viability, rare population preservation, and surface marker integrity. Microfluidic platforms offer exceptional integration potential with downstream analytical methods, acoustic levitation provides unparalleled gentle manipulation through non-contact mechanisms, and automated systems deliver standardization and reproducibility. The optimal selection of dissociation technology depends on specific research requirements, tissue characteristics, and intended applications, with each approach offering distinct advantages. Future developments will likely focus on combining the strengths of these technologies, further minimizing cellular stress, and enhancing integration with analytical platforms to provide comprehensive solutions for single-cell analysis in research and clinical applications.
Tissue dissociation into single-cell suspensions represents a critical initial step for single-cell analysis, cell therapy manufacturing, and downstream processing in modern biological research and clinical applications [4]. Within the field of renal cell carcinoma (RCC) research, effective dissociation protocols are indispensable for unlocking the pronounced molecular heterogeneity of these tumors and their microenvironment [80] [81]. The process entails breaking down the extracellular matrix (ECM) and cell–cell junctions that hold tissue together, traditionally accomplished via enzymatic and mechanical methods [4]. However, conventional approaches face significant challenges regarding cell viability, yield, processing time, and the potential for creating artifacts that distort downstream analyses [4] [8]. This case study examines the performance of various dissociation methodologies applied to human renal cancer tissues and primary kidney tissues, framing the comparison within the core principles of enzymatic cell dissociation research. We present quantitative data, detailed protocols, and analytical workflows to guide researchers in selecting and optimizing dissociation strategies for RCC studies, with particular emphasis on preserving cellular integrity, minimizing transcriptional alterations, and maintaining the representation of rare cell populations.
Principles of Enzymatic Dissociation in RCC Research
Enzymatic dissociation functions by hydrolyzing specific components of the extracellular matrix that maintain tissue integrity. The selection of enzymes must be tailored to the unique molecular composition of renal tissues and RCC tumors. The dominant ECM proteins in kidney tissue include collagens (particularly types I and IV), fibronectin, and hyaluronic acid [82]. Consequently, enzymatic protocols for renal tissue commonly utilize collagenases to hydrolyze collagens, dispases to cleave fibronectin and collagen IV, and hyaluronidases to degrade hyaluronic acid [82] [30].
The enzymatic dissociation process must account for the substantial histological and molecular heterogeneity among RCC subtypes. Clear cell RCC (ccRCC), which accounts for 75–80% of cases, papillary RCC (pRCC, 10–15%), and chromophobe RCC (chRCC, ~5%) each present distinct challenges [80]. For instance, ccRCC is characterized by inactivation of the VHL tumor suppressor gene, leading to sustained activation of HIF signaling pathways and extensive remodeling of the tumor microenvironment [80]. The selection of dissociation enzymes, their concentrations, and incubation times must be optimized for these divergent biological contexts to successfully liberate cells while preserving viability and surface markers essential for downstream analysis.
A critical consideration in protocol design is the trade-off between dissociation efficiency and cellular integrity. Overly aggressive enzymatic treatment or prolonged incubation can compromise cell surface proteins, destroy fragile cell types, and activate stress response pathways that alter transcriptional profiles [4] [82]. Conversely, insufficient dissociation reduces yield and fails to adequately release cells for analysis. This balance is particularly crucial in RCC research, where the tumor microenvironment contains diverse immune populations, endothelial cells, and stromal elements that collectively influence tumor behavior and therapeutic responses [81] [83].
Comparison of Dissociation Methodologies and Performance Metrics
Established and Emerging Dissociation Technologies
The following table summarizes the performance characteristics of various dissociation methods when applied to renal tissues, based on recent experimental findings:
Table 1: Performance comparison of tissue dissociation technologies for renal tissues
| Technology | Dissociation Type | Tissue Utilization | Time | Viability | Key Advantages |
|---|---|---|---|---|---|
| Hypersonic Levitation & Spinning (HLS) [8] | Non-contact acoustic | 90% | 15 minutes | 92.3% | Preserves rare cell populations, maintains cell integrity |
| Traditional Enzymatic-Mechanical [8] | Enzymatic + Mechanical | 70% | 60 minutes | ~80-85% | Established protocol, widely accessible |
| Cryogenic Enzymatic Dissociation (CED) [23] | Enzymatic (low temperature) | High yield (10× conventional) | Not specified | High RNA integrity | Minimizes RNA degradation, suitable for FFPE samples |
| Mixed Modal Microfluidic [4] | Microfluidic + Enzymatic | ~400,000 cells/mg (mouse kidney) | 20-60 minutes | ~90% (epithelial) | Integrated workflow, controlled shear forces |
| Electric Field Dissociation [4] | Electrical | 5× higher than traditional (GBM) | 5 minutes | ~80% (GBM) | Rapid processing, minimal reagent use |
Quantitative Cell Yield and Viability Across Methods
The efficacy of dissociation protocols is quantitatively assessed through cell yield and viability metrics, which vary significantly across methodologies:
Table 2: Cell yield and viability metrics across dissociation methods
| Method | Cell Yield | Cell Viability | Tissue Type | Notes |
|---|---|---|---|---|
| HLS [8] | Not specified | 92.3% | Human renal cancer | Superior preservation of rare cells |
| Optimized Enzymatic [4] | ~24,000 cells/4mm biopsy | 92.75% | Human skin biopsy | Protocol-specific optimization |
| Mixed Modal Microfluidic [4] | ~75,000 epithelial cells/mg | ~90% (epithelial) | Mouse kidney tissue | Cell type-specific variability |
| Ultrasound + Enzymatic [4] | 72% ± 10% (vs. 53% sonication alone) | 91-98% | Bovine liver tissue | Combination approach enhances yield |
| Automated Mechanical [4] | 1×10^5 to 6×10^5 cells | 60-80% | Mouse lung tissue | Viability varies by tissue type |
Impact on Downstream Applications
The choice of dissociation method significantly influences the quality and reliability of subsequent analytical procedures:
Single-Cell RNA Sequencing: The HLS method demonstrates exceptional performance in preserving transcriptional fidelity, with improved gene detection sensitivity and reduced stress response signatures compared to conventional methods [8]. Similarly, the CED approach maintains RNA integrity while minimizing ribosomal contamination, crucial for accurate transcriptomic quantification [23].
Flow Cytometry and Cell Sorting: Methods that preserve surface antigen integrity, such as non-enzymatic approaches or gentle enzymatic treatments with collagenase D, are preferred for immunophenotyping applications [82]. Traditional trypsin-based protocols often degrade key surface markers, compromising cell identification and sorting purity.
Primary Cell Culture and Organoid Generation: The success of RCC organoid establishment is highly dependent on the dissociation approach. Harsh mechanical dissociation can shear fragile tumor cells, while extended enzymatic digestion may degrade surface proteins necessary for self-organization [80]. Optimal protocols balance viability with preservation of regenerative capacity.
Experimental Protocols for Renal Tissue Dissociation
Hypersonic Levitation and Spinning (HLS) Protocol
The HLS method represents a groundbreaking non-contact approach for tissue dissociation that leverages acoustic energy to generate precise hydrodynamic forces [8].
Apparatus Setup: Configure the automated tissue dissociation apparatus incorporating a triple-acoustic resonator probe within a conical confinement structure. Ensure the system integrates dissociation, fluid replacement, filtration, and output functions [8].
Sample Preparation: Place human renal cancer tissue samples (2-4 mm³ fragments) into the digestion chamber containing appropriate buffer solution. For renal tissues, utilize a balanced salt solution with calcium and magnesium to maintain tissue integrity during initial processing.
Acoustic Energy Application: Activate the hypersonic probe to generate GHz-frequency acoustic waves. This creates hypersonic streaming jets that levitate the tissue sample and induce rapid self-rotation (approximately 50-100 rpm). The 'press-and-rotate' operation generates microscale liquid jets that apply precise shear forces to tissue surfaces.
Enzyme Integration (Optional): For particularly robust renal tissues, introduce a mild enzymatic solution (e.g., low-concentration collagenase at 50-100 U/mL) to enhance dissociation. The acoustic streaming augments enzyme penetration into tissue layers while reducing required incubation time.
Collection and Filtration: Process the dissociated cell suspension through the integrated filtration system. Collect the output containing single cells while directing debris to waste chambers. Typical processing time is 15 minutes for complete dissociation of renal tissue samples.
Quality Assessment: Determine cell viability via trypan blue exclusion or automated cell counting. Assess dissociation efficiency by microscopy examination for single cells versus remaining aggregates. Expected viability exceeds 90% with minimal debris [8].
Cryogenic Enzymatic Dissociation (CED) Protocol for FFPE Tissues
The CED method specializes in nuclei extraction from formalin-fixed paraffin-embedded (FFPE) tissues, particularly valuable for archived RCC specimens [23].
Tissue Preparation: Cut FFPE tissue sections at 50 μm thickness using a microtome. Deparaffinize sections using xylene substitutes and rehydrate through graded ethanol series to aqueous buffer.
Dissociation Buffer Preparation: Prepare CED buffer containing proteinase K (optimized concentration: 0.8-1.0 mg/mL for mouse brain, may require titration for renal tissues) and sarcosyl as an anionic surfactant. Sarcosyl preferentially interacts with nuclear membranes over cell membranes, enhancing nuclear isolation.
Cryogenic Incubation: Incubate tissue sections in CED buffer at 4°C for 4-24 hours. The low temperature protects nuclear membranes while allowing gradual enzymatic digestion of cross-linked proteins. Gently agitate samples periodically to enhance penetration.
Nuclei Liberation: Following incubation, gently pipette the solution to liberate nuclei from the partially digested tissue matrix. Avoid vigorous mechanical disruption that could damage nuclear integrity.
Purification: Without filtration or sucrose cushion centrifugation (which can reduce yields), allow larger debris to settle briefly and collect the nuclei-containing supernatant. This approach preserves smaller nuclei that might be lost through standard purification methods.
Quality Control: Assess nuclear integrity and count via fluorescence microscopy with DAPI staining. Nuclear size distribution should center around 6-8 μm with minimal cytoplasmic contamination. Expected yield exceeds 100,000 nuclei per gram of starting tissue, representing a tenfold improvement over conventional methods [23].
Conventional Enzymatic Dissociation Protocol for Fresh Renal Tissues
Traditional enzymatic dissociation remains widely used for processing fresh RCC specimens obtained during nephrectomy [82] [30].
Tissue Collection and Transportation: Place freshly resected RCC tissue in cold transportation medium (e.g., HBSS with antibiotics) and process within 1-2 hours of resection. Prolonged cold ischemia time compromises cell viability and organoid-forming potential [80].
Tissue Preparation: Transfer tissue to a sterile Petri dish and mince into 3-4 mm fragments using sterile scalpels or scissors. Wash tissue pieces several times with HBSS containing calcium and magnesium to remove blood cells and debris.
Enzyme Selection and Formulation: Prepare enzyme cocktail based on tissue characteristics. For typical RCC specimens, use Collagenase D (50-200 U/mL) in HBSS with calcium and magnesium. Collagenase D is recommended when functionality and integrity of cell-surface proteins are important [82]. For fibrotic tumors, supplement with hyaluronidase (100-500 U/mL) to digest hyaluronic acid.
Digestion Process: Submerge tissue fragments in enzyme solution using a ratio of 1-4 mL enzyme solution per 100 mg tissue. Incubate at 37°C with gentle agitation on a rocker platform for 1-2 hours. Monitor dissociation progress visually and microscopically.
Termination and Dispersion: When tissue fragments appear partially digested, add complete media with serum to inhibit enzymatic activity. Gently disperse remaining tissue fragments by pipetting or passing through a 100-200 μm sterile mesh filter.
Cell Recovery and Washing: Centrifuge cell suspension at 100-300 × g for 5-10 minutes. Discard supernatant and resuspend cell pellet in appropriate buffer or culture medium. Perform viability assessment and cell counting before proceeding to downstream applications.
Essential Reagents and Research Solutions
Successful dissociation of renal tissues requires carefully selected reagents and materials. The following table outlines key components of an effective dissociation toolkit:
Table 3: Essential research reagents and materials for renal tissue dissociation
| Reagent/Material | Function | Application Notes | Representative Examples |
|---|---|---|---|
| Collagenase D [82] | Hydrolyzes collagens | Preferred for RCC when surface protein integrity is crucial; gentle on receptors | Worthington Biochemical, Sigma-Aldrich |
| Dispase [82] [30] | Cleaves fibronectin and collagen IV | Gentle enzyme; often combined with collagenase for complex tissues | Gibco Dispase, Corning |
| Hyaluronidase [4] [82] | Degrades hyaluronic acid | Supplemental enzyme for RCC with extensive ECM; enhances penetration of other enzymes | STEMCELL Technologies |
| TrypLE Express [30] | Recombinant fungal trypsin | Animal origin-free; direct substitute for trypsin with consistent activity | Gibco TrypLE |
| Cell Dissociation Buffer [30] | Non-enzymatic chelation | Gentle detachment; preserves surface markers for flow cytometry | Gibco Enzyme-Free PBS-based buffer |
| Proteinase K [23] | Broad-spectrum serine protease | Essential for FFPE tissue digestion in CED protocol; digests cross-linked proteins | Molecular biology grade |
| Sarcosyl [23] | Anionic surfactant | Nuclear membrane-friendly detergent for CED method; replaces harsh SDS | Laboratory grade |
| Triple-Acoustic Resonator Probe [8] | Generates hypersonic streaming | Core component of HLS system; enables non-contact tissue manipulation | Custom apparatus |
| Automated Tissue Dissociator [4] | Standardized mechanical disruption | Reproducible processing; reduces operator variability | Miltenyi Biotec GentleMACS |
| Shaking Water Bath [82] | Temperature-controlled incubation | Superior heat transfer for enzymatic digestion compared to air incubators | Julabo SW Series |
Analytical Workflows and Technology Comparison
Workflow Comparison: Traditional vs. Advanced Dissociation Methods
Technology Mechanism Comparison Diagram
This comprehensive performance comparison of dissociation methodologies for human renal cancer and primary kidney tissues demonstrates significant advancements beyond conventional enzymatic-mechanical approaches. The emerging Hypersonic Levitation and Spinning (HLS) technology establishes a new benchmark with 90% tissue utilization achieving 92.3% cell viability within just 15 minutes—markedly superior to traditional methods offering 70% utilization and lower viability after 60 minutes processing [8]. Similarly, Cryogenic Enzymatic Dissociation (CED) delivers a tenfold improvement in nuclei yield from FFPE tissues while maximizing RNA integrity [23]. These innovative approaches address fundamental limitations of conventional techniques, particularly in preserving rare cell populations, maintaining surface marker integrity, and minimizing stress-induced artifacts.
The optimal dissociation strategy must be selected based on specific research objectives, tissue characteristics, and downstream applications. For single-cell RNA sequencing of fresh RCC specimens, HLS provides exceptional cellular integrity and representation of tumor heterogeneity. When working with archived FFPE tissues, CED offers unprecedented nuclei yield and transcriptomic fidelity. Traditional enzymatic approaches remain valuable for standard applications where equipment for advanced methods is unavailable, though they require careful optimization of enzyme cocktails and processing parameters. As renal cell carcinoma research increasingly focuses on tumor microenvironment interactions, immune contexture, and spatial biology [80] [81] [83], the critical importance of dissociation quality in generating biologically relevant data cannot be overstated. These technological advancements in tissue dissociation thus represent enabling methodologies that will accelerate discoveries in RCC biology and therapeutic development.
Tissue dissociation, the process of breaking down complex tissue architectures into single-cell suspensions, serves as a foundational step in modern biological research and therapeutic development. This technique is vital for downstream applications including single-cell analysis, cell therapy manufacturing, and drug discovery [84] [4]. The global tissue dissociation market, valued at approximately USD 326.65 billion in 2024, is projected to grow at a CAGR of 8.62% to reach USD 746.76 billion by 2034, reflecting its critical role in advancing biomedical science [84].
Despite its importance, conventional dissociation methods present significant challenges. Traditional enzymatic and mechanical techniques often suffer from batch-to-batch variability, extended processing times (sometimes requiring hours or overnight digestion), and compromised cell viability due to mechanical stress or enzymatic damage to cell surface markers [4]. These limitations introduce substantial technical artifacts and variability that compromise experimental reproducibility and clinical outcomes, creating an urgent need for more standardized, efficient, and gentle dissociation technologies [4].
The integration of artificial intelligence (AI), automation, and standardized systems represents a paradigm shift in tissue dissociation, addressing these longstanding challenges. This transformation is enabling unprecedented levels of reproducibility, efficiency, and scalability essential for both basic research and clinical applications [85] [86]. This whitepaper examines the current state, technological advances, and future trajectory of these integrated systems within the broader context of enzymatic cell dissociation research principles.
Market Dynamics and Growth Drivers
Current Market Landscape and Segmentation
The tissue dissociation market demonstrates robust growth driven by technological advancements and increasing applications in precision medicine. Key market segments show distinct growth patterns and revenue contributions, as detailed in Table 1.
Table 1: Tissue Dissociation Market Segmentation and Growth Projections
| Segment | Market Share Dominance (2024) | Highest Growth Projection | Key Drivers |
|---|---|---|---|
| Tissue Type | Human Tissue | Animal Tissue | Direct relevance to disease modeling; use in preclinical studies |
| Product Type | Enzymatic Dissociation Kits | Mechanical Dissociation Kits | Specificity and efficiency; cost-effectiveness for fibrous tissues |
| Technology Type | Automated Systems | Manual Systems (fastest growth) | Reproducibility in clinical settings; flexibility in resource-limited settings |
| Application | Cancer Research | Stem Cell Research | Tumor heterogeneity analysis; rise in regenerative medicine |
| End User | Pharmaceutical & Biotechnology Companies | Hospitals & Diagnostic Labs | Drug discovery pipelines; personalized medicine diagnostics |
| Region | North America | Asia Pacific | Established research infrastructure; rapid industrialization and investment |
Source: [84]
The enzymatic dissociation segment currently dominates product offerings, holding a 47.9% market share, due to its extensive use in tissue and cell detachment applications [71]. However, non-enzymatic dissociation methods are gaining traction as viable alternatives, particularly in applications requiring maximal preservation of cell surface markers and viability [71].
Key Growth Drivers
Several interconnected factors are propelling the adoption of advanced tissue dissociation technologies:
- Expanding Single-Cell Analysis Market: Rapid advancements in genomics and transcriptomics have increased the demand for high-quality single-cell suspensions, driving the need for optimized dissociation protocols that maintain cell integrity while maximizing yield [84].
- Rising Investments in Cell and Gene Therapies: The growing pipeline of advanced therapeutic medicinal products (ATMPs), including CAR-T cell therapies, requires scalable and reproducible dissociation processes for manufacturing [71] [16].
- Increasing Cancer Research Funding: Institutes such as the National Cancer Institute (NCI) and pharmaceutical companies are investing heavily in oncology research, where tissue dissociation enables critical analysis of tumor heterogeneity and drug efficacy testing [84].
- Growth in Regenerative Medicine: Applications like 3D bioprinting, organ regeneration, and autologous cell therapies require precise isolation of progenitor or stem cells from various tissue sources, creating strong incentives for companies offering GMP-compliant dissociation solutions [84].
Current State of Tissue Dissociation Technologies
Conventional Methods and Limitations
Traditional tissue dissociation relies primarily on enzymatic, mechanical, or combined approaches. Enzymatic methods utilize enzymes like collagenase, trypsin, dispase, papain, and hyaluronidase to digest extracellular matrix components and intercellular junctions [4]. Mechanical methods employ physical forces through homogenizers, grinders, or pipetting to disrupt tissue architecture [84]. While widely used, these conventional approaches present significant limitations:
- Extended Processing Times: Enzymatic digestion can require several hours to complete, with some protocols extending to overnight processing, delaying downstream applications and increasing contamination risks [4].
- Cell Viability Compromise: Mechanical dissociation inflicts substantial stress on cells, leading to membrane damage, reduced viability, and apoptosis. One study noted viability rates as low as 50-60% for challenging tissues like heart tissue using automated mechanical dissociation [4].
- Enzyme-Induced Artifacts: Enzymatic treatments can damage cell surface proteins, altering phenotypic characterization and affecting downstream functional assays [4] [16]. The biological variability of enzyme preparations also introduces batch-to-batch inconsistencies that compromise experimental reproducibility [84].
- Tissue-Specific Optimization Requirements: The heterogeneous nature of different tissues necessitates extensive optimization of dissociation protocols for each tissue type, creating workflow inefficiencies and hindering standardization [4].
Emerging Non-Enzymatic and Novel Dissociation Technologies
Recent research has focused on developing alternative dissociation strategies that minimize or eliminate enzymatic digestion:
- Hypersonic Levitation and Spinning (HLS): This revolutionary contact-free approach utilizes a triple-acoustic resonator probe to levitate and spin tissue samples within a confined flow field, generating microscale "liquid jets" that exert precise hydrodynamic forces. HLS achieves 92.3% cell viability and 90% tissue utilization within just 15 minutes, dramatically outperforming traditional methods [8].
- Electric Field Facilitated Dissociation: This technique applies controlled electrical fields to disrupt tissue integrity, achieving 95% dissociation efficiency for bovine liver tissue in only 5 minutes while maintaining 90% cell viability [4].
- Ultrasound-Assisted Methods: High-frequency sonication, sometimes combined with minimal enzymatic treatment, enhances dissociation efficiency (72% with sonication plus enzymatic vs. 53% with sonication alone for bovine liver tissue) while preserving cell integrity [4].
Table 2: Performance Comparison of Tissue Dissociation Technologies
| Technology | Dissociation Efficacy | Cell Viability | Processing Time | Key Advantages |
|---|---|---|---|---|
| Traditional Enzymatic | Variable by tissue type | Often compromised | 1-3 hours (up to overnight) | Wide applicability; established protocols |
| Mechanical Dissociation | High for fibrous tissues | 50-80% (tissue dependent) | ~1 hour | Cost-effective; no enzyme variability |
| Hypersonic Levitation & Spinning (HLS) | 90% tissue utilization | 92.3% | 15 minutes | Non-contact; preserves rare cell populations |
| Electric Field Dissociation | 95% | 90% | 5 minutes | Rapid; minimal reagent requirements |
| Ultrasound Dissociation | 53-72% | 91-98% | 30 minutes | Compatible with existing workflows |
These emerging technologies demonstrate that efficient tissue dissociation can be achieved while maintaining superior cell viability and significantly reducing processing times, addressing critical limitations of conventional methods.
AI and Machine Learning in Tissue Dissociation
AI-Powered Enzyme Engineering
Artificial intelligence is revolutionizing enzyme engineering for tissue dissociation through autonomous experimental systems that dramatically accelerate the optimization process. Recent breakthroughs include:
- Autonomous Enzyme Engineering Platforms: A generalized platform integrating machine learning with biofoundry automation has demonstrated the capability to engineer enzyme variants with 16 to 26-fold improvements in specific activity within just four weeks [86]. This system combines protein large language models (LLMs) like ESM-2 with epistasis models to design high-quality mutant libraries, which are then constructed and screened using fully automated workflows.
- Machine Learning-Guided Optimization: ML algorithms efficiently navigate the vast mutational space of enzymes used in dissociation cocktails, identifying variants with enhanced specificity, stability, and activity. These systems require construction and characterization of fewer than 500 variants to achieve significant improvements, representing orders-of-magnitude increases in efficiency compared to traditional directed evolution approaches [86].
- Large Language Models for Protein Design: Protein LLMs trained on global protein sequences predict the likelihood of amino acids occurring at specific positions based on sequence context, interpreting this likelihood as variant fitness. This approach successfully identified beneficial mutations, with 59.6% of AI-designed variants performing above wild-type baseline in one case study [86].
AI-Enhanced Process Control and Optimization
Beyond enzyme engineering, AI systems are being deployed to optimize dissociation parameters and outcomes:
- Predictive Modeling for Protocol Selection: AI algorithms can recommend optimal dissociation parameters (enzyme concentrations, incubation times, mechanical forces) based on tissue characteristics (type, size, cellular composition) and desired output specifications (viability, yield, specific cell populations) [85].
- Real-Time Process Monitoring and Adjustment: Integrated AI systems monitor dissociation progress through various sensors and dynamically adjust parameters to maintain optimal conditions, ensuring consistent results despite biological variability [85] [86].
- Data Integration and Knowledge Management: AI platforms aggregate dissociation outcomes across multiple experiments and laboratories, identifying subtle correlations between protocol variations and outcomes that would be undetectable through manual analysis [85].
The following diagram illustrates the integrated AI-powered enzyme engineering workflow:
Diagram 1: AI-Powered Autonomous Enzyme Engineering Workflow. This integrated system combines machine learning with biofoundry automation to rapidly engineer improved enzymes for tissue dissociation.
Automation and Standardized Systems
Automated Tissue Dissociation Platforms
Automation technologies are transforming tissue dissociation from a manual, artisanal process into a standardized, reproducible operation:
- Integrated Automated Systems: Bench-top instruments like the STEMprep Tissue Dissociator System automate and standardize tissue dissociation for research purposes, reducing operator-dependent variability [87]. These systems integrate dissociation, fluid replacement, filtration, and output functions into seamless workflows [8].
- Biofoundry Environments: Facilities like the Illinois Biological Foundry for Advanced Biomanufacturing (iBioFAB) implement fully automated protein engineering workflows that execute mutagenesis PCR, DNA assembly, transformation, colony picking, plasmid purification, protein expression, and enzyme assays without human intervention [86]. These systems are organized into modular units for robustness and ease of troubleshooting.
- Microfluidic Dissociation Platforms: Miniaturized devices perform enzyme-free dissociation of stem cell aggregates and tissue fragments, integrating sample preparation with downstream applications. One mixed-modal microfluidic platform achieved dissociation of multiple mouse tissue types (kidney, breast tumor, liver, heart) with cell-type-specific viabilities ranging from 50% to 95% in 1-60 minutes [4].
Standardized Workflows and Protocols
Standardization efforts are addressing the critical reproducibility challenges in tissue dissociation:
- Commercially Standardized Kits: Pre-formulated dissociation kits from commercial vendors are replacing in-house preparations due to their consistency, regulatory compliance, and validation data [84]. These kits accounted for the largest revenue share in the tissue dissociation market in 2024.
- GMP-Compliant Processes: For clinical applications, standardized protocols compliant with Good Manufacturing Practice (GMP) guidelines are essential for manufacturing cell therapies. Regulatory bodies are placing increased emphasis on standardized protocols for clinical-grade cell manufacturing [84].
- Modular Workflow Design: Automated dissociation workflows are increasingly designed as interconnected modules, allowing customization while maintaining standardization within each module. This approach enables recovery from errors without restarting entire processes [86].
The following diagram illustrates an automated tissue dissociation workflow integrating multiple technologies:
Diagram 2: Automated Tissue Dissociation Workflow. This integrated system demonstrates the modular approach to standardizing tissue dissociation processes with AI-enabled monitoring and control.
Integrated Experimental Protocols
Protocol 1: AI-Optimized Enzymatic Dissociation for Complex Tissues
This protocol leverages AI-designed enzymes and automated systems for efficient dissociation of challenging tissues like tumors and connective tissues:
Tissue Preparation:
- Obtain fresh tissue sample (≤500 mg) and place in chilled dissection buffer.
- Using automated tissue processor, mince tissue into fragments of approximately 2-4 mm³.
- Transfer tissue fragments to enzyme blend solution containing AI-optimized collagenase variant (2-4 mg/mL) and neutral protease (0.5-1 mg/mL).
Automated Dissociation:
- Load sample into automated dissociator (e.g., gentleMACS Dissociator or STEMprep System).
- Program system to execute optimized dissociation protocol: 30-minute incubation at 37°C with intermittent mechanical agitation (every 7.5 minutes for 30 seconds).
- Monitor dissociation progress in real-time using integrated sensors tracking particle size distribution.
Reaction Termination and Cell Recovery:
- Automatically add enzyme inactivation solution (e.g., PBS with 10% FBS).
- Filter cell suspension through 70-100 μm strainer to remove undissociated tissue.
- Centrifuge at 300-400 × g for 5 minutes and resuspend in appropriate buffer.
- Assess cell viability and yield using automated cell counter.
Quality Control:
- Perform flow cytometry analysis for specific cell surface markers to verify preservation of epitopes.
- Conduct functional assays as required for downstream applications.
Expected Outcomes: This protocol typically yields >85% cell viability with >2×10⁶ cells per 100 mg tissue for most solid tissues, representing significant improvements over conventional methods [4] [86].
Protocol 2: Enzyme-Free Dissociation Using Hypersonic Levitation
This contact-free method maximizes cell viability and preserves delicate cell populations:
System Setup:
- Initialize Hypersonic Levitation and Spinning (HLS) apparatus with triple-acoustic resonator probe.
- Fill dissociation chamber with appropriate isotonic buffer (e.g., PBS without Ca²⁺/Mg²⁺).
- Calibrate acoustic field parameters for specific tissue type.
Sample Loading and Levitation:
- Introduce tissue sample (≤100 mg) into dissociation chamber.
- Activate hypersonic resonator to achieve tissue levitation and spinning.
- Monitor "press-and-rotate" operation via integrated camera system.
Dissociation Process:
- Maintain hypersonic streaming for 15 minutes at room temperature.
- The microscale "liquid jets" generated by acoustic streaming exert precise hydrodynamic forces to dissociate tissue while preserving cell integrity.
- Optionally, introduce minimal enzyme concentrations (10-20% of standard protocols) for particularly challenging tissues.
Cell Collection:
- Direct dissociated cell suspension to output chamber through integrated filtration system.
- Collect cells by gentle centrifugation (200 × g for 5 minutes).
- Resuspend in appropriate medium for downstream applications.
Expected Outcomes: This method achieves ~92% cell viability with 90% tissue utilization within 15 minutes, significantly outperforming traditional methods, especially for preservation of rare cell populations [8].
The Researcher's Toolkit: Essential Reagents and Systems
Table 3: Research Reagent Solutions for Advanced Tissue Dissociation
| Category | Specific Products/Systems | Key Features | Applications |
|---|---|---|---|
| AI-Designed Enzymes | Gibco TrypLE Express, Recombinant collagenases | Animal-origin-free, room temperature stable, preserved cell surface markers | Gentle dissociation of sensitive cells; GMP-compliant manufacturing |
| Non-Enzymatic Reagents | Enzyme-free dissociation buffers, Chelating agents (EDTA) | Eliminates enzyme variability, preserves surface epitopes | Flow cytometry; single-cell sequencing |
| Automated Instruments | STEMprep Tissue Dissociator, gentleMACS Dissociator | Standardized protocols, reduced operator variability | High-throughput labs; clinical applications |
| Novel Technology Platforms | Hypersonic Levitation systems, Microfluidic dissociation chips | Non-contact processing, minimal cell damage | Rare cell isolation; delicate tissues |
| Specialized Buffer Systems | Commercial dissociation media with customized additives | Optimized ionic composition, energy substrates | Specific tissue types (CNS, tumor, etc.) |
| Quality Control Tools | Viability dyes, Microfluidic cell counters, Multi-parameter flow cytometry | Rapid assessment, integrated with automated systems | Process validation; regulatory compliance |
Future Perspectives and Strategic Recommendations
Emerging Trends and Development Trajectories
The field of tissue dissociation is evolving toward increasingly integrated, intelligent, and standardized systems:
- Fully Autonomous Experimentation: Platforms that integrate AI-guided experimental design with robotic execution will become more prevalent, enabling continuous optimization of dissociation protocols with minimal human intervention [86]. These systems will leverage increasingly sophisticated protein LLMs and active learning algorithms to navigate complex experimental spaces.
- Personalized Dissociation Protocols: As single-cell technologies reveal greater heterogeneity in tissue composition, dissociation protocols will become increasingly tailored to specific tissue subtypes, disease states, and even individual patient characteristics [16].
- Point-of-Care Dissociation Systems: Miniaturized, automated dissociation platforms will enable processing of tissue samples in clinical settings, reducing processing delays and better preserving native cellular states for diagnostic applications [88].
- Enhanced Multi-Omics Integration: Dissociation technologies will be increasingly optimized for compatibility with downstream multi-omics applications (transcriptomics, epigenomics, proteomics), requiring preservation of not just viability but also molecular integrity [4] [8].
Strategic Imperatives for Stakeholders
To capitalize on these emerging opportunities, stakeholders should consider the following strategic priorities:
- Invest in Next-Generation Technologies: Prioritize investments in AI-driven automation platforms, enzyme-free dissociation solutions, and integrated systems that improve cell viability, reproducibility, and scalability [71].
- Develop Standardized Cross-Platform Protocols: Establish universally applicable standards and protocols that enable data comparison across different laboratories and platforms, addressing a critical current limitation [4] [88].
- Foster Collaborative Ecosystems: Build partnerships across academia, industry, and clinical settings to accelerate technology transfer and validate new approaches in biologically and clinically relevant contexts [71].
- Address Cost and Accessibility Barriers: Develop cost-effective solutions and flexible business models (e.g., reagent subscription services) to make advanced dissociation technologies accessible to smaller laboratories and emerging markets [71] [88].
The integration of AI, automation, and standardized systems is fundamentally transforming the landscape of tissue dissociation, addressing longstanding challenges in reproducibility, efficiency, and cell viability. These technological advances are enabling new capabilities in single-cell analysis, cell therapy manufacturing, and precision medicine that were previously constrained by the limitations of conventional dissociation methods.
As the field continues to evolve, the convergence of intelligent design systems, automated execution platforms, and standardized workflows will further accelerate progress, ultimately enabling personalized dissociation protocols tailored to specific research and clinical applications. Researchers and drug development professionals who strategically adopt and contribute to these emerging technologies will be positioned at the forefront of biomedical innovation, driving advances in our understanding of biology and development of novel therapeutics.
The future of tissue dissociation lies not in incremental improvements to existing methods, but in fundamentally reimagined approaches that leverage integrated AI and automation to achieve unprecedented levels of precision, reproducibility, and biological relevance.
Conclusion
Mastering enzymatic cell dissociation is pivotal for success in modern biomedical research, requiring a deep understanding of biochemical principles, meticulous protocol optimization, and rigorous validation. The key to success lies in carefully balancing enzyme selection and digestion parameters to maximize cell yield and viability while preserving critical cellular characteristics for downstream applications. As the field advances, future progress will be driven by the integration of AI-driven optimization, automated and closed-system platforms, and the development of gentler, more specific enzyme formulations. These innovations will be crucial for standardizing workflows, enhancing reproducibility in cell therapy manufacturing, and unlocking deeper biological insights through more accurate single-cell analyses, ultimately accelerating discoveries in regenerative medicine and personalized therapeutics.
References
- 1. Design and Applications of Extracellular Matrix Scaffolds in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12293650/]
- 2. Biomaterials in tissue repair and regeneration: key insights ... [https://www.frontiersin.org/journals/medical-technology/articles/10.3389/fmedt.2025.1565810/full]
- 3. A Comprehensive Review on Collagen Type I ... [https://www.mdpi.com/2227-9059/10/9/2307]
- 4. Recent advancements in tissue dissociation techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12622302/]
- 5. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOorloWNXdA0o9KOJYg0xqqfN6FBGWbxAxT1SRut9J4dVPgEH71Iu]
- 6. Matrix metalloproteinases as diagnostic and prognostic ... [https://pubmed.ncbi.nlm.nih.gov/40604111/]
- 7. Editorial: Interplay of immunity and ECM in cancer [https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2025.1704061/full]
- 8. Hypersonic levitation and spinning: paving the way for ... [https://www.nature.com/articles/s44172-025-00497-0]
- 9. Unlocking the Potential of Collagenases: Structures ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11458858/]
- 10. Neutral Protease (Dispase) [https://www.worthington-biochem.com/products/neutral-protease-dispase]
- 11. What is Trypsin Substrate and Its Application? [https://www.bocsci.com/resources/what-is-trypsin-substrate-and-its-application.html?srsltid=AfmBOoqWtfvDdhvhUAtCS5cKqkkjiYMskqK0132MrmEv1Q67pak3zEIZ]
- 12. Hyaluronidase and its substrate hyaluronan: biochemistry, ... [https://pubmed.ncbi.nlm.nih.gov/9839614/]
- 13. Identification of Amino Acid Residues Required for the ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3516757/]
- 14. Collagenase Chromophore-Substrate for use at 320 nm [https://www.sigmaaldrich.com/US/en/product/sigma/27667?srsltid=AfmBOorcOiTHHhM-ePqwrkANfMOarOJqIxPm3_4ArSGMqXvyoIPbLZrt]
- 15. Trypsin Substrates | SCBT [https://www.scbt.com/browse/trypsin-substrates?srsltid=AfmBOoouBNKDy_BgzyK7jOHTspQjZTyvOXqreDXdf6_P521dSKClGw0q]
- 16. Cell Dissociation in the Real World: 5 Uses You'll Actually ... [https://www.linkedin.com/pulse/cell-dissociation-real-world-5-uses-youll-actually-bazlf]
- 17. Extracellular matrix determinants of proteolytic and non- ... [https://www.sciencedirect.com/science/article/abs/pii/S0962892411001966]
- 18. Concepts of extracellular matrix remodelling in tumour ... [https://www.nature.com/articles/s41467-020-18794-x]
- 19. Pollen proteolytic enzymes degrade tight junctions [https://pubmed.ncbi.nlm.nih.gov/17986111/]
- 20. disruption of cell junctions by cellular and bacterial proteases [https://pmc.ncbi.nlm.nih.gov/articles/PMC8862089/]
- 21. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOoqPAnIx0sKmn4gJJVTkS1hHf8TcnoVcNrFSU5DFci7hWFqVZ_pT]
- 22. Proteinase Suppression by E-cadherin-mediated Cell ... [https://www.sciencedirect.com/science/article/pii/S0021925818362926]
- 23. snCED-seq: high-fidelity cryogenic enzymatic dissociation ... [https://www.nature.com/articles/s41467-025-59464-0]
- 24. Impact of enzymatic digestion on single cell suspension yield ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10592386/]
- 25. Automated—Mechanical Procedure Compared to Gentle ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9138555/]
- 26. Comparison of enzymatic digestion and mechanical ... [https://www.sciencedirect.com/science/article/pii/S0015028215003167]
- 27. An Optimized Tissue Dissociation Protocol for Single-Cell ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2022.872688/full]
- 28. Defining standard enzymatic dissociation methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5773844/]
- 29. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOoqM5Uwqdmiq_jdeqtzFUv7tT9RpvjcnrULVSSjSCMdBPnkO613J]
- 30. Enzymatic and Non-Enzymatic Cell Dissociation Protocols [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 31. Enzymatic and Mechanical Approaches for Mammalian ... [https://orbitalshakers.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation]
- 32. Automated tissue dissociation for the establishment of ... [https://www.nature.com/articles/s41598-025-03905-9]
- 33. Worthington Tissue Dissociation Guide [https://www.worthington-biochem.com/tools-resources/tissue-dissociation-guide]
- 34. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOopqqecQ-GyYvM5ozlVd08vayeuQg28aCQq-cJg4U07Mf9bWfYxz]
- 35. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOoqnKE66jMzIDMYrJBGoRJgh_8Y63zYJO_YOe-AZuam4KCeANjJY]
- 36. Tissue Dissociation and Digestion Protocols [https://www.stemcell.com/tissue-dissociation-and-digestion-protocols.html]
- 37. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOor7WMUkbZ007CS-QSIw6rhy9JSINMEasK2sUh4Dwn4q0Y2gZ7cJ]
- 38. Optical sensor reveals the hidden influence of cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11111754/]
- 39. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOoqcNa17Zmu_3uVKwTGvVVueir_LgwgsnVd2zxxSAUiEwvA5gcxq]
- 40. The Importance of Tissue Dissociation Enzymes in Cell & ... [https://levitasbio.com/the-importance-of-tissue-dissociation-enzymes-in-cell-tissue-culture/]
- 41. An Optimized Tissue Dissociation Protocol for Single-Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9096112/]
- 42. A non-aggressive, highly efficient, enzymatic method for ... [https://bmcneurosci.biomedcentral.com/articles/10.1186/s12868-016-0262-y]
- 43. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOop6QiLgrC_edBzx4nEM1ALsp4kpy5yWZlW1c5ggJGUennN8sfkG]
- 44. Factors Affecting Cell Viability during the Enzymatic ... [https://www.mdpi.com/2079-7737/13/9/665]
- 45. Nicheformer: a foundation model for single-cell and spatial ... [https://www.nature.com/articles/s41592-025-02814-z]
- 46. Recent advancements in tissue dissociation techniques for ... [https://pubmed.ncbi.nlm.nih.gov/41248140/]
- 47. Cell detachment: A review of techniques, challenges, and ... [https://www.sciencedirect.com/science/article/abs/pii/S0079610725000173]
- 48. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOoqKSNelczmn7NMYzf3XI0MHTELxS0cMmejwjFBgswxM-NrxgZwk]
- 49. Impact of enzymatic isolation on the propagation efficiency ... [https://www.nature.com/articles/s41598-025-97650-8]
- 50. Other Cells - VitaCyte [https://www.vitacyte.com/applications/other-cells/]
- 51. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOorOZD-ACtqwAl7S4TvrRf0n5rSY1BcYPK4Hnq0YFLCT7vwOh33D]
- 52. Hepatocyte Horizons: Advancing Liver Research & Drug ... [https://www.linkedin.com/pulse/hepatocyte-horizons-advancing-liver-research-drug-development-j6b4e]
- 53. Cell Dissociation and Trypsin for Cell Culture [https://www.thermofisher.com/us/en/home/life-science/cell-culture/mammalian-cell-culture/reagents/cell-dissociation.html]
- 54. Semi-rational approaches to engineering enzyme activity [https://pubmed.ncbi.nlm.nih.gov/15994074/]
- 55. Evaluation of enzymatic protocols to optimize efficiency ... [https://www.nature.com/articles/s41538-024-00313-7]
- 56. Preserving the True Immune Landscape: Eliminating ... [https://blog.cellsonics.com/blog/preserving-immune-markers]
- 57. Enzymatic Dissociation Induces Transcriptional and ... [https://www.mdpi.com/1422-0067/21/21/7944]
- 58. A novel feature for monitoring the enzymatic harvesting ... [https://www.nature.com/articles/s41598-022-22561-x]
- 59. Protocol for single-cell dissociation of head and neck ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12272886/]
- 60. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOorfZwXZm9fr1RkD8GNSrLqlpBMXLzU0PZTd7geIpImnDxeurfn9]
- 61. A Cold-Active Protease Tissue Dissociation Protocol for the ... [https://en.bio-protocol.org/en/bpdetail?id=5293&type=0]
- 62. DNA and bovine serum albumin protein (BSA) interaction ... [https://www.sciencedirect.com/science/article/abs/pii/S1226086X24003484]
- 63. Inhibitory effect of bovine serum albumin on acid ... [https://pubmed.ncbi.nlm.nih.gov/6630163/]
- 64. The synthesis, characterization, DNA/BSA/HSA interactions ... [https://pubs.rsc.org/en/content/articlehtml/2020/ra/d0ra03436c]
- 65. Quality controls after tissue dissociation for single cell RNA ... [https://singleron.bio/quality-controls-after-tissue-dissociation-for-single-cell-rna-sequencing/]
- 66. Cell Viability Assays - Assay Guidance Manual - NCBI Bookshelf [https://www.ncbi.nlm.nih.gov/books/NBK144065/]
- 67. Population-level integration of single-cell datasets enables ... [https://www.nature.com/articles/s41592-023-02035-2]
- 68. Visualizing scRNA-Seq data at population scale with GloScope [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-024-03398-1]
- 69. How Cell Separation Reagent Works — In One Simple ... [https://www.linkedin.com/pulse/how-cell-separation-reagent-works-one-simple-flow-2025-tech-metrics-lwt5f/]
- 70. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOoqOg9_hcRHM8h_IjqdfZ9CnMpBj3HNManHSqM0cqfmVJW8FmDk9]
- 71. Cell Dissociation Market Demand 2025-2035 [https://www.futuremarketinsights.com/reports/cell-dissociation-market]
- 72. Cell Dissociation Market Size 2025 to 2034 [https://www.precedenceresearch.com/cell-dissociation-market]
- 73. North America Cell Dissociation Market Update 2025 [https://www.linkedin.com/pulse/north-america-cell-dissociation-market-update-2025-uq6bf]
- 74. Microfluidics applications for high-throughput single cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8507141/]
- 75. Microfluidics add-on technologies for single-cell analysis [https://www.sciencedirect.com/science/article/abs/pii/S0165993623003448]
- 76. An acoustic levitation platform for high-content histological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12053053/]
- 77. Comparing mechanical and enzymatic isolation procedures to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11584359/]
- 78. Different methods of detaching adherent cells and their ... [https://www.nature.com/articles/s41598-022-09605-y]
- 79. Mechanical Procedure Compared to Gentle Enzymatic ... [https://www.mdpi.com/2218-273X/12/5/701/review_report]
- 80. Renal cell carcinoma organoids for precision medicine [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-025-06949-7]
- 81. Single-cell Analysis Decodes Cellular Dynamics in Clear ... [https://www.sciencedirect.com/science/article/pii/S2001037025004210]
- 82. Enzymatic and Mechanical Approaches for Mammalian ... [https://labsup.net/blogs/blog/enzymatic-and-mechanical-approaches-for-mammalian-tissue-dissociation?srsltid=AfmBOop_i3YFu6fWyyPYUwH3Qxz6Xx5jG0zd-KR6HQMgw7dx-GrWPyI2]
- 83. Metabolite profiling of human renal cell carcinoma reveals ... [https://elifesciences.org/reviewed-preprints/95652]
- 84. Tissue Dissociation Market Size 2024 To 2034 [https://www.novaoneadvisor.com/report/tissue-dissociation-market]
- 85. Cell Lysis and Disruption Market Impact of AI and Automation [https://www.linkedin.com/pulse/cell-lysis-disruption-market-impact-ai-automation-h6off/]
- 86. A generalized platform for artificial intelligence-powered ... [https://www.nature.com/articles/s41467-025-61209-y]
- 87. Cell Dissociation Market 2025 | Industry Developments, Future [https://www.openpr.com/news/4216218/cell-dissociation-market-2025-industry-developments-future]
- 88. U.S. Tissue Dissociation Market Statistics 2025-2034 [https://www.statifacts.com/outlook/us-tissue-dissociation-market]