Organoid Biobanking and Cryopreservation: A Comprehensive Guide for Advancing Translational Research
Organoid biobanking has emerged as a transformative platform for personalized medicine, disease modeling, and drug development.
Organoid Biobanking and Cryopreservation: A Comprehensive Guide for Advancing Translational Research
Abstract
Organoid biobanking has emerged as a transformative platform for personalized medicine, disease modeling, and drug development. This article provides a comprehensive overview for researchers and drug development professionals, covering the foundational principles of establishing living biobanks, current methodological approaches for cryopreservation and application, key challenges and optimization strategies, and validation frameworks for ensuring biological fidelity. By synthesizing the latest advances in cryopreservation techniques, tissue viability maintenance, and quality control, this resource aims to support the development of robust, next-generation biobanking infrastructures that can accelerate translational research and clinical implementation.
The Rise of Living Biobanks: From Tissue Preservation to Functional Organoid Repositories
Defining Next-Generation Living Biobanks (NGLB) and Their Strategic Value
Next-Generation Living Biobanks (NGLB) represent a transformative paradigm in the storage and application of biological specimens, moving beyond simple archival repositories to become dynamic platforms that support long-term sample preservation coupled with unlimited expansion of cell cultures [1]. Traditional biobanks have primarily focused on the quantity of stored samples, whereas NGLBs are characterized by their emphasis on scientific utilization and the ability to generate living resources such as conditionally reprogrammed cells and patient-derived organoids that closely recapitulate original tissue biology [2] [3]. This shift addresses critical limitations in both basic and clinical cancer research, where traditional models including conventional cell lines and animal models have demonstrated restricted applications and genomic-based precision oncology currently benefits only a small fraction of patients [2].
The strategic value of NGLBs lies in their capacity to transform biological materials into readily deployable "off-the-shelf" resources for scientific research and clinical applications [1]. By integrating advanced technologies such as conditional reprogramming, organoid culture systems, and optimized cryopreservation protocols, NGLBs enable the establishment of extensive living collections that preserve the genetic and phenotypic profiles of original tissues while allowing for continuous expansion and experimentation [2] [4]. This capability marks a significant advancement toward realizing the full potential of personalized and precision medicine by providing inexhaustible cell populations for genetic, biochemical, metabolomic, proteomic, and biological assays, including chemosensitivity testing as a functional diagnostics tool [2].
Technological Foundations of NGLBs
Conditional Reprogramming Technology
Conditional reprogramming (CR) represents a cornerstone technology for NGLBs, enabling the rapid expansion of both normal and malignant epithelial cells from diverse anatomic sites and mammalian species. This method employs a simple co-culture system with a Rho kinase inhibitor (Y-27632) in combination with fibroblast feeder cells, requiring no transfection with exogenous viral or cellular genes [2]. The technical efficiency of this approach is demonstrated by its ability to produce 2 × 10⁶ cells within just five days from a core biopsy of tumor tissue, highlighting its robust potential for biobanking applications [2].
CR technology exhibits several distinctive advantages that make it particularly suitable for NGLBs. Establishment of CR cells from both normal and tumor tissue is highly efficient, with normal CR cell cultures retaining a normal karyotype and differentiation potential, while CR cells derived from tumors maintain their tumorigenic phenotype [2]. Furthermore, CR enables enrichment of cancer cells from various clinical sources including urine (for bladder cancer), blood (for prostate cancer), and pleural effusion (for non-small cell lung carcinoma), significantly expanding the potential sources for biobank specimen acquisition [2]. The National Cancer Institute has recognized the value of this technology by incorporating it into two key programs: the Patient-Derived Cancer Model Repository (PDMR) and the Human Cancer Model Initiative (HCMI), which will be distributed through ATCC [2].
Patient-Derived Organoid Cultures
Patient-derived organoids (PDOs) have emerged as powerful three-dimensional (3D) in vitro models that closely recapitulate the histological, genetic, and functional features of their parental primary tissues [4]. Unlike traditional monolayer cell lines that are grown on plastic, organoid cultures are established using three-dimensional growth of epithelial cells in Matrigel, providing a platform that more faithfully replicates the architecture and microenvironment of native organs [2] [4]. These cultures demonstrate improved fidelity in gene expression, protein synthesis, and metabolic activity compared to conventional Two-Dimensional (2D) cell cultures [1].
Organoid technology provides physiologically relevant models for disease research, drug discovery, and personalized medicine, while simultaneously reducing reliance on animal models [1]. PDOs retain key characteristics of original tissues, including: recapitulation of tissue-specific histological features; preservation of the full spectrum of differentiated cell types and stem-cell hierarchy; maintenance of disease-associated genetic mutations and related drug response; and exhibition of cell–cell and cell–matrix interactions that generate physiological gradients of oxygen, nutrients and growth factors [4]. These attributes make PDOs exceptionally reliable for functional analyses, personalized therapies, drug-response studies for prediction medicine, and disease modeling in translational research [4].
Table 1: Global Distribution of Select Patient-Derived Organoid Biobanks
| System or Body District | Organ | Number of Samples | Country | Diagnosis | Primary or Metastatic | Reference |
|---|---|---|---|---|---|---|
| Digestive | Colorectal | 55 | Japan | Colorectal carcinoma | Primary and metastatic | [4] |
| Digestive | Colorectal | 151 | China | Colorectal carcinoma | Primary and metastatic | [4] |
| Digestive | Stomach | 46 | China | Gastric tumor | Primary and metastatic | [4] |
| Digestive | Pancreas | 31 | Switzerland | Pancreatic carcinoma | Primary and metastatic | [4] |
| Reproductive | Mammary gland | 168 | The Netherlands | Breast carcinoma | Primary and metastatic | [4] |
| Reproductive | Ovaries | 76 | The United Kingdom | High-grade serous ovarian carcinoma | Primary and metastatic | [4] |
| Urinary | Kidney | 54 | The Netherlands | Renal cell carcinoma | Not specified | [4] |
Advanced Cryopreservation Methodologies
Cryopreservation serves as a critical enabling technology for NGLBs by allowing long-term storage of living biological specimens while maintaining their structural and functional integrity [1]. Conventional cryopreservation techniques encounter significant limitations when applied to complex living systems like organoids due to their multicellular architectures and cellular heterogeneity, which make them particularly vulnerable to damage during freeze-thaw cycles [1]. This damage can manifest as ice crystal formation, oxidative stress, osmotic pressure injury, cryoprotectant toxicity, and thermal stress, ultimately resulting in membrane damage, loss of intercellular connections, and impaired biological functionality [1].
Recent advances in cryobiology have introduced innovative approaches to overcome these challenges. The development of naturally derived, low-toxicity cryoprotectants (CPAs), along with optimization of CPA loading methods and refinement of cooling and warming protocols, has significantly mitigated cryoinjury [1]. Additionally, techniques such as hydrogel microencapsulation have emerged as effective approaches to reduce mechanical damage during cryopreservation by buffering against osmotic stress and ice crystal-induced injury [1]. The development of advanced rewarming techniques including Joule heating, magnetic nanoparticle-assisted rewarming, and electrically conductive rapid-warming chips have collectively contributed to improved post-thaw viability and function of complex living specimens [1].
Strategic Value and Translational Applications
Enhancing Precision Oncology
NGLBs provide indispensable resources for advancing precision oncology by enabling functional diagnostics that complement genomic approaches. Genomics-based precision oncology currently helps only 2-20% of patients with solid cancer, creating an urgent need for functional analyses using patient-derived models [2]. NGLBs address this gap by providing platforms for chemosensitivity testing that serve as functional diagnostics tools, allowing for direct assessment of therapeutic response in patient-derived models [2]. This approach is particularly valuable given that targeted therapies against known molecular drivers in certain cancers may not be effective in different cancer types, even when the same mutation is present [2].
The application of NGLBs in precision oncology extends to their use in drug response prediction and personalized treatment selection. Multiple studies have demonstrated the ability of patient-derived organoid biobanks to predict clinical drug responses, with applications across various cancer types including colorectal, gastric, pancreatic, breast, and ovarian carcinomas [4]. For instance, one study utilizing rectal carcinoma organoids demonstrated their utility in predicting both drug and radiation response, highlighting their potential to inform personalized treatment regimens [4]. The preservation of patient-specific characteristics in NGLB resources enables the development of tailored therapeutic strategies that account for individual variations in tumor biology and drug sensitivity.
Accelerating Drug Discovery and Development
NGLBs serve as powerful platforms for streamlining drug discovery and development processes by providing physiologically relevant models for high-throughput screening and target identification. Traditional drug development approaches utilizing conventional cell lines or animal models have demonstrated limited predictive value for clinical efficacy, with only approximately 5% of oncology drugs developed based on these models eventually receiving FDA approval [2]. In contrast, patient-derived models maintained in NGLBs more faithfully represent human disease pathophysiology, potentially improving the predictive accuracy of preclinical testing [2] [4].
The applications of NGLBs in drug discovery extend across multiple domains, including compound screening, target validation, and mechanism of action studies. The ability to generate matched normal and tumor models from the same patient enables identification of tumor-specific vulnerabilities while accounting for individual genetic backgrounds [2]. Furthermore, the establishment of extensive organoid biobanks from diverse patient populations facilitates the identification of patient subgroups that may respond differently to investigational therapies, supporting the development of stratified medicine approaches [4]. The integration of multi-omics data with drug response profiles in NGLBs further enhances their utility for identifying biomarkers of therapeutic response and resistance mechanisms [4].
Table 2: Cryopreservation Assessment Parameters for Cell-Based Formulations
| Assessment Category | Specific Parameters | Significance | Reference |
|---|---|---|---|
| Viability and Function | Post-thaw viability, recovery, proliferation, differentiation | Determines maintenance of cellular integrity and biological activity | [5] |
| Oxidative Damage | DNA damage, lipid peroxidation, protein oxidation | Identifies oxidative stress from reactive oxygen species during freezing | [5] |
| Cryoprotectant Interactions | Ice recrystallization inhibition, thermal hysteresis, ice shaping | Evaluates impact on ice formation and growth | [5] |
| Drug Formulation Stability | Drug entrapment efficacy, release profile, therapeutic efficacy | Assesses stability and functionality of drug-loaded cells | [5] |
| Structural Integrity | Membrane damage, mitochondrial damage, telomere length | Identifies physical and structural damage from cryopreservation | [5] |
Experimental Protocols and Methodologies
Protocol for Establishing Conditionally Reprogrammed Cells
The conditional reprogramming technique enables rapid expansion of primary epithelial cells from tissue specimens through a specific co-culture system. The following protocol outlines the key steps for establishing CR cultures:
Tissue Processing: Mechanically dissociate and enzymatically digest fresh tissue samples (e.g., core biopsies, surgical specimens) to generate single-cell suspensions or small cell clusters using collagenase/hyaluronidase solution. Filter through 70μm cell strainers to remove undigested fragments [2].
Co-culture Establishment: Plate the cell suspension onto a layer of irradiated or mitomycin-C-treated J2 fibroblast feeder cells in complete CR medium consisting of F-medium (3:1 mixture of Ham's F-12 and Dulbecco's modified Eagle's medium) supplemented with 5% fetal bovine serum, 0.4 μg/mL hydrocortisone, 5 μg/mL insulin, 8.4 ng/mL cholera toxin, 10 ng/mL epidermal growth factor, and 10 μM Y-27632 Rho kinase inhibitor [2].
Culture Maintenance: Incubate cultures at 37°C with 5% CO₂ and change medium every 2-3 days. The Y-27632 inhibitor should be maintained in the medium for the first 72 hours and can be subsequently withdrawn for differentiation studies or maintained for continuous proliferation [2].
Cell Passaging: Passage cells at 70-80% confluence using gentle dissociation with trypsin/EDTA or non-enzymatic cell dissociation buffers. Replate cells at appropriate densities onto fresh feeder layers with complete CR medium containing Y-27632 [2].
Cryopreservation: Harvest cells at optimal density and resuspend in cryopreservation medium (e.g., 90% FBS with 10% DMSO or commercial cryopreservation solutions). Utilize controlled-rate freezing containers and store in liquid nitrogen vapor phase for long-term preservation [2] [5].
This methodology typically enables the production of 2 × 10⁶ cells within five days from a core biopsy specimen, demonstrating the rapid expansion capability of the CR system [2]. The robust nature of this technique facilitates the establishment of CR cells from both normal and tumor tissue with high efficiency while retaining critical biological characteristics of the original tissue.
Cryopreservation Workflow for Organoids
The preservation of organoids within NGLBs requires specialized protocols that account for their three-dimensional architecture and cellular complexity. The following workflow outlines an optimized cryopreservation approach for patient-derived organoids:
Pre-cryopreservation Assessment: Evaluate organoid size, morphology, and viability prior to cryopreservation. Optimal organoids typically exhibit well-defined, compact structures with minimal cellular debris. Larger organoids (>200μm) may require special consideration due to diffusion limitations [1].
Cryoprotectant Loading: Gradually introduce cryoprotectant agents (CPAs) to minimize osmotic shock. For slow freezing methods, utilize CPA solutions containing 10% DMSO in combination with serum or defined alternatives. For vitrification approaches, employ higher CPA concentrations (e.g., 6-8M total cryoprotectants) in a stepwise manner. Microfluidic systems can optimize CPA loading by precisely controlling concentration gradients and exposure times [1].
Cooling Rate Optimization: Implement controlled-rate freezing protocols tailored to specific organoid types. Typical cooling rates range from -1°C/min to -10°C/min, with slower rates generally preferred for larger or more complex structures. Utilize ice nucleation controllers to initiate crystallization at higher temperatures (-4°C to -6°C), minimizing supercooling effects [1] [6].
Long-term Storage: Maintain cryopreserved organoids at temperatures below -150°C in liquid nitrogen vapor phase to ensure long-term stability. Implement robust inventory management systems with complete sample tracking and documentation [1] [7].
Thawing and Recovery: Rapidly warm cryopreserved organoids in a 37°C water bath with gentle agitation until only a small ice crystal remains. Immediately transfer to pre-warmed culture medium and gradually dilute CPAs to prevent osmotic shock. Centrifuge gently to remove cryoprotectants and plate in appropriate matrix with optimized recovery medium supplemented with Y-27632 for the first 48-72 hours to enhance viability [1].
This protocol emphasizes the critical balance between minimizing ice crystal formation and reducing CPA toxicity, both of which are essential for maintaining organoid viability and functionality post-thaw. The application of nanowarming technologies using magnetic nanoparticles has shown particular promise for improving recovery of larger organoid structures [1].
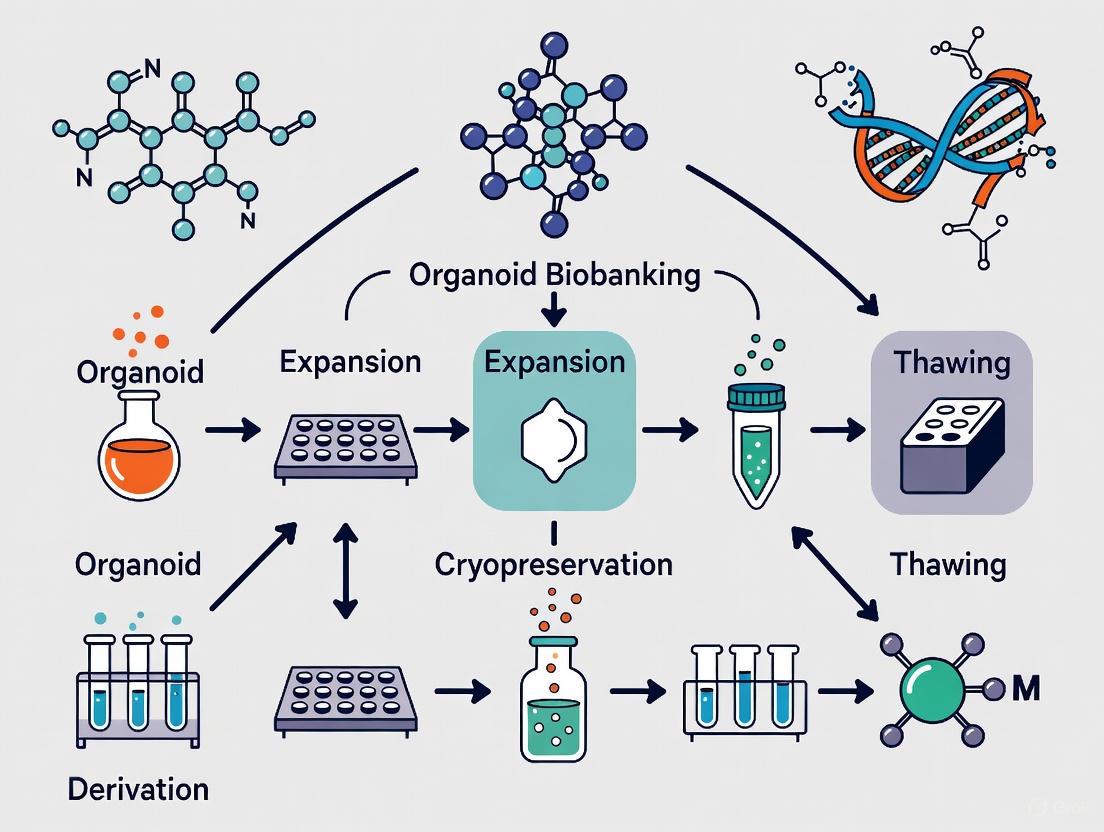
Diagram 1: Integrated Workflow for Next-Generation Living Biobanks. This diagram illustrates the key stages in establishing and utilizing NGLBs, from initial tissue processing through cryopreservation to final translational applications.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for NGLB Methodologies
| Reagent/Material | Function | Application Notes | Reference |
|---|---|---|---|
| Y-27632 (Rho kinase inhibitor) | Prevents apoptosis and enables indefinite proliferation of primary epithelial cells | Essential component of conditional reprogramming; typically used at 10μM concentration | [2] |
| J2 Fibroblast Feeder Cells | Provides necessary signaling environment for epithelial cell proliferation | Requires irradiation or mitomycin-C treatment to prevent proliferation; co-cultured with target cells | [2] |
| Matrigel | Extracellular matrix substitute for 3D organoid culture | Provides basement membrane components for structural support and signaling | [2] [4] |
| Dimethyl Sulfoxide (DMSO) | Penetrating cryoprotectant | Protects against intracellular ice formation; typically used at 5-10% concentration; potential toxicity concerns | [5] [6] |
| Trehalose | Non-penetrating cryoprotectant | Provides extracellular protection; stabilizes membranes; often combined with penetrating CPAs | [5] |
| Antifreeze Proteins | Ice-binding proteins that modify ice crystal formation | Reduces ice crystal damage; emerging as alternative to traditional CPAs | [1] [8] |
| Hydrogel Microcapsules | Physical protection during freezing/thawing | Buffers against mechanical stress from ice crystals; maintains structural integrity | [1] |
Implementation Challenges and Future Perspectives
Technical and Operational Challenges
The implementation of NGLBs faces several significant technical and operational challenges that must be addressed to realize their full potential. The complex three-dimensional structure and cellular heterogeneity of organoids make them particularly vulnerable to damage during freeze-thaw cycles, including ice crystal formation, oxidative stress, osmotic pressure injury, CPA toxicity, and thermal stress [1]. These insults can result in membrane damage, loss of intercellular connections, and impaired biological functionality, necessitating continued optimization of cryopreservation protocols [1]. Additionally, organoids typically require prolonged cultivation periods and possess limited lifespans, coupled with high heterogeneity and suboptimal reproducibility, factors that restrict their utility in long-term studies [1].
From an operational perspective, the establishment and maintenance of PDO biobanks remain technically demanding, particularly in terms of optimizing long-term culture conditions, preserving sample viability, and mimicking the tumor microenvironment [4]. The high heterogeneity of tumor tissues makes it impractical to culture organoids from every specimen and maintain individualized living biobanks for each patient to support personalized medicine, as this approach entails significant time, labor, and financial costs [1]. Furthermore, delays in processing often result in the loss of valuable research material due to limited viability of fresh tissue, and prolonged culture can lead to genetic drift, necessitating immediate processing after sample collection [1].
Emerging Solutions and Future Directions
Several innovative approaches are emerging to address the challenges facing NGLBs and enhance their capabilities. The development of decentralized biobanking platforms utilizing blockchain technology and non-fungible tokens (NFTs) shows promise for creating privacy-preserving specimen tracking and data sharing networks that connect patients, scientists, and physicians [9]. This approach has the potential to enhance efficiency, increase translational impact, and drive research discovery while bolstering ethical practices by fostering inclusion, ensuring transparency, and enhancing accountability across the research ecosystem [9].
Advancements in cryopreservation technology continue to improve the viability and functionality of preserved specimens. The comprehensive enhancement of cryopreservation technologies may facilitate the transformation of organoids into "off-the-shelf" products, enabling scalable production, batch standardization, and centralized distribution [1]. Such advancements will lay the foundation for the establishment of robust NGLBs that can support both research and clinical applications. Additionally, the integration of multi-omics technologies with functional drug response data in NGLBs is creating unprecedented opportunities for identifying novel biomarkers and understanding drug resistance mechanisms [4].
The future evolution of NGLBs will likely involve increased standardization and quality control measures to ensure reproducibility and reliability across different institutions and research programs. Furthermore, the development of specialized cryopreservation protocols for specific tissue types and disease states will enhance the utility of banked specimens for both basic research and clinical applications. As these technologies mature, NGLBs are poised to become indispensable resources that accelerate biomedical discovery and transform patient care through personalized medicine approaches.
Organoid biobanking has emerged as a cornerstone technology that bridges the gap between basic research and clinical application. By preserving living, patient-derived tissues in a biologically relevant state, these biobanks provide a robust platform for both personalized medicine and systematic drug discovery [4]. The development of reliable cryopreservation protocols has been transformative, enabling the creation of stable, living libraries of tumor tissues and patient-derived organoids (PDOs) that retain key characteristics of their source material even after long-term storage [10]. This advancement addresses a critical challenge in biomedical research: the inability of conventional frozen samples to generate living cell cultures for functional studies. The multidisciplinary rationale for organoid biobanking spans multiple domains, from clinical oncology to pharmaceutical development, creating an infrastructure that supports both individualized patient care and large-scale therapeutic screening.
Quantitative Applications of Organoid Biobanks
The translational value of organoid biobanks is demonstrated by their widespread application across multiple cancer types and research domains. The following table summarizes key quantitative data from established organoid biobanks, highlighting their scope and primary applications.
Table 1: Quantitative Overview of Patient-Derived Organoid Biobank Applications
| System/Body District | Organ | Number of Samples (Tumor/Healthy) | Country | Primary or Metastatic | Main Translational Applications |
|---|---|---|---|---|---|
| Digestive | Colorectal | 22/19 | The Netherlands | Primary | High-throughput screening [4] |
| Digestive | Colorectal | 55/41 | Japan | Primary & Metastatic | Disease modeling [4] |
| Digestive | Colorectal | 151/0 | China | Primary & Metastatic | Drug response prediction [4] |
| Digestive | Stomach | 46/17 | China | Primary & Metastatic | High-throughput screening, drug response prediction [4] |
| Digestive | Pancreas | 31/0 | Switzerland | Primary & Metastatic | Disease modeling, high-throughput screening [4] |
| Reproductive | Mammary gland | 168/0 | The Netherlands | Primary & Metastatic | Drug response prediction [4] |
| Reproductive | Ovaries | 76/0 | The United Kingdom | Primary & Metastatic | Disease modeling, drug response prediction [4] |
| Urinary | Kidney | 54/47 | The Netherlands | Information Missing | Information Missing [4] |
The data demonstrate substantial international efforts in organoid biobanking, with particular focus on gastrointestinal and reproductive cancers. The predominance of paired healthy tissue samples alongside tumor specimens enables comparative studies of disease mechanisms and therapeutic selectivity.
Core Experimental Protocols
Cryopreservation Protocol for Living Tumor Tissues
The ability to cryopreserve viable tumor tissues represents a significant advancement for creating functional biobanks. This protocol enables long-term storage with preservation of tissue viability for subsequent organoid generation [10].
Table 2: Key Reagents for Tumor Tissue Cryopreservation
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Cryopreservation Medium | CryoSure-DMSO [11], CryoStor CS10 [12] | Protects cell viability during freezing process |
| Basal Media | DMEM/F-12 with HEPES [12], RPMI [11], HBSS [11] | Provides ionic and nutrient support during processing |
| Dissociation Reagents | Gentle Cell Dissociation Reagent (GCDR) [12], Trypsin/EDTA [13] | Dissociates matrix and tissue integrity for processing |
| Supplements | Y-27632 (ROCK inhibitor) [13], Fetal Bovine Serum (FBS) [11] | Enhances survival of stem cells and vulnerable populations |
Procedure:
- Tissue Processing: Collect fresh tumor specimens in sterile conditions and transport in appropriate transport media on ice.
- Cryopreservation Solution Preparation: Prepare freezing medium containing appropriate cryoprotectants. For neural tissues, the MEDY formulation (Methylcellulose, Ethylene glycol, DMSO, and Y27632) has demonstrated efficacy [14].
- Tissue Preparation: Mince tissue into fragments of approximately 1-2 mm³ using sterile surgical blades or scalpels.
- Cryoprotectant Equilibration: Incubate tissue fragments in cryopreservation medium for 30-60 minutes at 4°C with gentle agitation.
- Controlled Rate Freezing: Transfer tissue fragments to cryovials and utilize a controlled-rate freezer, or place in an isopropanol-based freezing container at -80°C for 24 hours before transfer to long-term storage [12].
- Long-term Storage: Maintain samples in liquid nitrogen vapor phase (-135°C to -196°C) for indefinite preservation. Avoid storage at -80°C for extended periods [12].
Validation: Post-thaw viability assessment should demonstrate successful organoid generation with 95.2% success rate, with organoids retaining original tumor markers, histological features, and drug response profiles [10].
High-Throughput Screening Pipeline for 2D Organoid Models
This protocol adapts 3D organoids to 2D monolayers to enable automated, high-content screening applications while maintaining physiological relevance [13].
Procedure:
- Organoid Recovery and Expansion: Thaw cryopreserved organoids and culture in appropriate growth medium for 2 passages to ensure recovery and expansion [12].
- Monolayer Preparation:
- Coat 96-well plates with collagen IV (1:30 dilution in DI water) for 90 minutes at 37°C [13].
- Dissociate 3D organoids using 0.05% trypsin/0.5 mM EDTA for 5 minutes at 37°C [13].
- Neutralize trypsin with complete medium containing 10% FBS and pass through a 40-μm cell strainer to generate single-cell suspensions [13].
- Seed cells at optimized density (e.g., 10,000-20,000 cells/well) in L-WRN conditioned medium containing necessary growth factors and 10 μM Y-27632 [13].
- Experimental Treatment: After 24-48 hours, apply experimental conditions (e.g., drug compounds, microbial products) in triplicate with appropriate controls.
- Automated Imaging and Analysis:
- Fix and stain cells with appropriate fluorescent markers (e.g., nuclear stains, antibody conjugates).
- Image using high-throughput spinning disk confocal microscope with automated stage.
- Acquire multiple optical sections per well to ensure complete cellular capture.
- Process images using open-source software (e.g., ImageJ, CellProfiler) for quantitative analysis of fluorescence intensity, cell counting, and morphological parameters [13].
Validation: Compare pipeline results with traditional methods (e.g., flow cytometry) to ensure correlation. The system should detect inter-donor variability and cell-specific responses to experimental treatments [13].
Visualizing Workflows and Relationships
Organoid Biobanking and Application Workflow
Cryopreservation Protocol Integration
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of organoid biobanking and associated applications requires specialized reagents and materials optimized for 3D culture systems and cryopreservation.
Table 3: Essential Research Reagent Solutions for Organoid Biobanking
| Category | Specific Product/Type | Function | Application Notes |
|---|---|---|---|
| Cryopreservation Media | CryoStor CS10 [12] | Optimized cryoprotectant formulation | Maintains viability of fragile stem cell populations |
| Cryopreservation Media | MEDY Formulation [14] | Methylcellulose-based neural preservation | Specialized for neural organoids and brain tissues |
| Cryopreservation Media | 90% FBS + 10% DMSO [11] | Standard cryopreservation medium | For hematopoietic cells and PBMCs |
| Extracellular Matrices | Corning Matrigel Matrix [12] | Basement membrane extract | Provides 3D scaffolding for organoid growth |
| Dissociation Reagents | Gentle Cell Dissociation Reagent (GCDR) [12] | Enzymatic dissociation | Preserves cell surface receptors and viability |
| Dissociation Reagents | Trypsin/EDTA [13] | Proteolytic digestion | For complete dissociation to single cells |
| Basal Media | Advanced DMEM/F-12 [13] | Nutrient foundation | Standard for intestinal and many other organoid types |
| Specialized Supplements | Y-27632 (ROCK inhibitor) [13] | Inhibits apoptosis | Critical for survival post-thaw and after passage |
| Specialized Supplements | B-27 & N-2 Supplements [13] | Defined growth factors | Supports neural and various epithelial organoids |
| Coating Reagents | Collagen IV [13] | Attachment substrate | For 2D monolayer culture of organoid-derived cells |
Organoid biobanking represents a transformative multidisciplinary platform that seamlessly integrates personalized therapy with systematic drug discovery. The development of robust cryopreservation protocols that maintain tissue viability and function has been instrumental in realizing this potential, enabling the creation of living biobanks that faithfully preserve patient-specific biology. As the field advances, addressing challenges related to standardization, vascularization, and functional maturation will further enhance the translational power of these resources. The continued refinement of organoid biobanking methodologies promises to accelerate therapeutic development while simultaneously advancing precision medicine approaches that incorporate human biological diversity at their core.
Organoid technology has emerged as a transformative tool in biomedical research, creating new avenues for disease modeling, drug development, and personalized medicine. These three-dimensional, self-organizing microtissues derived from stem cells recapitulate the structural and functional complexity of their corresponding organs, offering unprecedented physiological relevance compared to traditional two-dimensional cell cultures [4] [15]. The growing importance of organoids has spurred the establishment of diverse biobanking initiatives worldwide, designed to standardize the collection, characterization, preservation, and distribution of these valuable biological resources. This document explores the current global landscape of organoid biobanks across academic, clinical, and commercial sectors, providing detailed protocols and application notes framed within a broader thesis on advancing organoid biobanking and cryopreservation research.
Global Distribution and Classification of Organoid Biobanks
The establishment of organoid biobanks has become a key strategic initiative at research institutions, medical centers, and companies worldwide. These repositories systematically collect, quality-control, and distribute living organoid models, serving as essential platforms for the scientific community.
Table 1: Global Distribution of Prominent Patient-Derived Tumor Organoid (PDTO) Biobanks
| Country/Region | Primary Institutions/Companies | Tumor Types Biobanked | Scale (Number of Organoid Lines) | Key Characteristics |
|---|---|---|---|---|
| The Netherlands | Hubrecht Institute, Utrecht University Medical Center, Royal Netherlands Academy of Arts and Sciences [15] | Colorectal, Breast, Pancreatic, Head and Neck, Intestinal, Liver, Lung, Ovarian [15] | >1,000 [15] | One of the most comprehensive; includes disease models like cystic fibrosis [15] |
| United States | Multiple academic centers and companies (e.g., Sigma-Aldrich, ATCC) [15] | Pancreatic, Breast, Colorectal, and various others from iPSCs or primary tissues [15] | Varies (e.g., 87 breast cancer lines [4]) | Includes commercial repositories; biobanks from healthy and diseased individuals [15] |
| China | Multiple academic institutions [15] | Colorectal, Rectal, Gastric, Breast, Nasopharyngeal Cancers [4] [15] | Varies (e.g., 96 rectal carcinoma lines [4]) | Focus on cancers prevalent in Asian populations; high-volume biobanks [4] [15] |
| Japan | Multiple academic institutions [4] | Colorectal, Gastroenteropancreatic Neuroendocrine Neoplasms [4] | Varies (e.g., 55 colorectal cancer lines [4]) | Mature culture techniques for specific gastrointestinal cancers [4] |
| United Kingdom | Multiple academic institutions [4] | Metastatic Colorectal, Gastroesophageal, Cholangiocarcinoma, Ovarian Cancers [4] | Varies (e.g., 110 samples from 71 patients [4]) | Focus on metastatic and post-treatment cancers [4] |
| Germany, Italy, Switzerland, Republic of Korea | Various academic institutions [4] | Colorectal, Breast, Liver, Pancreatic Cancers [4] | Varies (e.g., 106 colorectal cancer lines in Germany [4]) | Regional centers of excellence with specific organ specializations [4] |
Biobanks can be classified by their operational model:
- Academic Biobanks: Often focused on specific diseases or organs, driven by research questions, and facilitated by public funding. Examples include the Hubrecht Institute's comprehensive biobank and numerous university-associated collections [15].
- Clinical Biobanks: Integrated with hospital systems, these prioritize patient-derived models for translational research and personalized medicine applications. They are often paired with rich clinical annotation and outcome data [16].
- Commercial Biobanks: Operated by companies like Sigma-Aldrich and the American Type Culture Collection (ATCC), these provide organoids as off-the-shelf, standardized products for drug discovery and screening, ensuring broad accessibility for industrial and academic partners [15].
Establishment of an Organoid Biobank: A Protocol for Colorectal Cancer
The following detailed protocol for generating and cryopreserving colorectal cancer organoids is adapted from established methodologies [17] and serves as a representative workflow for constructing a living biobank.
Materials and Reagents
Table 2: Essential Reagents for Colorectal Cancer Organoid Culture
| Reagent/Solution | Function | Storage |
|---|---|---|
| B27 Supplement | Serum-free supplement providing essential nutrients and hormones | -20°C |
| N2 Supplement | Serum-free supplement for supporting neural and epithelial cell growth | -20°C |
| Nicotinamide | Promotes stem cell survival and self-renewal | -20°C |
| A-83-01 | TGF-β type I receptor inhibitor; prevents epithelial differentiation | -20°C |
| N-Acetyl-L-cysteine (NAC) | Antioxidant; reduces oxidative stress in culture | -20°C |
| Human Recombinant EGF | Epidermal Growth Factor; stimulates cell proliferation | -80°C |
| Human Recombinant FGF2 | Basic Fibroblast Growth Factor; supports cell growth and maintenance | -80°C |
| Y-27632 (ROCK inhibitor) | Inhibits ROCK kinase; prevents anoikis (cell death upon dissociation) | -80°C |
| Collagenase IV | Enzymatically digests tissue to isolate cells | -20°C |
| Matrigel | Extracellular matrix providing a 3D scaffold for organoid growth | -20°C |
| Advanced DMEM/F-12 | Base medium for organoid culture | 4°C |
Step-by-Step Protocol
Part A: Institutional Permissions and Tissue Collection
- Permissions: Obtain written informed consent from patients and secure approval from the relevant medical ethics committees [17].
- Tissue Collection: Aseptically collect fresh colorectal cancer tissue during surgical resection or biopsy.
- Transport: Immediately place the tissue in cold, antibiotic-supplemented tissue collection medium (e.g., DMEM/F12 with gentamicin) and transport to the laboratory on ice for prompt processing [17].
Part B: Processing and Primary Culture
- Wash and Mince: Wash the tissue in cold PBS to remove blood and contaminants. Using sterile instruments, mince the tissue into approximately 1 mm³ fragments.
- Enzymatic Digestion: Incubate the tissue fragments in tissue digestion medium (e.g., DMEM/F12 containing 1 mg/mL Collagenase IV and 10 µM Y-27632) for 30-60 minutes at 37°C with gentle agitation [17].
- Dissociation and Filtering: Mechanically dissociate the digested tissue by pipetting. Pass the resulting cell suspension through a 70-100 µm cell strainer to remove undigested fragments and debris.
- Cell Pellet Collection: Centrifuge the filtered suspension to pellet the cells. Wash the pellet with advanced DMEM/F-12.
- Embedding in Matrix: Resuspend the cell pellet in a small volume of cold Matrigel or a similar extracellular matrix. Plate the suspension as droplets in a pre-warmed culture dish and allow the matrix to polymerize at 37°C for 10-20 minutes.
- Overlay with Medium: Carefully add pre-warmed colorectal cancer organoid maintenance medium, supplemented with 10 µM Y-27632 for the first 2-3 days to enhance initial cell survival [17]. The medium is typically composed of Advanced DMEM/F-12, 1x B27, 1x N2, 1mM N-Acetyl-L-cysteine, 10mM Nicotinamide, 50 ng/mL EGF, 20 ng/mL FGF2, 500 nM A-83-01, 10 µM Y-27632, and antibiotics [17].
- Culture Maintenance: Culture the organoids at 37°C in a 5% CO₂ incubator. Refresh the medium every 2-3 days. Passage organoids every 1-2 weeks by mechanically breaking them up and/or enzymatically dissociating them, followed by re-embedding in fresh matrix.
Part C: Cryopreservation and Thawing
- Harvesting: Collect well-developed organoids from the matrix. For Matrigel cultures, this can be done by mechanically disrupting the dome and dissociating into small clusters.
- Cryopreservation Solution: Prepare a cryopreservation solution such as MEDY, which contains Methylcellulose, Ethylene glycol, DMSO, and Y27632, or use a standard solution like 90% FBS + 10% DMSO [14] [18].
- Freezing: Suspend the organoid fragments in the cryopreservation solution. Transfer to a cryovial and cool at a controlled rate of -1°C/minute to -80°C using a freezing container. The next day, transfer the vial to liquid nitrogen for long-term storage [18].
- Thawing: To recover, rapidly thaw the cryovial in a 37°C water bath. Immediately transfer the organoid suspension to a tube containing pre-warmed culture medium.
- Washing and Re-culture: Gently pellet the organoids by centrifugation to remove the cryoprotectant. Resuspend in fresh Matrigel and culture as described in Part B, ensuring the medium contains 10 µM Y-27632 for the first few days post-thaw [17].
Diagram 1: Workflow for Organoid Biobanking
Key Applications in Research and Drug Development
Disease Modeling and Drug Screening
Organoid biobanks are powerful platforms for understanding disease mechanisms and conducting drug screens. They preserve patient-specific genetic mutations and histological features, enabling the study of tumor heterogeneity and disease progression [4] [15]. High-throughput drug testing protocols using organoids have been established to identify effective therapeutic agents and predict patient responses.
Protocol for High-Throughput Drug Testing (e.g., Colorectal Cancer Organoids) [17]:
- Preparation: Thaw and expand colorectal cancer organoids as per the protocol in Section 3.2.
- Dissociation and Plating: Mechanically or enzymatically dissociate organoids into small fragments or single cells. Plate them in a 384-well plate pre-coated with Matrigel, ensuring uniform distribution.
- Drug Addition: After 24-48 hours, add a library of drugs or compounds to the wells using a liquid handler, creating a concentration gradient for each drug.
- Incubation: Incubate the plate for a predetermined period (e.g., 5-7 days), refreshing the drug-containing medium as needed.
- Viability Assay: Assess cell viability using assays like CellTiter-Glo 3D, which measures ATP content as a proxy for metabolically active cells.
- Image Analysis: Quantify organoid size, number, and morphology using high-content imaging systems to determine drug effects.
Personalized and Precision Medicine
The ability to generate organoids from individual patients and test drug sensitivities ex vivo positions biobanks at the forefront of personalized medicine. For example, a biobank of rectal cancer organoids was used to predict patient responses to neoadjuvant chemoradiotherapy with high accuracy (84.43%), sensitivity (78.01%), and specificity (91.97%) [15]. Beyond oncology, living biobanks of inflammatory bowel disease organoids have been used to identify molecular subtypes of Crohn's disease and link them to clinical outcomes, demonstrating their utility as predictive tools for complex inflammatory diseases [16].
Emerging Concepts and Standardization
Decentralized Biobanking and the "Biomediverse"
A novel paradigm known as decentralized biobanking ("de-bi") is emerging, which leverages blockchain technology and non-fungible tokens (NFTs) to create de-identified digital twins of patients, physicians, scientists, and biospecimens [9]. This approach aims to overcome the fragmentation of the current biobanking ecosystem by creating a transparent, auditable overlay network. It empowers patients by keeping them connected to the research use of their specimens in a privacy-preserving manner, thereby enhancing ethical practices, inclusion, and collaboration across a "biomediverse" [9].
The Critical Role of Cryopreservation and Next-Generation Living Biobanks (NGLB)
Cryopreservation is a cornerstone technology for viable organoid biobanking, enabling long-term storage, quality control, and distribution. The ultimate goal is to develop robust protocols that transform organoids into "off-the-shelf" products, facilitating scalable production and batch standardization [18]. This lays the foundation for Next-Generation Living Biobanks (NGLB). Key challenges and innovative strategies in cryopreservation include:
- Challenges: Cryoinjury from ice crystal formation, osmotic stress, and CPA toxicity, exacerbated by organoids' complex 3D structure [18].
- Innovative Strategies:
- Development of low-toxicity, natural cryoprotectants (e.g., antifreeze proteins) [18].
- Optimized CPA loading and unloading using microfluidic systems [18].
- Advanced rewarming techniques (e.g., magnetic nanoparticle-assisted Joule heating) for ultra-rapid and uniform warming [18].
- Hydrogel microencapsulation to provide a protective buffer during freezing and thawing [18].
Diagram 2: Signaling in Colorectal Cancer Organoid Culture
Moving Toward International Standards
The rapid growth of the field has highlighted the pressing need for standardized practices to ensure reproducibility and quality. In response, international standards are being developed. For instance:
- The ISO/WD 25630-1 standard is under development, specifying requirements for biobanking human intestinal organoids and intestinal cancer organoids [19].
- The ISoOR International Standard for Organoid Biobanking (ISoOR-ISOB) was officially released in 2025, providing a unified framework for sample collection, processing, storage, and ethical considerations [20].
These standards are critical for harmonizing biobanking practices, enhancing data comparability, supporting regulatory compliance, and fostering global collaboration.
The global landscape of organoid biobanks is dynamic and expanding, playing an indispensable role in bridging basic research and clinical application. Academic, clinical, and commercial repositories collectively provide the biomedical community with well-characterized, physiologically relevant models that drive innovation in disease modeling, drug discovery, and personalized therapy. The continued refinement of establishment and cryopreservation protocols, the adoption of international standards, and the exploration of innovative concepts like decentralized biobanking are poised to enhance the scalability, reproducibility, and ethical foundation of these vital resources. As these elements converge, organoid biobanks will increasingly become the cornerstone of translational research and precision medicine.
Key Technical and Logistical Challenges in Biobanking
Biobanking has evolved from simple collections of frozen specimens into complex infrastructures integral to modern translational research and precision medicine [21]. This evolution is particularly evident in the realm of organoid biobanking, where living, patient-derived three-dimensional (3D) microtissues are preserved for disease modeling, drug screening, and regenerative medicine applications [4] [22]. These "living biobanks" preserve not only biological material but also crucial physiological functions, offering unprecedented opportunities to study human biology and disease. However, the establishment and maintenance of such biobanks present a unique set of technical and logistical challenges that must be systematically addressed to ensure their scientific utility, reproducibility, and clinical translatability. This application note details these challenges within the context of a broader thesis on organoid biobanking and cryopreservation research, providing structured data, detailed protocols, and visual workflows to guide researchers and biobank operators.
Key Challenges in Organoid Biobanking
The transition from traditional biobanks to living biobanks based on 3D organoid models introduces complexities related to culture stability, preservation fidelity, functional reproducibility, and data management.
Table 1: Key Technical Challenges in Living Organoid Biobanks
| Challenge Category | Specific Technical Hurdles | Impact on Research & Translation |
|---|---|---|
| Long-Term Culture & Viability | Gradual loss of cellular heterogeneity and function; Internal necrosis due to lack of vascularization; Maintaining stem-cell hierarchy and differentiated cell types [4] [23]. | Compromised model fidelity; Reduced predictive power for drug screening and disease modeling [24]. |
| Cryopreservation & Revival | Low post-thaw viability rates; Compromised structural integrity and function; Cryoprotectant agent (CPA) cytotoxicity; Ice crystal formation causing physical damage [10] [23]. | Limited scalability and on-demand availability; Batch-to-batch variability; Impediment to high-throughput applications [10]. |
| Standardization & Reproducibility | Batch-to-batch variability of extracellular matrix (ECM) materials like Matrigel; Lack of uniform protocols for culture and differentiation; Difficulties in controlling organoid size, shape, and cell composition [24] [23]. | Poor inter-lab reproducibility; Hindered data sharing and collaboration; Slowed clinical adoption [22] [25]. |
| Data Integration & Management | Managing large, multi-modal datasets (genomic, clinical, imaging); Ensuring data privacy and interoperability; Tracking chain of custody and sample provenance [21] [9] [25]. | Ethical concerns over patient data; Inefficient use of biobank resources; Fragmented research ecosystem [9]. |
The establishment of patient-derived organoid (PDO) biobanks is a growing international effort. The table below summarizes a selection of existing tumor PDO biobanks, highlighting the scale, geographic distribution, and primary applications of these resources.
Table 2: Global Distribution and Characteristics of Selected Tumor PDO Biobanks
| System/Body District | Organ | Number of Samples (Tumor/Healthy) | Country | Diagnosis | Main Translational Applications |
|---|---|---|---|---|---|
| Digestive | Colorectal | 55 / 41 | Japan | Colorectal Carcinoma | Disease modeling [4] |
| Digestive | Colorectal | 151 / 0 | China | Colorectal Carcinoma | Drug response prediction [4] |
| Digestive | Stomach | 46 / 17 | China | Gastric Tumor | High-throughput screening, drug response prediction [4] |
| Reproductive | Mammary Gland | 168 / 0 | The Netherlands | Breast Carcinoma | Drug response prediction [4] |
| Reproductive | Ovaries | 76 / 0 | The United Kingdom | High-Grade Serous Ovarian Carcinoma | Disease modeling, drug response prediction [4] |
| Urinary | Kidney | 54 / 47 | The Netherlands | Renal Cell Carcinoma | Not Specified in Excerpt [4] |
Detailed Experimental Protocols
Protocol: Cryopreservation and Revival of Viable Tumor Organoids
This protocol, adapted from a 2025 study, enables the successful cryopreservation of live tumor tissues with a 95.2% success rate in post-thaw organoid generation, preserving the tumor microenvironment (TME) for downstream applications [10].
3.1.1 Principle The protocol utilizes optimized freezing and thawing conditions with specific cryoprotectants to minimize ice crystal formation and maintain tissue viability and organizational structure for deriving organoids and air-liquid interface (ALI) cultures.
3.1.2 Reagents and Materials
Table 3: Key Research Reagent Solutions for Organoid Cryopreservation
| Reagent/Material | Function/Description | Example/Note |
|---|---|---|
| Cryoprotectant Solution | Penetrates cells to depress freezing point and minimize ice crystal formation. | Typically contains 10% DMSO and a high concentration (e.g., 90%) of FBS [10]. |
| Programmable Freezer | Enables controlled-rate freezing for reproducible and controlled cooling. | Critical for standardizing the freezing process to maximize viability. |
| Liquid Nitrogen Storage | Provides long-term storage at temperatures below -135°C for sample preservation. | - |
| Recovery Culture Medium | Supports organoid growth and proliferation post-thaw. | Organoid-specific medium with growth factors (e.g., Wnt3A, Noggin, R-spondin) [4] [24]. |
| Extracellular Matrix (ECM) | Provides a 3D scaffold for organoid attachment and growth after thawing. | Matrigel or advanced synthetic hydrogels [10] [23]. |
3.1.3 Workflow Diagram
3.1.4 Step-by-Step Procedure
- Tissue Processing: Immediately after resection, wash the fresh tumor tissue thoroughly with cold, sterile phosphate-buffered saline (PBS) to remove blood and debris. Mince into approximately 1-2 mm³ fragments using sterile surgical blades.
- Cryoprotectant Agent (CPA) Equilibration: Transfer the tissue fragments to a cryovial containing a pre-chilled cryopreservation solution (e.g., 10% DMSO in 90% FBS). Gently mix and keep the vial on ice for 30-60 minutes to allow for full penetration of the CPA.
- Controlled-Rate Freezing: Place the cryovials in a programmable freezer. Initiate the freezing protocol: -1°C per minute until the temperature reaches -80°C. This slow, controlled cooling is critical to prevent lethal intracellular ice formation.
- Long-Term Storage: After completing the controlled-rate freezing, quickly transfer the cryovials to a long-term storage system, such as the vapor phase of a liquid nitrogen tank (typically below -135°C).
- Rapid Thawing: When needed, retrieve the vial and immediately thaw it by gentle agitation in a 37°C water bath for 2-3 minutes, or until only a small ice crystal remains.
- CPA Removal and Washing: Transfer the thawed tissue fragment to a tube containing 10 mL of pre-warmed organoid recovery medium. Gently invert to mix. Let the tissue settle or centrifuge briefly at low speed (100-200 x g for 5 minutes). Carefully aspirate the supernatant. Repeat this washing step twice to ensure complete removal of the cytotoxic CPA.
- Plating and Culture: Embed the washed tissue fragment in a droplet of ECM (e.g., Matrigel) and plate it in a pre-warmed culture dish. Polymerize the ECM at 37°C for 20-30 minutes, then carefully overlay with organoid recovery medium. Refresh the medium every 2-3 days. Organoids can typically be observed and passaged within 1-3 weeks. Alternatively, the tissue can be used to establish an ALI culture to preserve the native TME for immunotherapy applications [10].
3.1.5 Quality Control and Validation
- Viability Assessment: Use viability stains (e.g., Trypan Blue) on dissociated cells from thawed tissues.
- Characterization: Confirm that organoids derived from frozen tissues retain key features of the original tumor through histology (H&E staining), immunohistochemistry (for tumor markers), and genomic analyses (Whole Genome Sequencing - WGS) [10].
- Functional Validation: Perform drug sensitivity tests on organoids derived from frozen tissues and compare the response profiles to those from fresh tissue-derived organoids to ensure predictive fidelity is maintained [10].
Protocol: Establishing an Organoid-Immune Co-Culture for Immunotherapy Screening
This protocol outlines the establishment of a co-culture system combining tumor organoids and immune cells, a critical tool for evaluating cancer immunotherapies like immune checkpoint inhibitors (ICIs) and CAR-T cell therapies [24].
3.2.1 Principle This model reconstitutes critical elements of the tumor immune microenvironment (TIME) by co-culturing patient-derived tumor organoids with autologous immune cells, such as peripheral blood mononuclear cells (PBMCs) or tumor-infiltrating lymphocytes (TILs). This allows for the ex vivo study of immune cell recruitment, tumor cell killing, and response to immunotherapeutic agents.
3.2.2 Workflow Diagram
3.2.3 Step-by-Step Procedure
- Generate Tumor Organoids: Establish and expand PDOs from the patient's tumor tissue using standard 3D culture protocols in an ECM like Matrigel [4] [24].
- Isolate Autologous Immune Cells:
- From Blood: Isolate PBMCs from the patient's blood sample using a Ficoll density gradient centrifugation.
- From Tumor Tissue: Isolate TILs from a portion of the fresh tumor tissue using mechanical disaggregation and enzymatic digestion (e.g., with collagenase and DNase), followed by expansion in vitro with T-cell media containing cytokines like IL-2.
- Establish Co-culture: Harvest mature, well-established organoids and plate them in a low-attachment plate. Add the isolated immune cells (e.g., PBMCs or TILs) at a desired effector-to-target ratio (e.g., 10:1) in an appropriate co-culture medium, often supplemented with IL-2 (e.g., 100 IU/mL) to support T-cell survival.
- Treatment: Introduce the immunotherapeutic agent of interest. For example, add anti-PD-1 or anti-PD-L1 antibodies at clinically relevant concentrations to the co-culture system.
- Analysis and Monitoring: Monitor the co-culture over 3-7 days using various endpoints:
- Imaging: Use live-cell imaging to track immune cell- organoid interactions and organoid integrity.
- Cytotoxicity: Measure tumor cell death using assays like lactate dehydrogenase (LDH) release or Caspase-3 activation.
- Flow Cytometry: Analyze immune cell phenotypes (e.g., activation/exhaustion markers like PD-1, Tim-3, Lag-3) and proliferation.
- Cytokine Profiling: Quantify cytokine secretion (e.g., IFN-γ, TNF-α, IL-10) in the supernatant using multiplex ELISA.
Emerging Solutions and Future Perspectives
Technological Innovations
Several advanced technologies are being developed to overcome the inherent challenges of organoid biobanking:
- Nanomaterial-Based Strategies: Nanomaterials are used to improve ECM properties and cryopreservation outcomes. For instance, calcium silicate nanowires or magnetic nanoparticles (MNPs) can be incorporated into hydrogels to provide better mechanical support and mimic the natural ECM [23]. In cryopreservation, MNPs enable "nanowarming," which ensures uniform and rapid thawing of samples, significantly improving post-thaw survival rates compared to conventional water bath methods [23].
- Decentralized Biobanking Platforms: Blockchain technology and Non-Fungible Tokens (NFTs) are being explored to create decentralized biobanking platforms ("de-bi"). These platforms create de-identified "digital twins" of patients, specimens, and processes, enabling privacy-preserving tracking of sample provenance and usage. This enhances transparency, fosters donor inclusion, and facilitates collaboration across a networked "biomediverse" while maintaining ethical standards [9].
- AI and Data Integration: Artificial Intelligence (AI) and machine learning are increasingly applied to manage and analyze the vast, multi-modal datasets generated by biobanks. AI algorithms can enhance phenotype categorization from biobank data, improve image analysis speed, and help identify biomarkers from complex organoid drug-response data, thereby unlocking greater value from biobank resources [21] [24].
Logistical and Ethical Considerations
Beyond technical hurdles, establishing a biobank requires navigating significant logistical and ethical landscapes:
- Standardization and Quality Control: Implementing rigorous quality management standards (e.g., ISO 20387:2018) is essential to ensure sample and data quality, compliance, and reliability. The principle of "garbage-in, garbage-out" underscores the need for strict protocols from sample acquisition to data annotation [21] [25].
- Data Privacy and Informed Consent: The collection and use of biological samples and associated clinical data raise critical issues of informed consent, privacy, and confidentiality. Balancing open data access for innovation with the protection of donor information is a persistent challenge, especially in a global research environment with varying regulations [26] [9] [25].
- Cost and Infrastructure: Maintaining biobanks, particularly living biobanks requiring specialized culture facilities and cryogenic storage, demands significant financial investment and robust physical infrastructure, posing a substantial challenge for long-term sustainability [26] [25].
The establishment of organoid biobanks is a cornerstone of advancing personalized medicine and translational research. While the challenges related to culture stability, cryopreservation, standardization, and data management are significant, they are not insurmountable. By adopting robust, detailed protocols like those outlined for cryopreservation and immune co-culture, and by leveraging emerging technologies such as nanomaterials, blockchain, and AI, researchers can enhance the fidelity, reproducibility, and utility of these invaluable resources. Successfully navigating both the technical and logistical hurdles will pave the way for biobanks to fully realize their potential as dynamic platforms for discovery, ultimately accelerating the development of novel therapeutics and personalized treatment strategies for patients.
Cryopreservation Protocols and Translational Applications in Research and Medicine
1. Introduction Within the framework of organoid biobanking and advanced therapy medicinal product (ATMP) development, cryopreservation is a cornerstone technology for ensuring the long-term viability and functionality of living biospecimens. The selection of an optimal cryopreservation protocol—primarily between the conventional method of slow freezing and the more rapid vitrification technique—is critical for maintaining the complex cellular architecture and biological fidelity of these sophisticated models. This application note provides a comparative analysis of these two fundamental techniques, synthesizing recent empirical evidence and meta-analyses to guide researchers and drug development professionals in establishing robust biobanking protocols.
2. Summary of Comparative Performance Data The efficacy of slow freezing and vitrification has been extensively evaluated across diverse tissue types, including ovarian tissue, testicular tissue, and oocytes. Key quantitative outcomes from recent studies are consolidated in the table below for direct comparison.
Table 1: Comparative Outcomes of Slow Freezing vs. Vitrification Across Different Tissues
| Tissue Type / Metric | Slow Freezing Performance | Vitrification Performance | Study Conclusions |
|---|---|---|---|
| Human Ovarian Tissue (Post-Transplant) | Lower estradiol levels at 6 weeks; Higher stromal cell apoptosis at 4 weeks [27]. | Higher estradiol levels (VF2 protocol); Lower stromal cell apoptosis; Better normal follicle proportion [27]. | Vitrification, particularly the VF2 protocol, showed superior recovery of endocrine function and follicular health [27]. |
| Follicular Viability (Meta-Analysis) | No statistically significant difference from vitrification (RR = 0.96, 95% CI: 0.84–1.09) [28] [29]. | No statistically significant difference from slow freezing [28] [29]. | Both techniques provide comparable outcomes for follicular viability and primordial follicle integrity [28] [29]. |
| Human Oocytes | Survival rate: 65.1% (traditional thaw) vs. 89.8% (modified thaw). Clinical pregnancy rates improved with modified protocol [30]. | Survival rate: 89.7%. Clinical pregnancy rate: 30.1% [30]. | With an optimized thawing protocol, slow freezing outcomes can be equivalent to vitrification [30]. |
| Neonatal Calf Testicular Tissue | Better seminiferous tubule integrity; Uncontrolled method showed higher apoptosis [31]. | Lower tubule integrity, but equivalent germ cell density, proliferation, and gene expression. Protected against apoptosis [31]. | Vitrification is an effective alternative, preserving key cellular functions despite structural changes [31]. |
| General CGT Industry Adoption | High adoption (87%), especially for late-stage clinical products. Considered the standard [32]. | Less prevalent, but valued for its rapidity and lower equipment cost in specific applications [27] [32]. | Controlled-rate freezing is the industry norm for its process control, though vitrification has niche advantages [32]. |
3. Detailed Experimental Protocols To facilitate replication and standardization, detailed methodologies for key cited experiments are provided below.
3.1. Protocol: Ovarian Tissue Vitrification and Warming (VF2 Method) This protocol, adapted from a 2024 study, demonstrated superior endocrine function recovery post-transplantation [27].
Cryoprotectant Solutions:
- Equilibration Solution: 10% Ethylene Glycol (EG) + 10% Dimethylsulphoxide (DMSO) + 20% Serum Substitute Supplement (SSS) in M199 medium.
- Vitrification Solution: 20% EG + 20% DMSO + 0.5 M sucrose + 20% SSS in M199 medium.
- Warming Solutions: Thawing solution (1 M sucrose + 20% SSS), followed by serial dilutions (0.5 M, 0 M, 0 M sucrose, all with 20% SSS).
Procedure:
- Equilibration: Incubate ovarian tissue cubes in Equilibration Solution for 25 minutes at room temperature (RT).
- Vitrification: Transfer tissue to Vitrification Solution for 15 minutes at RT.
- Cooling: Plunge tissue directly into liquid nitrogen.
- Warming: Submerge vitrified tissue in pre-warmed (37°C) Thawing Solution for 1 minute.
- Dilution: Sequentially incubate tissue in sucrose solutions (0.5 M, 0 M, 0 M) for 5 minutes each at RT [27].
3.2. Protocol: Slow Freezing of Oocytes and Modified Thawing This protocol highlights a critical modification in the thawing process that significantly improves outcomes for slow-frozen oocytes, making them comparable to vitrified ones [30].
Freezing Solution: 1.5 M Propanediol (PrOH) + 0.2 M or 0.3 M sucrose in a base medium.
Freezing Procedure:
- Loading: Incubate oocytes in freezing solution for 15 minutes at RT and load into straws.
- Programmable Freezing: Use a controlled-rate freezer.
- Cool from 20°C to -6.5°C at -2°C/min.
- Hold for 5 minutes, then seed (induce ice formation).
- Hold for an additional 10 minutes.
- Cool from -6.5°C to -30°C at -0.3°C/min.
- Cool rapidly to -150°C at -50°C/min.
- Transfer to liquid nitrogen for storage [30].
Modified Thawing/Rehydration Procedure:
- Thaw: Warm straw in air for 30 seconds, then in a 30°C water bath.
- PrOH Removal: Remove oocytes and place them directly into a 1.0 M sucrose solution (instead of a step-down PrOH/sucrose mixture).
- Further Dilution: Transfer oocytes to decreasing concentrations of sucrose (e.g., 0.5 M, 0.25 M, 0.125 M).
- Final Rinse: Rinse in a sucrose-free medium [30]. This "sucrose-only" rehydration minimizes osmotic shock and improves survival rates.
4. Workflow Visualization: Method Selection for Organoid Cryopreservation The following diagram outlines a logical decision pathway for selecting and implementing a cryopreservation method for organoids, based on research priorities and practical constraints.
5. The Scientist's Toolkit: Key Reagents and Materials Successful cryopreservation relies on a suite of specialized reagents and equipment. The table below details essential components for setting up these protocols.
Table 2: Essential Research Reagent Solutions for Cryopreservation
| Reagent / Material | Function | Application Notes |
|---|---|---|
| Permeating Cryoprotectants (e.g., DMSO, EG, PrOH) | Penetrate cells to depress freezing point and prevent intracellular ice crystal formation [27] [30] [31]. | DMSO is common in slow freezing [31]; EG and DMSO are often combined for vitrification [27]. PrOH is used for oocytes [30]. |
| Non-Permeating Cryoprotectants (e.g., Sucrose) | Create an osmotic gradient, drawing water out of cells to dehydrate them before freezing [27] [30]. | Concentration is critical (e.g., 0.2-1.0 M). Higher concentrations often used in vitrification solutions and modern thawing protocols [27] [30]. |
| Serum Substitute Supplement (SSS) | Provides macromolecules that stabilize cell membranes and reduce cryo-injury [27]. | A defined, consistent replacement for fetal bovine serum in cryomedium formulations. |
| Programmable Controlled-Rate Freezer | Precisely controls cooling rates according to defined protocols, ensuring reproducibility [30] [32]. | Essential for standardized slow freezing. Industry standard for late-stage clinical products [32]. |
| Liquid Nitrogen | Provides the ultra-low temperature (-196°C) required for long-term storage of cryopreserved samples [27] [31]. | Used for both storage and as the cooling medium for vitrification. |
| Metal Containers (for closed vitrification) | Enables rapid cooling rates necessary for vitrification while maintaining a closed system, reducing contamination risk [33]. | An important alternative to open carrier systems for adhering to strict Good Manufacturing Practice (GMP). |
| Nanomaterials (e.g., Magnetic Nanoparticles) | Emerging tools for advanced cryopreservation. Act as heterogeneous nucleation sites to control ice crystal formation or enable uniform "nanowarming" [23]. | A developing technology to improve survival rates for complex tissues and organoids. |
6. Conclusion and Future Perspectives The choice between slow freezing and vitrification is not absolute but context-dependent. While meta-analyses indicate comparable follicular viability between the two methods [28] [29], specific applications—such as preserving the endocrine function of ovarian tissue [27] or the functionality of male germ cells [31]—may benefit from the optimized vitrification protocols. For organoid biobanking and ATMPs, slow freezing currently dominates the industrial landscape due to its superior process control and scalability [32]. However, the future points toward protocol harmonization rather than competition. The integration of novel nanomaterials for ice inhibition and nanowarming [23], the development of closed vitrification systems [33], and the refinement of thawing protocols [30] are poised to enhance the effectiveness of both foundational techniques, ultimately ensuring the reliable preservation of complex biological systems for research and therapy.
Optimized Media Formulations for Different Tissue Types
Organoid technology has revolutionized biomedical research by enabling the three-dimensional culture of stem cell-derived microtissues that mimic the structural and functional aspects of human organs [34]. The success of organoid cultures critically depends on precisely formulated media containing specific growth factors and signaling molecules that direct stem cell fate toward target tissues [35]. Unlike traditional 2D cell culture systems, organoid media must recapitulate the complex stem cell niche of the corresponding in vivo environment, providing not only nutrients but also precise developmental cues [36]. The core principle underlying organoid media formulation involves activating and inhibiting specific signaling pathways to mimic the natural processes of organ development and tissue homeostasis [35].
The foundational components of most organoid media include R-spondins (potentiating Wnt pathway activity) and BMP signaling antagonists such as Noggin or Gremlin 1, which prevent differentiation and maintain stemness [35]. Additionally, tissue-specific factors including epidermal growth factor (EGF), fibroblast growth factors (FGFs), and various small molecule inhibitors are required to direct differentiation along particular lineages [37]. Current challenges in organoid media formulation include batch-to-batch variability of growth factors, high costs of commercial preparations, and the need for tissue-specific optimization [35]. This protocol addresses these challenges by providing standardized, cost-effective media formulations of defined cellular activity for various tissue types, with particular emphasis on applications within organoid biobanking and cryopreservation research contexts.
Essential Signaling Pathways and Growth Factors
Core Signaling Pathways in Organoid Development
The self-organization and differentiation of organoids are governed by the precise regulation of several evolutionarily conserved signaling pathways. The Wnt/β-catenin pathway is fundamental for maintaining stemness and promoting proliferation in many epithelial organoids, particularly in the gastrointestinal tract [35]. This pathway is typically activated in organoid media through the addition of R-spondin proteins, which potentiate Wnt signaling by binding to LGR receptors and protecting Wnt ligands from degradation [35]. The BMP (Bone Morphogenetic Protein) pathway serves as a counterbalance to Wnt signaling, promoting differentiation in many tissue contexts; thus, organoid media commonly include BMP antagonists such as Noggin or Gremlin 1 to inhibit this differentiation signal and maintain the stem cell compartment [35].
The EGF pathway supports proliferation and survival across numerous organoid types, while FGF signaling plays particularly important roles in directing differentiation in lung, liver, and pancreatic organoids [37]. Additionally, Notch signaling often requires modulation through small molecule inhibitors or specific growth factors to control cellular differentiation decisions, especially in neural and intestinal organoids [37]. The precise combination and temporal regulation of these pathways must be tailored to each specific tissue type to successfully recapitulate organ development in vitro.
Growth Factor Production and Quality Control
A significant advancement in organoid media formulation has been the development of recombinant growth factors with defined cellular activities, which overcome the batch-to-batch variability associated with conditioned media preparations [35]. Bacterial expression systems have been successfully employed to produce highly pure recombinant Gremlin 1 and R-spondin 1 with minimal endotoxin contamination at a fraction of the cost of commercial eukaryotic-expressed factors [35]. For R-spondin production, the expression vector is modified to include a solubility-enhancing Avi-tag and co-expresses disulphide isomerase DsbC to ensure correct protein folding, followed by nickel-NTA purification and size exclusion chromatography to remove inactive aggregates [35].
The cellular activity of purified R-spondin is quantified using the WPC50 assay (Wnt potentiation concentration 50%), which typically yields values of approximately 4.0 nM for properly folded protein [35]. Similarly, Gremlin 1 activity is measured by its IC50 for inhibition of BMP2-induced ALP activity in C2C12 cells, with effective preparations showing IC50 values of approximately 6.4 nM [35]. This rigorous quantification of growth factor activity enables the formulation of organoid media with reproducible potencies, which is essential for standardized organoid culture and reliable biobanking applications [35].
Tissue-Specific Media Formulations
Quantitative Comparison of Media Components
Table 1: Essential Growth Factors for Different Organoid Types
| Organoid Type | R-spondin | Noggin/ Gremlin1 | Tissue-Specific Factors | Small Molecule Inhibitors | Key References |
|---|---|---|---|---|---|
| Intestinal | Required (25 nM) | Required (25 nM) | Wnt3a, EGF | A83-01 (TGF-β inhibitor) | [35] |
| Hepatic | Required | Required | FGF7, FGF10, HGF | - | [37] [4] |
| Pancreatic | Required | Required | FGF10, Nicotinamide | A83-01 | [37] [4] |
| Neural | Not required | Required | FGF2, EGF, N2, B27 | SB202190 (p38 inhibitor) | [37] |
| Pulmonary | Required | Required | FGF7, FGF10, BMP4 | A83-01, SB202190 | [37] [4] |
| Mammary | Condition-dependent | Condition-dependent | Heregulin β-1, Prolactin | - | [4] |
Table 2: Bacterial vs. Commercial Growth Factor Comparison
| Parameter | Bacterial R-spondin 1 | Commercial R-spondin 1 | Bacterial Gremlin 1 | Commercial Gremlin 1 |
|---|---|---|---|---|
| Purity | >95% (single band on SDS-PAGE) | Multiple bands (glycosylation variants) | >95% (single band) | Multiple bands |
| Cellular Activity | WPC50 = 4.0 ± 0.53 nM | WPC50 = 1.2 ± 0.69 nM | IC50 = 6.4 ± 0.65 nM | IC50 = 6.0 ± 0.33 nM |
| Endotoxin Level | <0.5 EU/ml (20x below threshold) | Similar to bacterial | 250x below threshold | Variable |
| Production Cost | <£10/L media | >£5,000/L media | <£10/L media | >£3,500/L media |
| Yield | ~2.5 mg (MBP-fusion) or 1 mg (cleaved) per L culture | N/A | ~10 mg per L culture | N/A |
Tissue-Specific Protocol Variations
Intestinal Organoid Media: For both murine and human intestinal organoids, the base medium (Advanced DMEM/F12) must be supplemented with 25 nM of bacterially-derived R-spondin 1 and 25 nM Gremlin 1, alongside additional factors including 50 ng/mL EGF, 1 μg/mL Wnt3a, 10 mM Nicotinamide, 1× N2, 1× B27, and 1.25 mM N-Acetylcysteine [35]. The small molecule inhibitor A83-01 (TGF-β inhibitor) at 500 nM is essential to prevent epithelial-mesenchymal transition [37]. This formulation supports the long-term expansion of intestinal stem cells and the development of crypt-villus structures containing all major intestinal cell types [35].
Hepatic Organoid Media: Liver organoids require a distinct combination of growth factors including FGF7 and FGF10 to promote differentiation toward hepatic lineages [37]. The precise formulation depends on the cell source—adult liver stem cells versus pluripotent stem cell-derived progenitors—with the former requiring higher concentrations of EGF and FGFs for expansion [4]. Recent advances have enabled the long-term culture of genome-stable bipotent stem cells from adult human liver, facilitating the establishment of comprehensive hepatic organoid biobanks [4].
Pancreatic Organoid Media: Pancreatic ductal adenocarcinoma organoids and normal pancreatic organoids have been successfully established using media containing FGF10, Nicotinamide, and the TGF-β inhibitor A83-01 [37]. The specific factor requirements vary significantly between normal and tumor-derived organoids, with cancer organoids often showing growth factor independence for certain pathways [4]. Large-scale biobanking efforts for pancreatic organoids have revealed substantial inter-patient heterogeneity in factor requirements [4].
Neural Organoid Media: Cerebral organoids require different signaling modulation compared to epithelial organoids, with emphasis on FGF2 and EGF for neural progenitor expansion, and the absence of Wnt agonists in most protocols [37]. The p38 inhibitor SB202190 is commonly included to prevent excessive differentiation [37]. The extracellular matrix environment also differs substantially, with neural organoids often being initially embedded in Matrigel but then transferred to suspension culture to allow for complex morphogenesis [23].
Experimental Workflows and Protocols
Growth Factor Production Protocol
Recombinant R-spondin 1 Expression and Purification:
Expression: Transform NEB Shuffle T7 E. coli with pR-spondin 1 vector and plate on selective LB agar. Inoculate a single colony into 50 mL LB with antibiotic and grow overnight at 30°C. Dilute the culture 1:100 into fresh medium and grow until OD600 reaches 0.6. Induce expression with 0.5 mM IPTG and incubate overnight at 16°C [35].
Purification: Harvest cells by centrifugation at 4,000 × g for 20 min. Resuspend pellet in lysis buffer (20 mM Tris-HCl pH 8.0, 500 mM NaCl, 20 mM imidazole) and lyse by sonication. Clarify lysate by centrifugation at 15,000 × g for 30 min. Incubate supernatant with Ni-NTA agarose for 1 h at 4°C. Wash with 10 column volumes of lysis buffer, then elute with elution buffer (20 mM Tris-HCl pH 8.0, 500 mM NaCl, 250 mM imidazole) [35].
In Vitro Refolding: Dialyze eluted protein against refolding buffer (20 mM Tris-HCl pH 8.0, 150 mM NaCl, 1 mM EDTA, 0.5 mM oxidized glutathione, 5 mM reduced glutathione) overnight at 4°C [35].
Size Exclusion Chromatography: Apply refolded protein to Superdex 200 column equilibrated with storage buffer (20 mM Tris-HCl pH 8.0, 150 mM NaCl). Collect fractions corresponding to monomeric MBP-R-spondin 1 (58 kDa). Analyze purity by SDS-PAGE and determine protein concentration by absorbance at 280 nm [35].
Activity Assessment: Determine WPC50 using Wnt reporter cell line. Serial dilute R-spondin preparations and measure Wnt pathway activation after 24 h. Calculate WPC50 as the concentration yielding half-maximal response [35].
Recombinant Gremlin 1 Expression and Purification:
Expression and Inclusion Body Isolation: Transform BL21(DE3) E. coli with His-ΔN-Gremlin 1 vector. Grow and induce as described for R-spondin 1. Harvest cells and resuspend in inclusion body isolation buffer (20 mM Tris-HCl pH 8.0, 150 mM NaCl, 1% Triton X-100). Lyse by sonication and pellet inclusion bodies by centrifugation at 15,000 × g for 20 min [35].
Solubilization and Refolding: Solubilize inclusion bodies in denaturing buffer (6 M GuHCl, 20 mM Tris-HCl pH 8.0, 150 mM NaCl, 1 mM EDTA). Dilute dropwise into refolding buffer (20 mM Tris-HCl pH 8.0, 150 mM NaCl, 1 mM EDTA, 0.5 mM oxidized glutathione, 5 mM reduced glutathione) and stir overnight at 4°C [35].
Purification: Dialyze refolded protein against anion exchange buffer A (20 mM Tris-HCl pH 8.0). Apply to Q-Sepharose column and elute with linear gradient of 0-500 mM NaCl over 20 column volumes. Analyze fractions by SDS-PAGE and pool pure Gremlin 1 fractions [35].
Activity Assessment: Determine IC50 using BMP2-induced ALP assay in C2C12 cells. Pre-treat cells with serial dilutions of Gremlin 1 for 1 h, then stimulate with 8 nM BMP2 for 24 h. Measure ALP activity and calculate IC50 as the concentration yielding 50% inhibition of BMP2 response [35].
Media Preparation and Quality Control
Complete Organoid Media Formulation:
Base Medium Preparation: Start with 500 mL Advanced DMEM/F12 supplemented with 10 mM HEPES, 1× GlutaMAX, and 1× Penicillin-Streptomycin (optional) [35].
Growth Factor Addition: Add purified recombinant growth factors to the following final concentrations: 25 nM R-spondin 1, 25 nM Gremlin 1, 50 ng/mL EGF, 1 μg/mL Wnt3a (conditionally required based on organoid type) [35].
Supplement Addition: Add 1× B27 supplement, 1× N2 supplement, 1.25 mM N-Acetylcysteine, and 10 mM Nicotinamide (for intestinal organoids) [35].
Small Molecule Inhibitors: Add A83-01 to 500 nM final concentration for intestinal, gastric, and pancreatic organoids. Add SB202190 to 10 μM for lung and neural organoids [37].
Sterile Filtration: Filter complete medium through 0.22 μm PES membrane and store at 4°C for up to 2 weeks. For long-term storage, aliquot and freeze at -20°C for up to 3 months [35].
Quality Control Measures:
Performance Validation: Test each new media batch by plating freshly dissociated organoid cells at standardized density (1,000 cells/50 μL Matrigel droplet) and monitoring organoid formation efficiency over 7 days [35].
Morphological Assessment: Compare organoid morphology to reference images after 5-7 days in culture. Intestinal organoids should display clear budded structures with distinct lumen [35].
Viability Staining: Perform live/dead staining using calcein-AM/propidium iodide to confirm viability >85% in established organoids [35].
Differentiation Capacity: For stem cell-derived organoids, validate multilineage differentiation potential through immunohistochemistry for tissue-specific markers after removing niche factors from the media [37].
Diagram 1: Organoid media formulation workflow showing the sequential steps from base medium preparation to quality-controlled final product.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Organoid Media Formulation
| Reagent Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| Base Media | Advanced DMEM/F12 | Nutrient foundation | Provides essential amino acids, vitamins, and salts for cell survival and proliferation [35] |
| Wnt Agonists | R-spondin 1, Wnt3a | Stem cell maintenance | Critical for Lgr5+ stem cell expansion in epithelial organoids; concentration typically 25 nM [35] |
| BMP Antagonists | Noggin, Gremlin 1 | Differentiation inhibition | Prevents BMP-mediated differentiation; used at 25 nM concentration [35] |
| Mitogenic Factors | EGF, FGF7, FGF10 | Proliferation stimulation | Concentration typically 50-100 ng/mL; tissue-specific requirements vary [37] |
| Small Molecule Inhibitors | A83-01, SB202190 | Pathway modulation | A83-01 inhibits TGF-β signaling; SB202190 blocks p38 MAPK pathway [37] |
| Matrix Components | Matrigel, Collagen, Nanomaterials | 3D structural support | Provides physical scaffold; nanomaterials can enhance functionality and reduce necrosis [23] |
| Commercial Kits | IntestiCult, STEMdiff | Standardized protocols | Pre-formulated media systems that reduce batch variability but at higher cost [35] |
Applications in Biobanking and Cryopreservation
The development of optimized, defined media formulations has direct implications for organoid biobanking and cryopreservation—essential technologies for making organoids widely available for research and clinical applications [34]. Patient-derived organoid (PDO) biobanks have been established for numerous cancer types including colorectal, pancreatic, breast, and hepatic cancers, with media optimization being a critical factor in their success [4]. These living biobanks serve as invaluable resources for drug screening, disease modeling, and personalized medicine approaches [38] [4].
In the context of cryopreservation, optimized media formulations play a dual role: they ensure the healthy pre-culture state of organoids before freezing, and they provide the necessary components for recovery post-thaw [39]. The inclusion of nanomaterials in organoid culture systems has shown promise in improving cryopreservation outcomes by serving as heterogeneous nucleation sites that reduce supercooling and inhibit damaging ice crystal formation [23]. Magnetic nanoparticles enable uniform thawing through exposure to external magnetic fields, significantly enhancing post-thaw viability in complex organoid models such as heart organoids [23].
Standardized media formulations are particularly crucial for high-throughput applications in biobanking, where consistency across thousands of samples is essential for meaningful comparative analyses [37]. Automated liquid handling systems combined with defined media enable the reproducible culture and expansion of organoid collections, facilitating large-scale drug screening initiatives [37]. The integration of these technological advances with blockchain-based tracking systems creates a robust infrastructure for the next generation of organoid biobanking, enhancing both traceability and ethical stewardship of patient-derived materials [9].
Diagram 2: Organoid biobanking pipeline showing the complete workflow from tissue acquisition to research applications, highlighting the critical role of optimized media formulations throughout the process.
The optimization of media formulations for different tissue types represents a cornerstone of robust organoid technology, with far-reaching implications for basic research, drug development, and clinical applications. The move toward defined, cost-effective media utilizing recombinant growth factors of precise cellular activity addresses key limitations in scalability and reproducibility that have historically constrained organoid applications [35]. As organoid biobanking continues to expand globally, standardized media protocols will be essential for ensuring comparability across institutions and research initiatives [4].
Future developments in organoid media formulation will likely focus on several key areas: First, the integration of nanomaterials as structural components and delivery vehicles for growth factors promises enhanced control over the organoid microenvironment, potentially reducing central necrosis and improving functional maturation [23]. Second, high-throughput screening approaches for media optimization will enable systematic exploration of factor combinations, potentially revealing novel signaling requirements for challenging tissue types [37]. Finally, the development of xeno-free, clinically compliant media will be essential for translational applications of organoid technology in regenerative medicine and personalized therapy [35].
The combination of optimized media formulations with advanced biobanking infrastructure—including blockchain-based specimen tracking and decentralized research networks—heralds a new era of collaborative, ethically grounded biomedical research [9]. As these technologies mature, standardized organoid media will continue to serve as the fundamental enabler of reproducible, physiologically relevant human tissue models that accelerate our understanding of disease and enhance therapeutic development.
Protocols for Tissue Processing, Freezing, and Thawing
Organoid biobanking has emerged as a cornerstone of modern translational research, providing invaluable resources for drug screening, disease modeling, and personalized medicine. These living biobanks preserve complex three-dimensional cellular structures that recapitulate the histological and functional characteristics of their tissue of origin [34] [4]. The reliability of research outcomes derived from organoid models is fundamentally dependent on the standardized protocols governing their processing, cryopreservation, and revival. Optimization of these protocols ensures the preservation of genetic fidelity, phenotypic stability, and functional integrity of organoids through freeze-thaw cycles, enabling their scalable application across the research community [40] [41]. This application note delineates comprehensive, validated methodologies for the critical processes of tissue processing, cryopreservation, and thawing of patient-derived organoids, with a specific focus on intestinal systems, to enhance reproducibility and success in organoid-based research.
Materials and Methods
Key Reagent Solutions for Organoid Biobanking
Successful organoid biobanking relies on a carefully formulated suite of reagents. The table below catalogues the essential materials and their specific functions within the protocols.
Table 1: Essential Research Reagents for Organoid Processing and Culture
| Reagent Category | Specific Examples | Function and Application |
|---|---|---|
| Basal Medium | Advanced DMEM/F12 with HEPES [42] | Serves as the nutrient foundation for washing solutions and culture media. |
| Extracellular Matrix | Matrigel Matrix [43] [42] | Provides a 3D scaffold that mimics the in vivo stem cell niche for organoid growth. |
| Growth Supplement Media | IntestiCult Organoid Growth Medium [42]; L-WRN Conditioned Medium (Wnt3a, R-spondin, Noggin) [43] [44] | Supplies essential niche signals (ENR: EGF, Noggin, R-spondin) for stem cell maintenance and proliferation. |
| Cryoprotective Agents | Dimethyl sulfoxide (DMSO), Ethylene Glycol [43] [14] | Penetrate cells to prevent ice crystal formation and ensure viability during freezing. |
| Cryopreservation Additives | Methylcellulose, Y27632 (ROCK inhibitor) [14]; Fetal Bovine Serum (FBS) [43] | Enhance cell survival during freezing (MEDY formulation) and thawing by inhibiting apoptosis. |
| Antibiotics | Penicillin-Streptomycin, Gentamicin [43] [42] | Prevent microbial contamination in culture and during tissue transport. |
| Washing & Processing Solutions | Dulbecco's Phosphate Buffered Saline (DPBS); DMEM/F12 with 1% Bovine Serum Albumin (BSA) [42] | Used for tissue transport, crypt isolation, and gentle removal of cryoprotectants post-thaw. |
Quantitative Guidelines for Tissue Handling and Preservation
Decisions regarding tissue preservation directly impact cell viability and organoid formation efficiency. The following table summarizes key quantitative data to guide protocol selection.
Table 2: Comparative Analysis of Tissue Preservation Methods
| Parameter | Short-term Refrigerated Storage | Cryopreservation for Biobanking |
|---|---|---|
| Primary Application | Interim storage when processing is delayed [43] | Long-term storage for living biobanks [43] [40] |
| Recommended Duration | ≤ 6-10 hours [43] | Indefinite (long-term) [40] |
| Standard Protocol | Store tissue at 4°C in DMEM/F12 or RPMI supplemented with antibiotics [43] | Cryopreserve tissue or organoids in freezing medium (e.g., 10% FBS, 10% DMSO in 50% L-WRN medium) [43] |
| Impact on Viability | Higher cell viability compared to cryopreserved samples [43] | Approximately 20-30% reduction in live-cell viability compared to short-term storage [43] |
| Key Consideration | Prompt processing is critical; viability decreases with prolonged storage. | Requires optimized freeze-thaw cycle; essential for creating scalable, distributable biobanks. |
Experimental Protocols
Tissue Procurement and Initial Processing Protocol
The foundation of a successful organoid culture lies in the integrity of the starting material. The following protocol, adaptable for colorectal and other tissues, outlines critical steps from collection to initial processing, which should be completed within approximately 2 hours [43].
1. Sample Collection: Under sterile conditions, collect human colorectal tissue samples immediately following colonoscopy or surgical resection, in accordance with IRB-approved protocols and patient informed consent [43]. 2. Transport: Transfer the tissue to a 15 mL Falcon tube containing 5–10 mL of cold Advanced DMEM/F12 medium supplemented with antibiotics (e.g., penicillin-streptomycin). Prompt handling is imperative to preserve tissue integrity [43]. 3. Preservation Decision Point: * For processing within 6-10 hours: Wash tissues with an antibiotic solution and store at 4°C in an appropriate medium like DMEM/F12 with antibiotics [43]. * For processing delays exceeding 14 hours: Cryopreservation of the tissue is preferable. After an antibiotic wash, cryopreserve the tissue fragments using a validated freezing medium, such as one containing 10% Fetal Bovine Serum (FBS), 10% DMSO, and 50% L-WRN conditioned medium [43]. 4. Crypt Isolation: For intestinal tissues, mince the cleaned tissue and incubate it in a chelating buffer (e.g., DPBS with 2 mM EDTA) to dissociate and isolate crypts. These crypts are then embedded in Matrigel domes for culture initiation [43] [44].
Crypt Isolation and Organoid Establishment Workflow
The following diagram illustrates the core workflow for establishing an organoid culture from intestinal tissue, highlighting key decision points and processes.
Diagram 1: Organoid Establishment Workflow
Cryopreservation Protocol for Organoids
The MEDY cryopreservation protocol has been demonstrated to enhance the survival of complex neural organoids and can be adapted for other organoid types [14]. The principles also apply to the cryopreservation of tissue fragments for later organoid generation [43].
1. Prepare Cryopreservation Solution: Formulate a solution such as MEDY, which contains Methylcellulose, Ethylene glycol, DMSO, and Y27632 [14]. For intestinal organoids, a standard freezing medium can consist of 10% FBS and 10% DMSO in a suitable base like 50% L-WRN conditioned medium [43]. 2. Harvest and Mix: Gently harvest the organoids from the Matrigel dome. Pellet the organoids via gentle centrifugation and resuspend them thoroughly in the pre-cooled cryopreservation solution. 3. Controlled-Rate Freezing: Aliquot the organoid suspension into cryovials. Use a controlled-rate freezing device or an isopropanol-filled "Mr. Frosty" container placed at -80°C to achieve a consistent cooling rate of approximately -1°C per minute. This controlled process is critical to prevent lethal intracellular ice crystal formation. 4. Long-term Storage: After 24 hours, transfer the cryovials to a liquid nitrogen tank for stable long-term storage at -150°C to -180°C [44].
Thawing and Recovery Protocol for Cryopreserved Organoids
A standardized thawing procedure is vital for reviving viable and proliferative organoids. This protocol is critical for utilizing biobanked resources [42].
1. Preparation: * Thaw 120 μL of Matrigel Matrix on ice. * Pre-warm complete organoid growth medium (e.g., IntestiCult) to room temperature. * Prepare a DMEM/F12 washing solution containing 1% BSA and keep it at room temperature. * Place a 24-well tissue culture plate in a 37°C CO₂ incubator to warm [42]. 2. Rapid Thaw and Transfer: * Retrieve the cryovial from liquid nitrogen and immediately place it in a 37°C water bath. Thaw for 2-2.5 minutes, just until the ice crystal disappears. Avoid over-warming. * Wipe the vial with 70% ethanol. Using a pre-wetted pipette tip, add 1 mL of the 1% BSA washing solution to the cryovial and gently pipette to mix. * Immediately transfer the contents to a 15 mL conical tube containing 2 mL of the BSA washing solution [42]. 3. Wash and Plate: * Centrifuge the organoid suspension at 200 x g for 5 minutes at 2-8°C to pellet the organoids and remove the cryoprotectant-containing supernatant. * Carefully aspirate the supernatant, avoiding the pellet. * Resuspend the organoid pellet in 100 μL of complete growth medium. * Add 100 μL of thawed Matrigel, mix gently by pipetting to avoid bubbles, and plate 50 μL drops (domes) into the pre-warmed 24-well plate. * Let the Matrigel solidify for 10-15 minutes in the incubator [42]. 4. Initial Culture and Recovery: * After solidification, add 750 μL of pre-warmed complete growth medium gently to the side of each well. * Perform a full medium change three times per week. * For optimal recovery and experimental consistency, passage the organoids at least two times after thawing before initiating experiments. Organoid growth may be slow in the first passage but typically recovers by the second passage [42].
Discussion
The protocols detailed herein provide a robust framework for the establishment and management of living organoid biobanks. The quantitative data presented in Table 2 underscores a critical trade-off: while short-term storage maximizes viability, cryopreservation is indispensable for creating scalable, distributable resources, despite an expected 20-30% viability cost [43]. The success of these protocols hinges on several pivotal steps. The initial tissue processing speed and the choice of preservation method are primary determinants of organoid formation efficiency [43]. Furthermore, the use of specialized cryopreservation formulations like MEDY and the strict adherence to rapid thawing and gentle cryoprotectant removal are non-negotiable for maintaining organoid viability and functionality post-thaw [14] [42].
A significant challenge remains in standardizing these protocols across different tissues and research laboratories to ensure data reproducibility [41]. Future advancements are likely to focus on the development of chemically defined, xeno-free cryopreservation media and the integration of decentralized biobanking platforms using blockchain technology to enhance traceability and ethical stewardship of patient-derived samples [9]. In conclusion, the meticulous application of these detailed protocols for processing, freezing, and thawing will significantly advance the reliability and translational impact of organoid technology in drug development and personalized medicine.
Patient-derived organoid (PDO) biobanks represent a transformative platform in modern biomedical research, serving as living repositories that closely recapitulate the histological, genetic, and functional features of parental primary tissues [4]. These biobanks provide an essential bridge between traditional two-dimensional cell cultures and complex in vivo environments, enabling more physiologically relevant studies of disease mechanisms and therapeutic responses [4] [22]. The establishment of robust cryopreservation protocols ensures the long-term viability and genetic stability of these biospecimens, creating sustainable resources for the scientific community [45]. Within this framework, organoid biobanks have become indispensable tools for two critical downstream applications: high-throughput drug screening and sophisticated disease modeling. These applications leverage the biological fidelity of organoids to accelerate drug discovery and advance our understanding of human pathophysiology, ultimately supporting the development of personalized medicine approaches [4].
Table 1: Global Distribution of Select Patient-Derived Organoid Biobanks for Translational Research
| System/Body District | Organ | Number of Samples | Country | Primary Translational Applications |
|---|---|---|---|---|
| Digestive | Colorectal | 151 | China | Drug response prediction [4] |
| Digestive | Colorectal | 106 | Germany | High-throughput screening, gene-drug response correlation [4] |
| Digestive | Colorectal, gastroesophagus, bile ducts | 110 | United Kingdom | High-throughput screening (in vitro/in vivo) [4] |
| Digestive | Pancreas | 31 | Switzerland | Disease modeling, high-throughput screening [4] |
| Reproductive | Mammary gland | 168 | The Netherlands | Drug response prediction [4] |
| Reproductive | Ovaries | 76 | United Kingdom | Disease modeling, drug response prediction [4] |
| Urinary | Kidney | 54 | The Netherlands | Disease modeling, drug response prediction [4] |
High-Throughput Drug Screening Using Organoids
Automated Imaging and Analysis Pipelines
The application of organoids in high-throughput screening requires specialized methodologies to overcome the challenges associated with their three-dimensional architecture. Recent technological advances have led to the development of automated pipelines that enable rapid imaging and quantitative analysis of organoids in multi-well plate formats [13]. These systems utilize high-throughput confocal microscopy coupled with sophisticated image analysis software to quantify fluorescent labeling in organoids, dramatically reducing the time and labor requirements for large-scale drug screening campaigns [13]. For instance, researchers have established a 96-well plate-based automated platform that can efficiently characterize organoids at cellular and molecular levels, allowing for the quantification of varying proliferation rates among different donor-derived organoid lines in response to therapeutic compounds or microbial products [13].
A particularly significant innovation in this domain is the emergence of AI-driven analysis platforms that enable multi-scale segmentation and quantification at nuclear, cytoplasmic, and whole-organoid levels [46]. These systems employ convolutional neural networks, such as the DeepStar3D model based on StarDist principles, to perform rapid 3D segmentation of organoid structures under real-world laboratory conditions [46]. The resulting "digitalized organoids" approach provides comprehensive morphological and topological descriptors that can be mined for drug response signatures, offering a user-friendly interface (3DCellScope) that makes this advanced analytical capability accessible to researchers without specialized computational expertise [46].
Applications in Drug Discovery and Development
Organoid biobanks have demonstrated considerable utility across multiple stages of the drug discovery and development pipeline. In the context of cancer research, PDO biobanks enable high-throughput compound screening on models that preserve the genetic heterogeneity of original tumors, facilitating the identification of candidate therapeutics with increased clinical predictive value [4]. The ability to maintain patient-specific characteristics in these models allows for the correlation of drug responses with genetic mutations, supporting the discovery of biomarker-driven treatment strategies [4].
Cardiac organoids have emerged as valuable tools for cardiotoxicity screening, addressing a major cause of drug attrition during clinical development [47]. These simplified, miniature three-dimensional cellular model systems more accurately mimic the biological characteristics and functions of the human heart compared to conventional two-dimensional models, providing enhanced predictive capability for adverse cardiac effects [47]. Similarly, brain organoids are being utilized for neurotoxicity assessment and the evaluation of therapeutic candidates for neurological disorders, offering human-relevant models that recapitulate aspects of developing human brain architecture [48].
Table 2: Key Experimental Protocols for High-Throughput Drug Screening Using Organoids
| Protocol Step | Methodological Details | Application Examples |
|---|---|---|
| Organoid Culture in Multi-well Plates | 2D monolayer plating in 96-well plates with collagen IV coating; seeding density optimization [13] | Drug screening, toxicity testing, intestinal barrier integrity studies [13] |
| High-Content Imaging | Automated confocal microscopy; 3D image acquisition; multiple optical sections [13] [46] | Phenotypic profiling; quantification of cell proliferation, death, or differentiation [13] |
| AI-Based 3D Analysis | Multi-level segmentation (nuclear, cytoplasmic, organoid); DeepStar3D convolutional neural network; topology descriptors [46] | Morphological signature extraction; tissue patterning detection; mechanical constraint assessment [46] |
| Response Quantification | Fluorescence intensity measurement; nuclear and cytoplasmic marker quantification; correlation with flow cytometry validation [13] | Donor-specific response variability; cell-specific drug effects; mechanism of action studies [13] |
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Organoid-Based Screening
| Reagent/Material | Function | Examples & Specifications |
|---|---|---|
| Extracellular Matrix Substitutes | Provides 3D scaffolding for organoid growth and development | Matrigel; collagen IV; nanocellulose hydrogels; synthetic peptide hydrogels [45] [47] |
| Staged Culture Media | Supports organoid development through specific differentiation pathways | Media containing Wnt-3a, EGF, FGF, R-spondin, noggin, B-27 supplement [47] |
| Cell Staining Reagents | Enables visualization and quantification of cellular components | DAPI (nuclear); phalloidin (actin); immunolabeling antibodies; viability dyes [13] [46] |
| Functional Nanomaterials | Enhances organoid culture and maturation | Mesoporous silica nanoparticles (growth factor delivery); magnetic nanoparticles (3D suspension culture) [45] |
Disease Modeling with Organoids
Neurodevelopmental and Neurodegenerative Disorders
Brain organoids have emerged as powerful tools for modeling various neurological conditions, ranging from neurodevelopmental diseases to neurodegenerative disorders and brain cancers [48]. These self-organized, three-dimensional aggregates derived from human pluripotent stem cells exhibit cell types and cellular architectures resembling those of the developing human brain, providing unprecedented opportunities to study human-specific aspects of brain disorders [48]. The application of region-specific brain organoids allows for the investigation of disorders affecting particular brain areas, while assembloid approaches enable the modeling of interactions between different brain regions, offering insights into circuit-level abnormalities in neuropsychiatric conditions [48].
The use of brain organoids in disease modeling extends to pediatric brain cancers, where they serve as faithful models for studying tumor biology and treatment response [48]. Cancer neuroscience applications utilizing tumoroids derived from patient samples provide platforms for investigating neuron-cancer interactions and screening for therapeutics that target these relationships [48]. The ability to generate these models from human stem cells rather than relying solely on animal models has accelerated research into human-specific aspects of neurological disease pathogenesis.
Cardiovascular Diseases
Cardiac organoids represent increasingly sophisticated models for studying cardiovascular diseases, which remain the leading cause of mortality worldwide [47]. These simplified, miniature three-dimensional cellular systems are grown from progenitor or stem cells and mimic significant biological characteristics and functions of the normal cardiac system more accurately than conventional two-dimensional models [47]. Through self-organization strategies following developmental induction conditions, cardiac organoids achieve higher metabolic similarities and physiological relevance, making them valuable platforms for studying human cardiac biology and pathophysiology [47].
Current cardiac organoid models successfully incorporate multiple relevant cell types, including cardiomyocytes, endothelial cells, and fibroblasts, recapitulating aspects of the native cardiac microenvironment [47]. These models are being utilized to study cardiac development, model disease processes such as myocardial infarction and cardiomyopathy, and evaluate drug efficacy and toxicity [47]. Despite progress in cardiac organoid development lagging behind some other organ systems due to the heart's intricate cellular structure, recent advances in biofabrication techniques are accelerating their sophistication and application.
Cancer Modeling and Personalized Oncology
Patient-derived cancer organoids have revolutionized approaches to modeling human malignancies, with biobanks established for numerous cancer types including colorectal, pancreatic, breast, ovarian, and prostate cancers [4]. These models preserve the genetic mutations and cellular heterogeneity of the original tumors, providing valuable platforms for studying tumor biology and advancing personalized medicine approaches [4]. The ability to bank matched healthy tissue organoids alongside tumor models further enhances their utility by providing patient-specific controls for drug screening and toxicity assessment [4].
The translational applications of cancer organoid biobanks extend to functional precision medicine, where they enable the correlation of drug responses with genomic data to identify predictive biomarkers [4]. Large-scale drug screening on genomically characterized organoid collections facilitates the identification of gene-drug response relationships, supporting the development of stratified medicine approaches [4]. Additionally, these biobanks serve as resources for studying tumor evolution, drug resistance mechanisms, and the functional validation of newly discovered genetic alterations in cancer.
Integrated Protocols for Disease Modeling and Drug Screening
Protocol: High-Throughput Drug Screening Using Intestinal Organoids
Background: This protocol describes an automated pipeline for rapid imaging and quantitative analysis of human intestinal organoids (HIOs) in 96-well plate formats, enabling high-throughput drug screening applications [13].
Materials:
- Collagen IV-coated 96-well plates
- Single-cell suspension of intestinal organoids
- L-WRN conditioned medium (advanced DMEM/F-12 with supplements)
- Compound library of interest
- Fixation and permeabilization reagents
- Fluorescent antibodies or dyes (e.g., proliferation markers, cell type-specific markers)
- High-throughput confocal microscope
- Image analysis software (e.g., 3DCellScope [46])
Methodology:
- Plate Preparation: Coat inner wells of 96-well plates with collagen IV solution (1:30 dilution in DI water) for 90 minutes at 37°C, then remove solution [13].
- Organoid Dissociation: Harvest HIOs from Matrigel using ice-cold 0.5M EDTA in PBS. Pellet organoids, resuspend in 0.05% trypsin/0.5mM EDTA, and incubate for 5 minutes at 37°C [13].
- Single-Cell Suspension: Inactivate trypsin with complete medium containing 10% FBS. Vigorously pipette to dissociate organoids, then pass through a 40-μm cell strainer [13].
- Cell Seeding: Centrifuge cell suspension, resuspend in L-WRN conditioned medium, count cells, and seed at optimized density in collagen-coated wells [13].
- Compound Treatment: After 24-48 hours of culture, add compound library using automated liquid handling systems. Include appropriate controls (DMSO vehicle, positive/negative controls).
- Endpoint Processing: At experimental endpoint, fix cells, permeabilize, and stain with fluorescent markers (e.g., proliferation markers, apoptosis markers, cell type-specific antibodies) [13].
- High-Content Imaging: Image plates using high-throughput confocal microscope, acquiring multiple optical sections to capture 3D structure [13] [46].
- Quantitative Analysis: Process images using AI-based segmentation software (e.g., 3DCellScope) to extract quantitative parameters including cell number, viability, proliferation, and differentiation markers [46].
Validation: Correlate results with traditional methods such as flow cytometry to ensure pipeline accuracy [13].
Protocol: Disease Modeling Using Brain Organoids
Background: This protocol outlines the generation and application of brain organoids for modeling neurodevelopmental and neurodegenerative diseases, with specific modifications for region-specific organoids and assembloids [48].
Materials:
- Human pluripotent stem cells (embryonic stem cells or induced pluripotent stem cells)
- Neural induction medium
- Matrigel or synthetic ECM alternatives
- Region-patterning small molecules (e.g., SMAD inhibitors, WNT agonists/antagonists)
- Differentiation factors (e.g., BDNF, GDNF, cAMP)
- Immunostaining reagents for neural markers
Methodology:
- Neural Induction: Culture pluripotent stem cells in neural induction medium containing dual SMAD inhibitors to promote neural lineage commitment [48].
- 3D Culture Setup: Embed neural progenitor clusters in Matrigel droplets or alternative ECM supports to provide 3D scaffolding [48] [45].
- Region-Specific Patterning: For region-specific organoids, add patterning molecules during critical developmental windows (e.g., SHH for ventral forebrain, FGF8 for midbrain) [48].
- Extended Maturation: Culture organoids in spinning bioreactors or orbital shakers to enhance nutrient/waste exchange during extended maturation periods (up to several months) [48].
- Assembloid Formation: For circuit-level modeling, fuse region-specific organoids to create assembloids that model interactions between brain areas [48].
- Disease Modeling Applications:
- Neurodevelopmental Disorders: Model genetic mutations by starting with patient-derived iPSCs or introducing mutations via CRISPR editing [48].
- Neurodegenerative Diseases: Extend maturation periods to observe age-related pathology; consider introducing disease-relevant stressors [48].
- Brain Cancer Modeling: Incorporate patient-derived tumor cells or introduce oncogenic mutations to study tumor biology [48].
- Analysis: Utilize high-content imaging and AI-based segmentation to quantify structural and functional phenotypes [46].
Organoid biobanks represent a paradigm shift in biomedical research, providing robust platforms for high-throughput drug screening and disease modeling that bridge the gap between traditional cell culture and clinical studies. The integration of advanced technologies such as AI-driven image analysis, nanomaterials, and automated culture systems continues to enhance the scalability, reproducibility, and physiological relevance of these models [45] [46]. As these technologies mature, organoid biobanks are poised to become central components of drug discovery pipelines and personalized medicine approaches, potentially reducing the high failure rates of candidate therapeutics in clinical trials [47] [22].
Future developments in the field will likely focus on enhancing organoid complexity through improved vascularization, immune component integration, and the creation of multi-organ systems that better recapitulate systemic physiology [22]. Additionally, the integration of blockchain technology for decentralized biobanking may address challenges related to sample tracking, data sharing, and ethical governance, further enhancing the utility and accessibility of organoid resources for the global research community [9]. As these advancements converge, organoid biobanks will continue to transform our approach to understanding disease mechanisms and developing effective therapeutics.
Overcoming Technical Hurdles: Strategies for Enhanced Viability and Functionality
The establishment of reliable, living biobanks is a cornerstone for advancing organoid-based research in disease modeling, drug screening, and regenerative medicine [49]. Cryopreservation, which enables the long-term storage of these complex three-dimensional microtissues, is therefore not merely a storage formality but a critical determinant of experimental reproducibility and clinical translation [23] [10]. However, the path to effective cryopreservation is fraught with the twin challenges of cryoinjury: ice crystal formation and cryoprotectant (CPA) toxicity [50] [49]. These interlinked phenomena can compromise cellular viability, disrupt intricate cell-cell connections, and degrade the functional integrity of organoids, ultimately nullifying their value as physiological models [51] [49]. This document details the underlying mechanisms of these cryoinjuries and presents validated protocols and novel materials designed to mitigate them, providing a framework for enhancing the fidelity of organoid biobanking.
Mechanisms of Cryoinjury
Ice Crystal Formation and Growth
The phase change of water to ice is the primary source of mechanical damage during cryopreservation. The injury manifests differently depending on the cooling rate [50].
- Slow Freezing: At slow cooling rates, ice forms initially in the extracellular space. This extracellular ice increases the solute concentration outside the cell, creating an osmotic gradient that draws water out of the cell, leading to severe cellular dehydration and shrinkage. While some dehydration is beneficial, excessive shrinkage can cause irreversible damage to the cell membrane and internal structures [50].
- Fast Freezing (Intracellular Ice): With rapid cooling, water within the cell does not have sufficient time to efflux and instead freezes in place, forming intracellular ice crystals. These crystals are particularly lethal, as they can physically pierce and disrupt organelles and the plasma membrane, leading to immediate cell death [50].
- Ice Recrystallization during Thawing: The warming process presents its own hazard. As the temperature rises through the range of -15°C to -60°C, small, unstable ice crystals can melt and refreeze into larger, more damaging crystals. This process of recrystallization exacerbates mechanical injuries incurred during the freezing process [50].
Cryoprotectant Toxicity
Cryoprotectants (CPAs) are used to mitigate ice formation, but they introduce a second major challenge: cytotoxicity. The toxicity is influenced by several factors [52]:
- Chemical Nature and Concentration: Penetrating CPAs like dimethyl sulfoxide (DMSO) and ethylene glycol are small molecules that enter cells. While effective at suppressing ice formation, they can disrupt cellular metabolism, protein function, and the epigenetic landscape at high concentrations. The toxicity is often concentration- and time-dependent, making the exposure duration during CPA loading and unloading a critical parameter [50] [52].
- Osmotic Stress: The introduction and removal of CPAs create significant osmotic shifts. Cells first shrink during CPA addition as water leaves, and then swell as CPAs and water re-enter. If not carefully controlled, this "shrink-swell response" can rupture cell membranes [49] [52].
- Lipophilicity and Hydrogen Bonding: The lipophilicity of a CPA correlates with its ability to penetrate cell membranes and potentially destabilize lipid bilayers. Furthermore, CPAs that form strong hydrogen bonds can disrupt the hydration shells essential for macromolecular function [52].
Table 1: Key Challenges in Organoid Cryopreservation
| Challenge | Mechanism of Injury | Consequence for Organoids |
|---|---|---|
| Extracellular Ice | Osmotic dehydration and cell shrinkage [50] | Loss of structural integrity, dissociation of cell-cell contacts [49] |
| Intracellular Ice | Mechanical piercing of membranes and organelles [50] | Immediate cell death, destruction of 3D microarchitecture [50] |
| Ice Recrystallization | Growth of ice crystals during thawing [50] | Aggravation of mechanical damage from the freezing phase [50] |
| CPA Toxicity | Disruption of metabolism, protein function, and osmotic balance [50] [52] | Reduced post-thaw viability and functionality, altered differentiation potential [51] |
| Inadequate CPA Penetration | Failure to protect inner cell layers [51] | Central necrosis, loss of specific cell types (e.g., podocytes) [51] |
Advanced Strategies and Materials to Mitigate Cryoinjury
Novel Cryoprotectant Formulations
Innovative CPA cocktails aim to reduce reliance on high concentrations of traditional, toxic agents like DMSO.
- Natural and Bioinspired CPAs: Antifreeze proteins (AFPs) and their synthetic mimics are emerging as powerful tools. They function by binding to ice crystal surfaces, inhibiting growth and recrystallization, thereby providing protection at much lower concentrations [50] [49]. Deep eutectic solvents, derived from natural sources, are also being explored as low-toxicity alternatives for vitrification [49].
- Combination Vitrification Solutions: Protocols for neural organoids have demonstrated success with a cocktail termed MEDY, which combines Methylcellulose, Ethylene glycol, DMSO, and Y27632 (a Rho kinase inhibitor) [14]. This formulation leverages the synergistic effects of different CPAs and an apoptosis inhibitor, allowing for a reduction in the required concentration of any single toxic agent.
Nanomaterial and Hydrogel-Based Interventions
Nanomaterials offer a physical means to control the ice formation process and improve warming efficiency.
- Ice-Nucleating and Inhibiting Materials: Materials like mesoporous silica nanoparticles and graphene oxide can act as heterogeneous nucleation sites. This raises the ice nucleation temperature, reducing the degree of supercooling and promoting the formation of numerous small, less damaging ice crystals instead of large, destructive ones [23]. Conversely, other nanomaterials can inhibit ice recrystallization during thawing [23].
- Magnetic Nanoparticles for Nanowarming: A groundbreaking approach involves incorporating magnetic nanoparticles (e.g., Fe₃O₄) into the organoid or its surrounding medium. When subjected to an alternating magnetic field during thawing, these nanoparticles generate heat uniformly and rapidly, enabling ultra-fast "nanowarming." This method avoids the destructive ice recrystallization phase associated with conventional slow warming, significantly improving survival rates in complex organoids like cardiac models [23] [49].
- Hydrogel Microencapsulation: Encapsulating organoids in hydrogel matrices such as alginate provides a physical buffer against osmotic stress and ice crystal penetration. This technique confines the organoid in a protective microenvironment, enhancing its tolerance to the cryopreservation process [49].
Protocol-Driven Optimizations
The method of CPA delivery is as crucial as the formula itself.
- Microfluidic CPA Loading: Microfluidic systems enable precise, graded introduction and removal of CPAs. This technology minimizes the osmotic shock and reduces the duration of exposure to toxic CPA concentrations, leading to higher viability and better-preserved function [49].
- Optimized Equilibration: Simple adjustments, such as allowing sufficient equilibration time for CPAs to penetrate deeper cell layers, can be highly effective. For kidney organoids, extending the equilibration time beyond 0 minutes was critical for protecting internal podocyte structures [51].
Experimental Protocols
Protocol: Vitrification of Kidney Organoids
This protocol, adapted from Mashouf et al., demonstrated superior preservation of podocytes and tubular structures compared to slow-freezing methods [51].
1. Reagents:
- Vitrification Solution V1: 20% DMSO, 20% Ethylene Glycol, and sucrose in base medium [51].
- Base medium: Advanced RPMI with GlutaMAX.
- Sucrose solutions (for thawing and washing) at 1.0 M and 0.5 M in base medium.
- Culture medium.
2. Procedure:
- Equilibration: Transfer day-35 kidney organoids into Vitrification Solution V1 at room temperature for 15 minutes.
- Cooling: Quickly place the organoids on a pre-cooled metal surface or directly submerge them in liquid nitrogen to achieve vitrification (ultra-rapid cooling into a glassy state). Store in liquid nitrogen.
- Thawing: Rapidly warm organoids by transferring them from liquid nitrogen into a 37°C water bath for 60-90 seconds.
- CPA Removal & Washing:
- Immediately transfer organoids to 1.0 M sucrose solution at room temperature for 10 minutes.
- Transfer to 0.5 M sucrose solution for a further 10 minutes.
- Wash twice in culture medium.
- Recovery Culture: Plate organoids in culture medium and monitor recovery over 7 days.
3. Key Quantitative Outcome: This protocol achieved a post-thaw viability of 91%, significantly higher than slow-freezing methods, and successfully preserved both PODXL+ podocytes and LTL+ tubules [51].
Protocol: Slow Freezing with MEDY for Cortical Organoids
This protocol is designed for the cryopreservation of sensitive neural tissues [14].
1. Reagents:
- MEDY Cryopreservation Solution: Contains Methylcellulose, Ethylene Glycol, DMSO, and Y27632 [14].
- Sucrose solutions (for step-down dilution).
2. Procedure:
- Preparation: Incubate cortical organoids in MEDY solution. The methylcellulose increases viscosity, which can help reduce ice crystal growth.
- Cooling: Use a controlled-rate freezer or a specialized freezing container to achieve a slow, consistent cooling rate of approximately -1°C/min.
- Storage: Transfer to liquid nitrogen for long-term storage.
- Thawing & Washing: Rapidly thaw and perform a step-wise dilution to remove CPAs, using descending concentrations of sucrose.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Organoid Cryopreservation
| Reagent / Material | Function / Purpose | Example Usage |
|---|---|---|
| DMSO (Dimethyl sulfoxide) | Penetrating CPA; suppresses ice formation [52] | Common component of slow-freezing and vitrification solutions (e.g., 10% for slow freeze) [51] |
| Ethylene Glycol | Penetrating CPA; often used in combination with DMSO for vitrification [51] | Key component of high-efficiency vitrification cocktails (e.g., V1 solution) [51] |
| Sucrose | Non-penetrating osmolyte; reduces osmotic shock during CPA removal [51] | Used in thawing solutions (e.g., 1.0 M and 0.5 M) to draw out CPAs gradually [51] |
| Y27632 (Rho kinase inhibitor) | Inhibits apoptosis; improves survival of dissociated cells and post-thaw viability [14] | Added to cryopreservation and/or recovery media (e.g., MEDY protocol) [14] |
| Antifreeze Proteins (AFPs) & Mimics | Biological ice blockers; inhibit ice recrystallization [50] | Added to CPA solutions to reduce mechanical injury at low concentrations |
| Magnetic Nanoparticles (Fe₃O₄) | Enables uniform, rapid nanowarming to prevent recrystallization [23] [49] | Incorporated into organoids or matrix prior to freezing; thawed via alternating magnetic field |
| Hydrogels (e.g., Alginate) | Microencapsulation scaffold; buffers against osmotic and mechanical stress [49] | Organoids are encapsulated prior to the cryopreservation process |
Workflow and Pathway Visualizations
Pathway for Mitigating Organoid Cryoinjury
The following diagram summarizes the strategic approach to addressing the two primary sources of cryoinjury in organoids.
Experimental Workflow for Kidney Organoid Vitrification
This workflow outlines the specific steps for the successful vitrification of kidney organoids, highlighting critical steps for preserving sensitive structures.
The successful cryopreservation of organoids for biobanking hinges on a multifaceted attack on the dual problems of ice damage and CPA toxicity. Relying solely on traditional methods and CPAs like DMSO is insufficient for preserving complex, multicellular structures. The integration of advanced strategies—including novel nanomaterial-assisted ice control, optimized vitrification protocols, and microfluidic technology for CPA handling—provides a robust path forward. By adopting these sophisticated approaches, researchers can transform organoid biobanking from a bottleneck into a reliable, scalable resource, thereby unlocking the full potential of organoid technology in personalized medicine and drug development.
Novel Cryoprotectants and Penetration Enhancement Techniques
The advancement of organoid biobanking is fundamentally constrained by the inherent limitations of conventional cryopreservation methods. Traditional cryoprotectants (CPAs), such as dimethyl sulfoxide (DMSO), are often cytotoxic and struggle to penetrate the complex three-dimensional structures of organoids, leading to ice crystal formation, osmotic stress, and compromised post-thaw viability [23] [53]. For organoid technology to achieve its full potential in drug screening and personalized medicine, it must transition into an "off-the-shelf" resource, a goal that hinges on the development of novel, effective preservation strategies [53]. This Application Note details advanced cryoprotectants and innovative techniques designed to overcome these barriers, ensuring the high-fidelity, long-term storage of living organoids for translational research.
Novel Cryoprotectant Agents (CPAs)
Moving beyond traditional small-molecule CPAs, recent research has focused on developing agents that offer superior ice-recrystallization inhibition and reduced toxicity. The table below summarizes key categories of novel CPAs.
Table 1: Categories and Properties of Novel Cryoprotectants
| Category | Key Examples | Mechanism of Action | Reported Advantages | Organoid Models Demonstrated |
|---|---|---|---|---|
| Natural Antifreeze Proteins (AFPs) | Antifreeze Proteins (AFPs) [53] | Adsorb to specific ice crystal planes, inhibiting growth and recrystallization. | Reduced ice-crystal damage; Can be used at lower concentrations. | Glioblastoma organoids, Endometrial organoids [53] |
| Deep Eutectic Solvents (DES) | Not Specified (Natural derivatives) [53] | Form a glassy state (vitrification) with less toxicity; disrupts ice formation. | Low toxicity; Biocompatible; "Green" alternative. | Under investigation for organoids [53] |
| Nanomaterial-Based Agents | Graphene Oxide (GO) [23] | Serves as a heterogeneous nucleation site, reducing supercooling. | Inhibits ice crystal formation. | Cardiac organoids [23] |
| Non-Toxic Permeating Agents | Alternatives to DMSO (e.g., specific polymers) [54] | Cross cell membranes to depress freezing point, with reduced cellular toxicity. | Lower cytotoxicity; Improved post-thaw function. | Ovarian tissue, Testicular tissue [54] |
Penetration Enhancement Techniques
The multicellular architecture of organoids presents a significant diffusion barrier. The following techniques are designed to enhance CPA permeation uniformly throughout the tissue.
Microfluidic-Based Loading
Microfluidic systems allow for precise, controlled delivery of CPAs, drastically reducing osmotic shock and exposure time.
- Principle: Organoids or tissue samples are perfused with gradually increasing concentrations of CPAs within micro-channels, enabling a smooth osmotic transition [53].
- Key Benefit: This method mitigates the toxic and osmotic stresses typically associated with manual, bulk CPA loading, leading to significantly higher viability [53].
Hydrogel Microencapsulation
This technique provides a protective barrier that buffers against mechanical stress during freezing and thawing.
- Principle: Individual organoids are encapsulated within a biocompatible hydrogel matrix (e.g., alginate, synthetic polymers) [53].
- Key Benefit: The hydrogel acts as a physical shield, minimizing damage from ice crystals and osmotic swelling/shrinkage, thereby preserving intercellular connections and structural integrity [53].
Nanoparticle-Assisted Strategies
Functional nanomaterials can be employed to improve both warming efficiency and CPA delivery.
- Principle: Magnetic nanoparticles (MNPs), such as Fe₃O₄, are introduced into the organoid or its surrounding matrix. During thawing, exposure to an alternating magnetic field induces uniform and rapid "nanowarming" [23] [53].
- Key Benefit: Prevents the lethal ice recrystallization that occurs during slow, conventional thawing. This method has demonstrated enhanced survival in complex organoid models like cardiac organoids [23] [53].
Integrated Experimental Workflow
The following diagram illustrates a generalized workflow for applying novel cryoprotectants and penetration techniques in organoid cryopreservation.
Detailed Protocol: Cryopreservation of Patient-Derived Organoids Using Advanced CPAs and Microfluidic Loading
This protocol outlines the steps for cryopreserving patient-derived tumor organoids (PDOs) with a novel CPA cocktail and a microfluidic penetration enhancement system [10] [53].
Materials and Reagents
Table 2: Essential Research Reagents and Materials
| Item | Function/Description | Example Product/Catalog |
|---|---|---|
| Novel CPA Cocktail | A mixture of low-toxicity permeating CPAs and natural ice-binding agents. | Custom formulation (e.g., combining a low-toxicity permeating agent with Antifreeze Proteins) [53] [54] |
| Microfluidic Perfusion Device | For controlled, gradual introduction of CPAs to minimize osmotic shock. | Custom or commercial microfluidic chip [53] |
| Hydrogel for Encapsulation | A biocompatible matrix for protecting organoids from mechanical cryoinjury. | Alginate or PEG-based hydrogel [53] |
| Magnetic Nanoparticles (MNPs) | For uniform and rapid nanowarming during the thawing process. | Fe₃O₄ nanoparticles [23] [53] |
| Controlled-Rate Freezer | To ensure a consistent, optimal cooling rate (typically -1°C/min). | "Mr. Frosty", "CoolCell", or programmable freezer [55] [56] |
| Liquid Nitrogen Storage Tank | For long-term storage at or below -135°C. | N/A |
| Serum-Free Freezing Medium | A chemically defined base medium for cryopreservation. | Gibco Synth-a-Freeze [55] |
Pre-Freezing Procedure
- Organoid Preparation: Harvest organoids in the log phase of growth, ensuring high viability (>90%) [56]. Characterize and confirm the absence of microbial contamination prior to freezing.
- CPA Cocktail Preparation: Prepare the novel CPA cocktail on ice. A representative formulation may include:
- Base Medium: 80% serum-free cryopreservation medium (e.g., CryoStor CS10) [56].
- Novel Permeating Agent: 10% of a low-toxicity alternative to DMSO.
- Natural AFP: 10% (v/v) of a purified Antifreeze Protein solution.
- Microfluidic CPA Loading:
- Load the organoid suspension into the inlet reservoir of the microfluidic device.
- Perfuse with a stepwise gradient of the novel CPA cocktail (e.g., 25%, 50%, 75%, 100%) at a controlled flow rate, allowing 5-10 minutes equilibration per step [53].
- Collect the CPA-loaded organoids from the outlet.
- Hydrogel Encapsulation (Optional but Recommended):
- Mix the CPA-loaded organoids with a sterile hydrogel precursor solution.
- Form microdroplets using a droplet generator or manual pipetting into a crosslinking solution (e.g., CaCl₂ for alginate) [53].
- Incubate to form solid microbeads, each containing one or several organoids.
- Aliquoting and Sealing: Aliquot the organoid suspension (encapsulated or not) into pre-chilled, labeled cryogenic vials. Seal vials tightly.
Freezing and Storage
- Controlled-Rate Freezing: Place the cryovials into a controlled-rate freezing apparatus and initiate the following program:
- Long-Term Storage: Immediately transfer the vials to the vapor phase of a liquid nitrogen storage tank (-135°C to -196°C) for long-term preservation [55] [56].
Thawing and Assessment
- Nanowarming: For MNP-containing samples, thaw rapidly using an alternating magnetic field setup optimized for uniform Joule heating [23] [53]. For standard samples, thaw rapidly in a 37°C water bath with gentle agitation for approximately 2 minutes.
- CPA Removal: Immediately after ice dissolution, transfer the organoids to a pre-warmed culture medium. Use a stepwise dilution (e.g., 1:1, 1:3) to gradually remove CPAs and minimize osmotic stress.
- Viability and Functionality Assessment:
- Quantify viability using Trypan Blue exclusion or a fluorescent live/dead assay [55] [56].
- Assess recovery by monitoring re-attachment to matrix and re-initiation of growth.
- Validate functionality through tissue-specific assays (e.g., barrier integrity for gut organoids, contractility for cardiac organoids) and genomic stability checks (e.g., WGS, RNA-seq) to confirm retention of original tumor characteristics [4] [10].
Optimizing Cooling and Warming Rates for Complex Structures
The emergence of complex three-dimensional (3D) biological models, particularly organoids, has revolutionized biomedical research, disease modeling, and drug development. These structures recapitulate the architectural and functional complexity of native organs, providing invaluable platforms for scientific discovery and personalized medicine [23] [57]. However, their widespread application is constrained by significant challenges in long-term storage and distribution. Cryopreservation has emerged as a critical strategy to overcome these limitations, enabling the establishment of next-generation living biobanks [57].
The core challenge in cryopreserving organoids lies in their intricate multicellular architectures and relatively large size, which impede uniform cooling and warming throughout the tissue. Unlike conventional two-dimensional cell cultures, organoids are highly vulnerable to cryoinjury during freeze-thaw cycles, primarily due to ice crystal formation, osmotic stress, and cryoprotectant agent (CPA) toxicity [57] [58]. The optimization of cooling and warming rates is therefore paramount to minimize these damages, maintain structural integrity, and ensure post-thaw viability and functionality.
This application note provides a comprehensive framework for optimizing cooling and warming protocols for complex 3D structures, with a specific focus on organoid biobanking. We present quantitative data, detailed methodologies, and strategic visualizations to guide researchers in developing robust and reproducible cryopreservation strategies.
Quantitative Analysis of Cryopreservation Parameters
Comparative Performance of Cryopreservation Media
The choice of cryopreservation medium significantly impacts cell recovery and viability. The table below summarizes the performance of different media based on studies with human mononuclear cells (MNCs) and fibroblasts, which provide foundational principles applicable to organoid systems.
Table 1: Performance of Different Cryopreservation Media
| Cryopreservation Medium | Cell Recovery (%) | Viability (%) | Key Findings |
|---|---|---|---|
| 90% FBS + 10% DMSO | 80.9% [74.8–87.6] | 71.5% [68.3–78.7] (Trypan Blue); 92.6% [91.3–94.3] (Flow Cytometry) | High recovery and viability; optimal for fibroblasts; maintains collagen-I and Ki67 expression [59] [60]. |
| CryoStor CS10 | 78.0% [71.5–79.3] | 70.1% [64.4–74.8] (Trypan Blue); 94.7% [92.6–96.4] (Flow Cytometry) | Superior viability post-thaw; best preserves relative distribution of leukocyte populations [60]. |
| 70% RPMI/20% FBS/10% DMSO | 72.5% [65.9–76.7] | 63.7% [59.8–70.2] (Trypan Blue); 90.8% [87.6–92.7] (Flow Cytometry) | Moderate performance [60]. |
| Synth-a-Freeze | 68.4% [63.1–73.9] | 62.4% [55.9–68.9] (Trypan Blue); 88.4% [84.1–91.3] (Flow Cytometry) | Lower recovery and viability compared to other media [60]. |
| HPL + 10% DMSO | Not specified | >80% (Fibroblasts) | Maintains fibroblast viability but lower than FBS + DMSO for long-term storage [59]. |
Impact of Storage and Technical Parameters
Technical parameters beyond the medium formulation, such as storage duration and revival method, are critical for success.
Table 2: Impact of Storage Duration and Revival Methods on Cell Attachment
| Parameter | Condition | Performance / Outcome |
|---|---|---|
| Storage Duration | 0-6 months | Highest number of vials with optimal cell attachment [59]. |
| >24 months | Decreased performance and cell attachment [59]. | |
| Revival Method | Direct seeding (no centrifugation) | Higher cell attachment post-revival for some cell types [59]. |
| Indirect seeding (with centrifugation) | Significantly higher Ki67 expression in fibroblasts cryopreserved for 3 months [59]. | |
| Storage Location | Vapor phase of cryo tank (non-immersed) | Superior cell attachment compared to liquid phase [59]. |
Experimental Protocols for Organoid Cryopreservation
Protocol: Slow Freezing Cryopreservation of Human Intestinal Organoids
This protocol, adapted from a method for whole human intestine, is designed for preserving epithelial and immune cells for subsequent organoid generation and autologous co-culture [61] [62].
Key features: Facilitates analysis of region-specific characteristics and generation of a versatile biobank.
Materials and Reagents:
- Biological Material: Human intestinal tissue (e.g., from Novobiosis, I3PT biobank).
- Cryopreservation Medium: CryoStor CS10 or a custom buffer (see Recipes).
- Other Reagents: Sucrose, DMSO USP grade, Ethylene glycol, Trehalose, RNA later stabilization solution, Custodiol, DTT.
Solutions:
- Cryopreservation Buffer Recipe: 28.75% (v/v) Ethylene Glycol, 10% (w/v) Sucrose, 10% (v/v) CryoSure-DMSO, 1.5% (w/v) Trehalose in Custodiol solution.
Procedure:
- Tissue Processing: Dissect whole human intestine and separate into region-specific biopsies (e.g., duodenum, jejunum, ileum).
- Cryopreservation: Incubate tissue biopsies in cryopreservation buffer for 40 minutes at 4°C.
- Cooling: Transfer samples to a Mr. Frosty Freezing Container or CoolCell device. Place the container at -80°C for a minimum of 4 hours to ensure a consistent cooling rate of approximately -1°C per minute [59].
- Long-term Storage: After 24 hours, transfer the frozen vials to a liquid nitrogen tank for long-term storage in the vapor phase.
- Thawing (Rapid Warming): When needed, retrieve vials and immediately thaw by gently swirling in a 37°C water bath until only a small ice crystal remains (typically <1 minute) [59] [62].
- Revival: Wipe the vial with ethanol, transfer contents to a tube, and slowly add pre-warmed organoid culture medium. For direct seeding, plate the cell suspension immediately. For indirect seeding, centrifuge at 5000 rpm for 5 minutes, remove the supernatant containing CPAs, and then resuspend the pellet in fresh medium before plating [59].
Protocol: On-Plate Cryopreservation of Human Brain Organoids
This advanced protocol uses a pillar plate platform to minimize mechanical stress and improve CPA diffusion for neural organoids [14] [58].
Key features: Enables in-situ cryopreservation, eliminating the need for harvesting and centrifugation, thus preserving 3D structure.
Materials and Reagents:
- Biological Material: Early-stage human brain organoids (ES-HBOs) derived from iPSCs.
- Platform: 36-pillar plate with sidewalls and slits (36PillarPlate).
- CPA: PSC Cryopreservation Kit, CryoStor CS10, or 3dGRO.
- Culture Reagents: Matrigel, Neural Induction Medium (NIM), mTeSR Plus medium.
Procedure:
- Organoid Generation and Transfer: Differentiate iPSCs into neuroectoderm (NE) spheroids in an ultralow attachment 384-well plate. On day 7, transfer NEs to a Matrigel-loaded pillar plate.
- CPA Optimization: For ES-HBOs with a diameter of 400–600 μm, incubate with the PSC Cryopreservation Kit for 80 minutes at room temperature to ensure sufficient CPA diffusion [58]. Note: Smaller organoids (<400 μm) may require shorter incubation times.
- Cooling: Place the entire pillar plate in an isopropanol freezing jar and store at -80°C overnight.
- Long-term Storage: Transfer the pillar plate to a vapor-phase liquid nitrogen dewar.
- Thawing (Rapid Warming): Rapidly warm the pillar plate by placing it in a 37°C incubator or water bath for uniform heat transfer.
- CPA Removal and Culture: Gently wash the organoids on the pillar plate with fresh medium to remove CPAs. Continue culturing and differentiation for over 30 days to obtain mature HBOs.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Organoid Cryopreservation
| Reagent / Material | Function / Application | Examples & Notes |
|---|---|---|
| Cryoprotectant Agents (CPAs) | Prevent ice crystal formation; reduce freezing point. | DMSO: Penetrating CPA; standard but can be cytotoxic. Ethylene Glycol: Penetrating CPA; used in vitrification. Sucrose/Trehalose: Non-penetrating CPAs; provide osmotic balance [61] [57] [62]. |
| Cryopreservation Media | Formulated solutions to maximize cell survival. | CryoStor CS10: Pre-defined, xeno-free; shows high viability. FBS + 10% DMSO: High recovery/viability for fibroblasts. PSC Cryopreservation Kit: Low toxicity for neural organoids [59] [60] [58]. |
| Specialized Equipment | Control cooling/warming rates and sample handling. | CoolCell / Mr. Frosty: Provides -1°C/min cooling rate. Pillar Plate: Allows in-situ cryopreservation, minimizing mechanical stress [59] [58]. |
| Nanomaterials | Advanced strategies to mitigate cryoinjury. | Magnetic Nanoparticles (e.g., Fe3O4): Enable uniform "nanowarming" under magnetic fields. Mesoporous Silica/ Graphene Oxide: Act as ice inhibitors by providing nucleation sites [23] [57]. |
Strategic Workflow and Pathway Visualization
The following diagrams outline the core experimental workflow for organoid cryopreservation and the strategic decision-making pathway for optimizing cooling and warming rates.
Organoid Cryopreservation Workflow
Diagram 1: Standard organoid cryopreservation and revival workflow.
Optimization Strategy Pathway
Diagram 2: Strategic pathway for optimizing cooling and warming protocols based on organoid characteristics.
Optimizing cooling and warming rates is not a one-size-fits-all endeavor but a deliberate process that must account for the specific physical and biological properties of each organoid type. The integration of advanced CPAs, precise cooling devices like the CoolCell, and rapid warming techniques forms the foundation of successful cryopreservation. Emerging technologies, particularly nanomaterials for nanowarming and specialized platforms like pillar plates, are pushing the boundaries by enabling uniform heat transfer and reducing mechanical stress. By adhering to the detailed protocols and strategic frameworks outlined in this application note, researchers can significantly enhance the viability, functionality, and reproducibility of cryopreserved organoids. This progress is crucial for realizing the full potential of next-generation living biobanks, which will serve as centralized resources to fuel drug discovery and personalized medicine.
The field of organoid biobanking faces a significant challenge: the transition from long-term cryopreservation at ultra-low temperatures back to physiological conditions must be controlled with precision to ensure high post-thaw viability and functionality. Conventional water bath thawing methods generate uncontrolled warming rates and thermal gradients, causing massive ice recrystallization, osmotic stress, and mechanical damage that compromise organoid integrity [50]. Within the context of organoid biobanking and cryopreservation research, advanced rewarming technologies have emerged as critical solutions to these challenges, enabling the reliable recovery of complex, multicellular systems for research and clinical applications.
Nanowarming and conductive chip technologies represent two pioneering approaches that utilize fundamentally different mechanisms to achieve rapid and uniform warming. Nanowarming employs magnetic nanoparticles (MNPs) dispersed throughout the biological sample, which generate heat when exposed to an alternating magnetic field (AMF) [23] [63]. This volumetric heating method overcomes the limitations of surface-based heating, ensuring consistent temperature distribution even in larger samples like organoids. Conductive chip-based rewarming, alternatively, utilizes microfabricated chips with integrated metal thin films or electrodes that heat rapidly via Joule heating when an electric current is applied, bringing samples to physiological temperatures in milliseconds [63] [64]. Both strategies aim to surpass critical warming rates (estimated to be approximately 30,000 °C/s for cells) to vitrify the ice-liquid phase transition, thereby preventing destructive ice crystal growth [65] [50]. The integration of these technologies is foundational to the development of Next-Generation Living Biobanks (NGLB), where organoids can be stored as "off-the-shelf" products and recovered with minimal functional compromise [63] [64].
The following table summarizes the core characteristics, mechanisms, and applications of Nanowarming and Conductive Chip rewarming technologies.
Table 1: Comparative Analysis of Advanced Rewarming Technologies
| Feature | Nanowarming | Conductive Chip Rewarming |
|---|---|---|
| Fundamental Mechanism | Inductive heating of magnetic nanoparticles (e.g., Fe₃O₄) via an alternating magnetic field (AMF) [23] [63] | Resistive (Joule) heating of a conductive metal film or pattern on a microfabricated chip [63] [64] |
| Heating Profile | Volumetric, from within the sample | Surface-based, conducted into the sample |
| Key Components | Magnetic nanoparticles (MNPs), AMF generator, sample container [23] | Conductive chip (e.g., with metal films), electrical power supply, control unit [63] |
| Reported Warming Rates | Highly efficient, enables recovery of heart organoids [63] | Extremely high (enables rates >10,000 °C/s), suitable for droplet vitrification [65] [64] |
| Sample Compatibility | Suitable for larger volumes and complex tissues (e.g., heart organoids) [23] [63] | Ideal for small-volume samples (e.g., droplets, micro-encapsulated organoids) [65] [64] |
| Key Advantage | Superior uniformity in larger samples, overcoming thermal insulation limits | Unmatched speed for small samples, precise electronic control |
| Potential Limitation | Requires incorporation and potential removal of nanoparticles from the sample | Scaling to larger sample volumes can be challenging due to thermal mass |
Detailed Experimental Protocols
Protocol A: Nanowarming of Heart Organoids Using Fe₃O₄ Nanoparticles
This protocol, adapted from Lee et al. (2024), details the cryopreservation and subsequent nanowarming of heart organoids, achieving high viability and functional recovery [63].
3.1.1 Pre-warming Preparation and Cryoprotectant (CPA) Loading
- Nanoparticle Preparation: Prepare a stock solution of biocompatible iron oxide (Fe₃O₄) nanoparticles (e.g., 10-15 nm diameter) in a basal culture medium at a concentration of 2-5 mg/mL. Sterilize the solution by passing it through a 0.22 µm filter.
- CPA Cocktail Formulation: Prepare a vitrification solution. A common formulation is based on 40% (v/v) ethylene glycol and 30% (v/v) DMSO in a suitable culture medium, supplemented with sucrose (e.g., 0.5 M) as a non-penetrating osmolyte [64] [50].
- MNP-CPA Integration: Mix the Fe₃O₄ nanoparticle stock solution with the CPA vitrification solution at a 1:1 ratio to achieve the final working concentration. Keep this solution on ice.
- Equilibration: Transfer the heart organoids to the MNP-CPA solution in a stepwise manner to minimize osmotic shock. A typical protocol involves 3-minute equilibration steps at 4°C. Gently agitate to ensure uniform exposure.
3.1.2 Freezing and Storage
- Packaging: Following equilibration, aspirate the organoids in a minimal volume of the MNP-CPA solution and transfer them into a standard 2 ml cryovial.
- Superflash Freezing: Immediately plunge the cryovial directly into liquid nitrogen. The high cooling rate, combined with the CPA cocktail, promotes vitrification rather than crystalline ice formation.
- Storage: Maintain the organoids in a liquid nitrogen Dewar at -196°C for long-term storage.
3.1.3 Nanowarming Process
- System Setup: Pre-warm a water bath or bead bath to 37°C. Pre-warm standard culture medium to 37°C.
- AMF Configuration: Place the cryovial containing the frozen sample into a custom-built alternating magnetic field (AMF) applicator. The system typically operates at a frequency of 200-500 kHz and a field strength of 15-30 kA/m [63].
- Activation: Apply the AMF for a short duration (typically 60-90 seconds). The Fe₃O₄ nanoparticles will dissipate heat uniformly throughout the sample, resulting in rapid warming rates that devitrify the sample.
- Temperature Monitoring: Use a fiber-optic temperature probe placed near the sample to monitor the temperature in real-time, ensuring it does not exceed 37°C.
3.1.4 Post-warming Recovery and Analysis
- Immediate Transfer: As soon as the sample is thawed, immediately transfer the organoids to the pre-warmed 37°C culture medium.
- CPA Removal: Gently wash the organoids 3-5 times with fresh, pre-warmed culture medium to dilute and remove the CPAs and any residual nanoparticles.
- Viability Assessment: Quantify cell viability using a live/dead assay (e.g., Calcein-AM/Ethidium Homodimer-1) and imaging after 24 hours in culture.
- Functional Analysis: For heart organoids, assess functional recovery by measuring the spontaneous beating rate and rhythm via video microscopy after 48-72 hours in culture [63].
Protocol B: High-throughput Thawing of Organoid Droplets via Conductive Chips
This protocol leverages the IIDR (Impact-Induced Droplet Release) thawing method and conductive chip principles for CPA-free or low-CPA cryopreservation of organoid droplets, ideal for high-throughput drug screening applications [65].
3.2.1 Chip-based Freezing
- Droplet Generation: Use an inkjet-based printer or a microfluidic droplet generator to dispense organoid suspensions into uniform, small-volume droplets (e.g., 70-200 pL) directly onto a conductive chip substrate, such as a silicon chip with patterned gold or copper thin films.
- CPA-free or Low-CPA Formulation: For CPA-free freezing, use a pure culture medium. For low-CPA formulations, include low concentrations of permeating CPAs like ethylene glycol (e.g., <10%) [65].
- Superflash Freezing: Immediately after droplet deposition, transfer the entire conductive chip into a cooling chamber or directly plunge it into a cryogen (e.g., liquid nitrogen slush) to achieve ultra-fast cooling rates exceeding 15,000 °C/s, leading to vitrification.
3.2.2 Conductive Chip Rewarming
- System Preparation: Pre-warm a large volume (e.g., 50 mL) of recovery medium in a well of a multi-well plate to 37°C.
- Chip Activation: Place the frozen chip into a custom holder that makes electrical contact. Apply a short, controlled electrical pulse (e.g., milliseconds to seconds) to the conductive metal film on the chip. The resulting Joule heating rapidly increases the chip's temperature.
- Impact-Induced Droplet Release (IIDR): The rapid thermal expansion of the chip substrate mechanically ejects the frozen droplets from the surface. These droplets are directly released into the pre-warmed recovery medium below, ensuring instantaneous and uniform warming. This method has been shown to improve warming rates by approximately threefold for 200 pL droplets compared to substrate-based methods [65].
3.2.3 Post-thaw Collection and Analysis
- Collection: Gently swirl the recovery medium to ensure all thawed droplets are collected.
- Viability Assessment: After 24 hours in culture, assess viability. This protocol has achieved >80% viability in 200 pL droplets containing NIH 3T3 fibroblasts [65].
- Organoid Culture: Transfer the recovered organoid droplets into a standard 3D culture system (e.g., Matrigel dome) and monitor for regrowth and proliferation.
The Scientist's Toolkit: Key Research Reagent Solutions
The successful implementation of advanced rewarming protocols relies on a suite of specialized reagents and materials. The following table catalogues the essential components for researchers embarking on these techniques.
Table 2: Essential Reagents and Materials for Advanced Rewarming Studies
| Item | Function/Description | Example Application/Note |
|---|---|---|
| Iron Oxide (Fe₃O₄) Nanoparticles | Magnetic core for heat dissipation under an AMF [23]. | ~10-15 nm diameter, surface-functionalized for biocompatibility and dispersion [63]. |
| Alternating Magnetic Field (AMF) System | Generator and coil producing the oscillating magnetic field for nanowarming [23] [63]. | Typical parameters: 200-500 kHz frequency, 15-30 kA/m field strength. |
| Conductive Chip (Au/Cu薄膜) | Microfabricated substrate for Joule heating [63] [64]. | Features patterned metal thin films; can be custom-designed for specific sample formats. |
| Vitrification Cocktail (e.g., EG/DMSO) | Penetrating CPAs that suppress ice crystal formation during freezing [64] [50]. | High concentrations (e.g., 40% EG, 30% DMSO) required for vitrification; cytotoxicity is a key concern. |
| Sucrose | Non-penetrating osmolyte that aids in dehydration and reduces CPA toxicity [64]. | Commonly used at 0.3-0.5 M in vitrification solutions. |
| Rho-associated Kinase (ROCK) Inhibitor (Y-27632) | Small molecule inhibitor that reduces apoptosis in thawed cells and organoids [66]. | Often added to recovery medium at 10 µM for the first 24-48 hours post-thaw. |
| Deep Eutectic Solvents (DES) | Emerging class of potentially less toxic, naturally derived CPAs [64] [50]. | Considered as a partial replacement for traditional CPAs like DMSO. |
Nanowarming and conductive chip technologies are no longer speculative concepts but are now validated, powerful tools that directly address the critical bottleneck of the rewarming process in organoid cryopreservation. By enabling rapid and uniform warming, these strategies significantly improve post-thaw viability, preserve complex cellular architectures, and maintain functional integrity, which is paramount for reliable drug screening and disease modeling.
The future trajectory of these technologies points toward greater integration and automation. A key challenge for nanowarming is the standardization and potential regulatory pathway for nanoparticles within biological products [23]. Future work will focus on optimizing nanoparticle biocompatibility, coating, and clearance. For conductive chips, the primary challenge is scaling to accommodate larger and more complex organoid cultures and tissues. The convergence of these technologies with other advanced strategies—such as hydrogel-based microencapsulation for added mechanical protection and the use of ice-binding polymers to further control ice recrystallization—will create synergistic effects [64] [50]. Ultimately, the refinement and adoption of these advanced rewarming platforms are fundamental to realizing the vision of global, standardized, and clinically relevant Next-Generation Living Biobanks, transforming organoids into truly accessible "off-the-shelf" reagents for biomedicine.
Ensuring Fidelity: Validation Frameworks and Comparative Efficacy Analysis
Benchmarking Viability and Growth Post-Thaw
The establishment of robust and reproducible cryopreservation protocols is a critical component for the operational feasibility of patient-derived organoid (PDO) biobanks, which are indispensable platforms for precision medicine and drug development. This application note provides a detailed framework for benchmarking the viability and growth of organoids following thawing procedures. We summarize quantitative data from key studies, present standardized experimental protocols for post-thaw assessment, and delineate essential reagent solutions. By integrating recent advancements in cryopreservation media and viability assays, this document serves as a technical guide for researchers and biobank managers aiming to optimize recovery of functionally competent organoids for downstream translational applications.
Organoid biobanks represent living repositories of three-dimensional, patient-derived microtissues that closely recapitulate the histological, genetic, and functional characteristics of their parental primary tissues [4]. These biobanks are pivotal for high-throughput drug screening, disease modeling, and the advancement of personalized therapeutic strategies. A major technical bottleneck in the widespread utilization and distribution of these biospecimens is the development of reliable cryopreservation methods that ensure high post-thaw viability and retention of key biological functions [67].
The complex, multicellular architecture of organoids renders them particularly vulnerable to cryoinjury, which can manifest as ice crystal formation, osmotic stress, loss of intercellular connections, and ultimately, impaired proliferation and functionality [23] [67]. Therefore, systematic benchmarking of post-thaw recovery is not merely a measure of cell survival but a critical quality control metric for ensuring that cryopreserved organoids are fit for purpose in subsequent research and clinical applications. This document outlines standardized protocols and analytical tools for this essential benchmarking process, framed within the broader context of establishing next-generation living biobanks (NGLBs) [67].
Quantitative Benchmarking Data from Recent Studies
The following tables consolidate key quantitative findings from recent investigations into organoid cryopreservation, providing benchmarks for expected performance across different organoid types and preservation strategies.
Table 1: Post-Thaw Viability and Success Rates in Organoid Cryopreservation Studies
| Organoid Type | Tissue Source | Cryopreservation Method | Key Viability/Success Metric | Reference |
|---|---|---|---|---|
| General Tumor Organoids | Patient-derived tumor tissues | Novel live-tissue freezing technique | 95.2% success rate for PDO generation from frozen tissues | [10] |
| Cortical Organoids | Human pluripotent stem cells | MEDY solution (Methylcellulose, Ethylene glycol, DMSO, Y27632) | Reliable storage and resuscitation of diverse neural organoids | [14] |
| Liver Organoids | Human liver cells | WellReady hypothermic hydrogel (5 days at 20°C) | High viability, maintained 3D structure, and functional capacity post-preservation | [68] |
| Cardiac Organoids | Human pluripotent stem cells | Fe₃O₄ nanoparticle-assisted nanowarming | Enhanced post-thaw survival rate via uniform thawing | [23] |
Table 2: Functional Benchmarking of Organoids Derived from Cryopreserved Tissues
| Functional Assessment | Methodology | Key Finding | Implication for Biobanking | Reference |
|---|---|---|---|---|
| Drug Response | High-throughput screening | Similar drug responses between PDOs from frozen and fresh tissues | Cryopreserved libraries are suitable for drug discovery | [10] |
| Histological & Molecular Integrity | Histology, tumor marker expression | Structural features and marker expression maintained post-thaw | Preserves original tumor characteristics | [10] |
| Tumor Microenvironment (TME) Preservation | Air-liquid interface (ALI) culture | TME maintained, enabling immune checkpoint inhibitor testing | Facilitates immunotherapy research | [10] |
| Model Reproducibility | Long-term expansion, passaging | Organoids from frozen tissues can be routinely passaged and re-frozen | Enables scalable, high-throughput screening | [10] |
Experimental Protocols for Post-Thaw Assessment
Protocol: Thawing and Initial Recovery of Cryopreserved Organoids
This protocol is adapted from established methods for resuscitating cryopreserved organoids [14] [10].
Principle: To successfully recover viable organoids from liquid nitrogen storage while minimizing osmotic stress and cryoprotectant agent (CPA) toxicity during the thawing process.
Reagents:
- Pre-warmed complete organoid culture medium (37°C)
- Basal washing medium (e.g., Advanced DMEM/F12)
- Cryopreserved vial of organoids
Procedure:
- Rapid Thawing: Retrieve a cryovial from long-term storage and immediately place it in a 37°C water bath. Gently agitate until only a small ice crystal remains (approximately 2-3 minutes).
- CPA Dilution: Transfer the thawed suspension to a 15 mL conical tube. Slowly add 10 mL of pre-warmed basal washing medium dropwise over 5 minutes to gently dilute the CPA.
- Pellet Collection: Centrifuge the tube at 200-300 x g for 5 minutes at room temperature.
- CPA Removal: Carefully aspirate the supernatant, ensuring not to disturb the soft pellet.
- Reseeding: Resuspend the organoid pellet in an appropriate, chilled extracellular matrix (e.g., Matrigel). Plate the mixture in pre-warmed culture plates and polymerize at 37°C for 15-20 minutes.
- Culture Initiation: Overlay the polymerized drops with pre-warmed complete organoid culture medium. Culture at 37°C, 5% CO₂, with medium changes every 2-3 days.
Protocol: Assessing Viability and Growth Kinetics
Principle: To quantitatively measure the viability and proliferative recovery of organoids following the thawing process.
Reagents:
- CellTiter-Glo 3D Cell Viability Assay or equivalent ATP-based assay
- Calcein-AM / Propidium Iodide (PI) staining solution
- Phosphate Buffered Saline (PBS)
- 4% Paraformaldehyde (PFA)
- Reagents for immunostaining (e.g., primary and secondary antibodies, blocking buffer)
Procedure:
- Metabolic Activity (ATP Assay):
- At defined time points post-thaw (e.g., days 1, 3, 7), transfer organoids to a white-walled 96-well plate.
- Add an equal volume of CellTiter-Glo 3D reagent to each well.
- Shake the plate for 5 minutes on an orbital shaker to induce cell lysis, then incubate for 25 minutes at room temperature to stabilize the luminescent signal.
- Record luminescence using a plate reader. Normalize values to a fresh, non-cryopreserved organoid control.
Live/Dead Staining:
- Harvest organoids and wash once with PBS.
- Prepare a working solution of 2 µM Calcein-AM and 4 µM Propidium Iodide in PBS.
- Incubate the organoids in the staining solution for 30-45 minutes at 37°C, protected from light.
- Wash the organoids twice with PBS and image immediately using a confocal or fluorescence microscope.
- Quantification: Viability (%) = [Calcein-AM positive area / (Calcein-AM positive area + PI positive area)] x 100.
Growth Kinetics and Morphology:
- Monitor organoid size and morphology daily using bright-field microscopy.
- Quantify the increase in organoid diameter or cross-sectional area over time using image analysis software (e.g., ImageJ).
- Passage organoids when they reach a critical size and record the doubling time across subsequent passages to assess long-term functional recovery.
Diagram 1: Organoid Post-Thaw Recovery and Benchmarking Workflow.
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and their specific functions in organoid cryopreservation and post-thaw analysis, as cited in recent literature.
Table 3: Essential Reagents for Organoid Cryopreservation and Viability Benchmarking
| Reagent / Solution | Function / Application | Specific Example / Note |
|---|---|---|
| MEDY Cryopreservation Solution | A specialized formulation for reliable cryopreservation of neural organoids and human brain tissue. | Comprises Methylcellulose, Ethylene glycol, DMSO, and Y27632 (a Rho kinase inhibitor) [14]. |
| Rho Kinase (ROCK) Inhibitor (Y27632) | Promotes cell survival and inhibits apoptosis during the critical post-thaw recovery phase. | Commonly added to recovery media for 24-48 hours after thawing [14]. |
| Hypothermic Hydrogel (WellReady) | Enables ambient temperature storage and shipment of organoids by stabilizing cell membranes. | Alginate-based hydrogel; used for liver organoid storage at 20°C for 5 days [68]. |
| Magnetic Nanoparticles (Fe₃O₄) | Facilitates uniform "nanowarming" during thawing to mitigate thermal stress and improve viability. | Applied in cardiac organoid cryopreservation protocols for enhanced recovery [23]. |
| ATP-based Viability Assay (CellTiter-Glo 3D) | Quantifies metabolically active cells in 3D structures; core assay for benchmarking viability post-thaw. | Provides a quantitative luminescent readout proportional to the number of viable cells [10]. |
| Calcein-AM / Propidium Iodide | Fluorescent live/dead staining for direct visualization and quantification of viability in 3D organoids. | Calcein-AM (green) stains live cells; PI (red) stains dead cells with compromised membranes. |
Discussion and Concluding Remarks
Systematic benchmarking of viability and growth post-thaw is the cornerstone of quality assurance for any functional organoid biobank. The data and protocols presented herein demonstrate that with optimized techniques, it is feasible to achieve high success rates in generating and expanding organoids from cryopreserved tissues, with fidelity to the original tumor's drug response and histological profile [10]. The field is moving beyond simple viability metrics toward a more holistic assessment of functional recovery, including the preservation of complex cellular interactions and the tumor microenvironment.
Future directions in organoid cryopreservation benchmarking will likely involve the integration of advanced nanomaterials to improve cryoprotectant delivery and rewarming uniformity [23], the adoption of hypothermic preservation as a complementary strategy to cryopreservation for simplified logistics [68], and the establishment of standardized, scalable protocols to support the vision of next-generation living biobanks. By adhering to rigorous, quantitative benchmarking practices, researchers can ensure that cryopreserved organoids serve as reliable and predictive tools in the transition toward personalized medicine and more efficient drug development pipelines.
Validating Genetic, Structural, and Functional Integrity
Within organoid biobanking and cryopreservation research, rigorous validation of genetic, structural, and functional integrity is paramount to ensure that preserved organoids faithfully recapitulate in vivo physiology upon recovery. Organoids, as three-dimensional in vitro models, must maintain key characteristics of their tissue of origin—including cellular heterogeneity, tissue-specific architecture, and physiological functions—to be reliable tools for disease modeling, drug screening, and regenerative medicine [34] [69]. The process of cryopreservation and subsequent thawing introduces significant stress that can compromise these essential qualities, making comprehensive validation protocols a critical component of the biobanking workflow. This document outlines standardized methodologies for assessing organoid integrity across genetic, structural, and functional domains, providing a framework for quality assurance in organoid biobanking operations.
Validation of Genetic Integrity
Genetic integrity refers to the stability and accuracy of genomic information within organoids before and after cryopreservation. Maintaining a consistent genetic profile is essential for disease modeling applications, particularly for patient-derived organoids used in personalized medicine.
Genomic Stability Assessment
Protocol: Whole Genome Sequencing for Genetic Stability
- Extract genomic DNA from pre-cryopreservation and post-thaw organoids using a commercial kit suitable for 3D culture models.
- Assess DNA quality using agarose gel electrophoresis and quantify using fluorometric methods.
- Prepare sequencing libraries using a PCR-free library preparation kit to minimize bias.
- Sequence libraries on a high-throughput sequencing platform to achieve at least 30x coverage.
- Analyze data by aligning sequences to a reference genome and identifying single nucleotide variations (SNVs), copy number variations (CNVs), and structural variations (SVs).
- Compare pre- and post-cryopreservation profiles to identify cryopreservation-induced genetic alterations.
Multi-omics approaches, particularly genomic analysis, allow researchers to test whether organoids maintain directional differentiation and genomic stability after cryopreservation [70]. Genomic studies help reassess the suitability of complex organoid culturing conditions and can reveal the anatomical origin of organoids from different sources [70].
Epigenetic Consistency
Protocol: DNA Methylation Analysis
- Extract DNA from organoid samples using a kit that preserves methylation patterns.
- Treat DNA with bisulfite to convert unmethylated cytosines to uracils.
- Perform genome-wide methylation profiling using array-based or sequencing-based methods.
- Analyze differential methylation between pre-cryopreservation and post-thaw organoids, focusing on regions known to regulate tissue-specific gene expression.
Organoids maintain segmental DNA methylation patterns and age stages during cellular differentiation, making them reliable tools for investigating stem cell-intrinsic aging in vitro [70]. Preservation of these epigenetic patterns after cryopreservation indicates maintained cellular identity.
Table 1: Genetic Integrity Assessment Methods
| Assessment Method | Key Parameters Measured | Acceptance Criteria | Technical Considerations |
|---|---|---|---|
| Whole Genome Sequencing | SNVs, CNVs, SVs | >95% concordance between pre- and post-cryopreservation | PCR-free library prep recommended to reduce bias |
| DNA Methylation Array | Genome-wide methylation patterns | Consistent methylation at tissue-specific regulatory elements | Bisulfite conversion efficiency must be >99% |
| Karyotyping | Chromosomal number and structure | Normal diploid karyotype maintained | Requires single-cell suspension from organoids |
| RT-qPCR | Expression of housekeeping and tissue-specific genes | Stable expression of reference genes | Normalize to multiple reference genes |
Validation of Structural Integrity
Structural integrity encompasses the histological, morphological, and ultrastructural characteristics that define organoid architecture and cellular composition.
Histological and Morphological Assessment
Protocol: Immunohistochemistry for Tissue-Specific Markers
- Fix organoids in 4% paraformaldehyde for 30-60 minutes at room temperature.
- Embed in paraffin or optimal cutting temperature (OCT) compound following standard histological protocols.
- Section at 4-7μm thickness using a microtome or cryostat.
- Perform antigen retrieval using appropriate buffer (e.g., citrate buffer) and heating method.
- Block nonspecific binding with 5% normal serum from the secondary antibody host species.
- Incubate with primary antibodies targeting tissue-specific markers overnight at 4°C.
- Incubate with fluorophore-conjugated secondary antibodies for 1-2 hours at room temperature.
- Counterstain with DAPI for nuclear visualization.
- Image using confocal microscopy to capture 3D architecture.
Organoids must recapitulate the cellular diversity and spatial organization of native tissues [71]. Histological analysis restores the full picture of architectures on a macro-scale to assess the similarity to in vivo organs [70]. For intestinal organoids, this includes the formation of crypt-villus structures with appropriate cellular polarization; for cerebral organoids, the presence of layered cortical structures; and for hepatic organoids, the formation of bile canaliculi-like structures.
Ultrastructural Analysis
Protocol: Transmission Electron Microscopy
- Fix organoids in 2.5% glutaraldehyde in 0.1M cacodylate buffer for 2 hours at room temperature.
- Post-fix in 1% osmium tetroxide for 1 hour.
- Dehydrate through a graded ethanol series (50%, 70%, 90%, 100%).
- Infiltrate and embed in epoxy resin.
- Section at 60-90nm thickness using an ultramicrotome.
- Stain sections with uranyl acetate and lead citrate.
- Image using a transmission electron microscope.
This protocol allows assessment of subcellular structures including tight junctions, desmosomes, microvilli, and specialized organelles that are critical for organoid function.
Diagram 1: Structural validation workflow for organoid assessment.
Validation of Functional Integrity
Functional integrity confirms that organoids not only look like their in vivo counterparts but also perform their characteristic physiological activities after cryopreservation.
Metabolic and Secretory Functions
Protocol: LC-MS/MS for Metabolic Profiling
- Extract metabolites from organoids using a methanol:water:chloroform (4:3:1) extraction method.
- Centrifuge at 14,000 × g for 15 minutes to separate phases.
- Collect aqueous phase and dry using a speed vacuum concentrator.
- Reconstitute in appropriate LC-MS solvent.
- Analyze using a high-resolution LC-MS/MS system with reverse-phase chromatography.
- Identify metabolites by comparing retention times and mass spectra to authentic standards.
- Quantify using stable isotope-labeled internal standards.
Targeted analysis of central carbon metabolites in intestinal organoids has shown that donor-donor variability can be maintained at manageable levels, enabling robust experimental design for studying metabolic processes [70]. Metabolic profiling validates that essential biochemical pathways remain intact after cryopreservation.
Electrophysiological Activity
Protocol: Multi-Electrode Array Recording for Neural Organoids
- Transfer cerebral organoids to a multi-electrode array (MEA) chamber containing pre-warmed artificial cerebrospinal fluid.
- Allow equilibration for 10-15 minutes.
- Record spontaneous activity for 10 minutes at a sampling rate of at least 10 kHz.
- Apply pharmacological agents (e.g., glutamate receptor antagonists) to confirm specific neural network activity.
- Analyze data for spike rate, burst patterns, and network synchronization.
Studies have demonstrated that human cerebral organoids maintain viability and functional integrity after transport, with preserved spontaneous electrophysiological activity confirmed through MEA recordings [72]. This functional assessment is crucial for neurological disease modeling.
Barrier Function and Transport
Protocol: TEER Measurement for Epithelial Organoids
- Seed intestinal organoids on Transwell filters and allow to form confluent monolayers.
- Measure transepithelial electrical resistance (TEER) using an epithelial voltohmmeter.
- Calculate TEER values by subtracting blank filter resistance and multiplying by the membrane area.
- Perform transport assays by adding marker compounds to the apical chamber and sampling from the basolateral chamber at timed intervals.
- Analyze samples using LC-MS or scintillation counting for radiolabeled compounds.
Intestinal organoids recapitulate barrier functions including selective permeability and polarized transport, essential features for drug absorption studies [69].
Table 2: Functional Assessment Methods for Different Organoid Types
| Organoid Type | Functional Assays | Key Readouts | Quality Thresholds |
|---|---|---|---|
| Hepatic | Albumin production, CYP450 activity, urea synthesis | Albumin: >100ng/mL/day; CYP3A4 activity: inducible >2-fold | Metabolic competence maintained post-thaw |
| Intestinal | TEER, digestive enzyme secretion, mucus production | TEER: >150Ω·cm²; alkaline phosphatase: detectable | Barrier function preserved |
| Neural | MEA recording, calcium imaging, neurotransmitter release | Synchronized bursting activity; responsive to receptor agonists | Network activity patterns maintained |
| Renal | Albumin uptake, electrolyte transport, hormone response | Selective molecular transport; response to vasopressin | Transport specificity retained |
| Cardiac | Contractility analysis, calcium transients, electrophysiology | Regular beating rate; coordinated calcium waves | Spontaneous contraction recovery |
Integrated Validation Workflow
A comprehensive validation strategy integrates genetic, structural, and functional assessments in a sequential workflow to fully characterize organoid integrity after cryopreservation.
Diagram 2: Integrated validation workflow for organoid integrity assessment.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Organoid Integrity Validation
| Reagent/Category | Specific Examples | Function in Validation | Technical Notes |
|---|---|---|---|
| Extracellular Matrices | Matrigel, collagen-based hydrogels, synthetic PEG hydrogels | Provide 3D structural support mimicking native microenvironment | Matrix composition influences organoid differentiation and function [23] |
| Cryoprotectants | DMSO, ethylene glycol, sucrose, trehalose | Protect cellular structures during freeze-thaw cycles | Nanomaterials show promise as advanced cryoprotectants [23] |
| Growth Factors & Small Molecules | R-spondin, Noggin, EGF, Wnt agonists, TGF-β inhibitors | Maintain stemness or direct differentiation | Controlled release using nanoparticles improves stability [23] |
| Antibodies for Characterization | Tissue-specific markers (e.g., LGR5 for intestine, TUJ1 for neurons) | Identify cellular composition and differentiation status | Validate specificity for organoid models using appropriate controls |
| Viability Assays | Calcein-AM/EthD-1 (Live/Dead), ATP-based assays, TUNEL | Quantify cell survival and apoptosis post-thaw | 3D-optimized protocols required for accurate assessment |
| Functional Probes | Calcium indicators (Fluo-4), membrane potential dyes, pH sensors | Monitor dynamic physiological processes | Rationetric probes preferred for quantitative measurements |
| Nuclease Inhibitors | RNase inhibitors, DNase inhibitors | Preserve nucleic acid integrity during processing | Critical for obtaining high-quality omics data |
Implementing a comprehensive validation framework for assessing genetic, structural, and functional integrity is essential for establishing reliable organoid biobanking systems. The protocols outlined here provide standardized methodologies that enable researchers to verify that cryopreserved organoids maintain their essential characteristics post-thaw. As organoid technology continues to advance, with emerging applications in nanomaterials-assisted culture and cryopreservation [23], these validation approaches will ensure that biobanked organoids remain faithful models of human physiology and disease. Integrating these assessment protocols into routine biobanking operations will enhance reproducibility, facilitate data comparison across institutions, and ultimately strengthen the translational potential of organoid-based research.
Correlating Drug Response Between Fresh and Cryopreserved Organoids
The emergence of patient-derived organoids (PDOs) has revolutionized preclinical cancer research and personalized medicine by providing three-dimensional models that faithfully recapitulate the histological, genetic, and functional features of parental tumors [4]. However, the clinical translation of organoid technology faces significant logistical challenges, primarily due to the limited availability of fresh tumor tissues and the need for immediate processing to maintain viability [10] [73]. Cryopreservation has emerged as a critical strategy to overcome these limitations by enabling long-term storage and flexible utilization of biological specimens [73].
Establishing a robust correlation between drug responses in organoids derived from fresh and cryopreserved tissues is fundamental for validating biobanking approaches. This protocol summarizes recent advances in cryopreservation techniques that maintain the viability and drug sensitivity of organoids, enabling their use in high-throughput drug screening and personalized therapy planning [10] [74]. The application of these methods ensures that cryopreserved organoids retain the essential characteristics of their fresh counterparts, thereby enhancing the reproducibility and scalability of cancer research.
Success Rates and Correlation Metrics Across Studies
Table 1: Comparative Analysis of Organoid Generation and Drug Response from Fresh vs. Cryopreserved Tissues
| Tumor Type | Cryopreservation Method | Success Rate | Key Correlation Findings | Reference |
|---|---|---|---|---|
| Multiple solid tumors | Novel tissue freezing technique | 95.2% | Structural features, tumor marker expression, and drug responses similar to fresh tissue-derived organoids | [10] |
| Acute Myeloid Leukemia | Viable freezing of mononuclear cells | High viability post-thaw | Strong correlation in ex vivo drug sensitivity (139 compounds); frozen samples showed slightly stronger responses | [74] |
| Breast cancer xenografts | Slow freezing in DMSO media | Viable organoids generated | OMI index correlated in drug response; DMSO frozen tissue yielded more accurate response than flash-frozen | [75] |
| Glioblastoma | Optimized recovery with Matrigel + 20% FBS | Significant improvement | Recovered cells suitable for TMZ drug testing in 2D and 3D cultures after >10 years cryopreservation | [76] |
Morphological and Functional Preservation Metrics
Table 2: Morphological and Functional Characteristics of Organoids from Cryopreserved Tissues
| Parameter | Fresh Tissue Organoids | Cryopreserved Tissue Organoids | Statistical Significance |
|---|---|---|---|
| Viability Markers | Baseline Ki67 and cleaved caspase 3 expression | Similar expression levels | Not significant [75] |
| Structural Features | Native architecture maintained | Retained main features of original tumors | Not significant [10] |
| Drug Response Accuracy | Reference OMI index and IF staining | Strong correlation in drug sensitivity | Highly correlated (p<0.05) [75] |
| Passaging Capacity | Routinely passaged and frozen | Ideal for high-throughput screening | Comparable functionality [10] |
| TME Preservation | Native microenvironment | Suitable for ALI culture maintaining TME | Not significant [10] |
Experimental Protocols
Protocol for Cryopreserving Tumor Tissues for Organoid Generation
This protocol, adapted from a novel technique for cryopreserving live tumor tissues, demonstrates a 95.2% success rate in generating viable organoids after long-term freezing [10].
Materials and Equipment
- Tumor tissue specimens (surgical or biopsy)
- DMSO-supplemented cryopreservation medium
- Cryogenic vials
- Controlled-rate freezing apparatus
- Liquid nitrogen storage system
- Collagenase/hyaluronidase enzyme mixture
- TrypLE Express enzyme
- Basement membrane extract (BME) or Matrigel
- Organoid culture medium with growth factors
Procedure
Tissue Processing:
- Process fresh tumor tissues within 1 hour of resection.
- Remove non-epithelial tissue (muscle, fat) with surgical instruments.
- Cut primary tumor tissues into 1-3 mm³ pieces using scalpel or scissors.
Cryopreservation:
- Prepare cryopreservation medium with 10% DMSO in FBS.
- Place tissue fragments in cryovials with freezing medium.
- Implement controlled-rate freezing at -1°C/minute to -80°C.
- Transfer to liquid nitrogen for long-term storage after 24 hours.
Recovery and Organoid Generation:
- Rapidly thaw cryopreserved tissues in 37°C water bath.
- Wash twice with cold PBS to remove DMSO.
- Digest tissues with collagenase/hyaluronidase and TrypLE Express enzymes.
- Monitor digestion progress; complete when 2-10 cell clusters are visible.
- Filter through 70-100μm strainers based on tumor type.
- Mix cell clusters with BME/Matrigel and plate as hemispherical drops.
- Solidify at 37°C for 15-30 minutes before adding organoid culture medium.
- Include 10μM ROCK inhibitor during initial culture to improve viability.
Protocol for Drug Sensitivity Testing in Recovered Organoids
This protocol enables functional assessment of drug response in organoids derived from cryopreserved tissues, validated against fresh tissue-derived organoids [74] [75].
Materials and Reagents
- Organoids derived from fresh and cryopreserved tissues
- 96-well or 384-well plates for high-throughput screening
- Small molecule drug library (FDA-approved and investigational)
- Temozolomide (for GBM studies) or other tumor-specific agents
- Cell Counting Kit-8 (CCK-8) or similar viability assays
- Immunofluorescence staining reagents (Ki67, cleaved caspase 3 antibodies)
- Optical Metabolic Imaging (OMI) components if available
Drug Screening Procedure
Organoid Preparation:
- Harvest organoids at similar growth stages (typically 7-14 days).
- Dissociate into single cells or small clusters using TrypLE Express.
- Adjust density to 1×10⁶ cells/mL for spheroid formation.
Drug Treatment:
- Seed organoids in 96-well plates pre-coated with BME/Matrigel.
- Include triplicate wells for each drug concentration and DMSO controls.
- Implement drug treatment at two concentrations (typically 1μM and 10μM).
- Incubate for 72 hours with daily medium refreshment.
Response Assessment:
- Viability Analysis: Use CCK-8 assay per manufacturer's instructions.
- Immunofluorescence: Fix organoids and stain for Ki67 (proliferation) and cleaved caspase 3 (apoptosis).
- OMI Assessment: Measure NAD(P)H and FAD fluorescence lifetimes to detect metabolic shifts.
- Image Analysis: Quantify cell numbers and drug-induced cytotoxicity using high-content imaging.
Signaling Pathways in Cryopreservation Response and Recovery
Understanding the molecular mechanisms activated during cryopreservation and recovery is essential for optimizing protocols. Research indicates that specific pathways are modulated in response to freezing stress and subsequent revival.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Cryopreservation and Drug Response Studies
| Reagent/Category | Specific Examples | Function & Application |
|---|---|---|
| Cryoprotectants | DMSO, Natural antifreeze proteins | Prevent ice crystal formation, reduce cryoinjury during freezing/thawing |
| Extracellular Matrices | Matrigel, BME, Geltrex, Nanocellulose hydrogels | Provide 3D scaffold for organoid growth, mimic native microenvironment |
| Recovery Enhancers | ROCK inhibitor (Y-27632), 20% FBS | Improve post-thaw viability and outgrowth during initial culture |
| Viability Assays | CCK-8, Trypan blue exclusion, OMI | Quantify cell survival, metabolic activity, and drug response |
| Drug Screening Tools | Small molecule libraries, TMZ, targeted inhibitors | Assess therapeutic efficacy and resistance patterns |
| Characterization Antibodies | Ki67, cleaved caspase 3, SOX2, NESTIN | Evaluate proliferation, apoptosis, and stemness markers |
The established correlation between drug responses in organoids derived from fresh and cryopreserved tissues validates cryopreservation as a reliable strategy for biobanking and scalable drug screening applications. The quantitative data and standardized protocols presented herein provide researchers with robust methodologies for implementing cryopreservation approaches that maintain the phenotypic and functional characteristics of original tumors. As cryopreservation technologies continue to advance with innovations in nanowarming, natural cryoprotectants, and microfluidic CPA loading, the fidelity of drug response preservation is expected to further improve, ultimately enhancing the role of biobanked organoids in personalized medicine and drug discovery pipelines.
Quality Control Metrics for Biobank Standardization
Biobanks serve as indispensable pillars in biomedical research, functioning as organized repositories for vast collections of biological specimens and associated data [77]. These resources are crucial for advancing personalized medicine, accelerating drug discovery, and supporting public health initiatives [78]. The value of biobanked materials is fundamentally dependent on their quality, integrity, and fitness for purpose in downstream research applications [77] [79]. Standardization of quality control (QC) metrics across biobanking operations ensures that biological samples maintain their molecular integrity and research utility throughout collection, processing, storage, and distribution workflows [79].
Within the specific context of organoid biobanking and cryopreservation research, standardization faces unique challenges. Organoids, as three-dimensional, self-organizing microtissues that emulate native organ architecture and functionality, present particular difficulties for quality assessment due to their structural complexity, cellular heterogeneity, and sensitivity to cryoinjury [23] [80]. The emergence of Next-Generation Living Biobanks (NGLB) necessitates the development of robust, standardized QC protocols to ensure that cryopreserved organoids retain their phenotypic and functional characteristics post-thaw, enabling their reliable use in disease modeling, drug screening, and regenerative medicine [80]. This application note details comprehensive QC metrics and standardized protocols to address these challenges across diverse biobanked sample types, with particular emphasis on living systems such as organoids.
Essential Quality Control Metrics for Biospecimens
Quality control in biobanking requires a multi-parameter approach that assesses sample quality throughout the pre-analytical and storage phases. The following metrics provide a standardized framework for evaluating biospecimen integrity.
Table 1: Core Quality Control Metrics for Different Biospecimen Types
| Biospecimen Type | Key QC Metrics | Acceptance Criteria | Assessment Method |
|---|---|---|---|
| Plasma/Serum | Hemolysis Index, miRNA Integrity [79] | Consistent expression of ubiquitous miRNAs (e.g., miR-16, miR-451) [79] | Spectrophotometry, qRT-PCR |
| Peripheral Blood Mononuclear Cells (PBMCs) | Cell Viability, Immunophenotyping [79] | Viability >80%, Identifiable lymphocyte subsets (CD45+, CD3+, CD19+, CD56+) [79] | 7-AAD staining, Flow Cytometry |
| Tissue (RNA Quality) | RNA Integrity Number (RIN), 28S/18S Ratio [81] | RIN >7.0, 28S/18S ~2.0 [81] | Bioanalyzer, qRT-PCR |
| Organoids | Post-Thaw Viability, Structural Integrity, Functional Markers [10] [80] | High viability, retained original architecture & marker expression, similar drug response to fresh controls [10] | Metabolic assays, Histology, Immunofluorescence |
Standardized metrics must also account for pre-analytical variables. Storage temperature is a universal diagnostic measure, with long-term storage recommended at -196°C in liquid nitrogen, contrary to the still-common practice of storage at -80°C [81]. Transit time from collection to stabilization and transit temperature are critical predictive measures that are often poorly recorded; establishing and adhering to standardized SOPs for these parameters is essential [81].
For cryopreserved living tissues and organoids, additional viability and functionality assessments are paramount. Research demonstrates that with optimized cryopreservation protocols, tumor tissues can achieve a 95.2% success rate in generating patient-derived organoids (PDOs) that retain the structural features, marker expression, and drug responses of their fresh counterparts [10].
Standardized Experimental Protocols for Quality Control
Protocol: Viability and Immunophenotyping of Cryopreserved PBMCs
This protocol outlines a standardized method for assessing the quality of cryopreserved PBMCs, a common biospecimen in biobanks [79].
Principle: Cell viability is determined by staining with 7-Aminoactinomycin D (7-AAD), a fluorescent dye that is excluded by viable cells. Immunophenotyping characterizes lymphocyte subsets via cell surface markers using flow cytometry, confirming the preservation of immunologically relevant cells.
Materials and Reagents:
- Thawed PBMC sample
- 7-AAD Viability Staining Solution (e.g., Beckman Coulter, #IM3422)
- Antibody Mix for immunophenotyping (e.g., CD45-FITC, CD56-PE, CD19-ECD, CD3-PC5)
- Flow Cytometry Staining Buffer
- Phosphate Buffered Saline (PBS)
- Positive Control: Freshly isolated PBMCs
- Negative Control: PBMCs treated with 30% DMSO for 3 hours
Procedure:
- Sample Thawing: Rapidly thaw the cryovial containing PBMCs in a 37°C water bath. Immediately transfer cell suspension to a pre-filled tube with warm culture medium.
- Cell Washing: Centrifuge the cell suspension at 400 x g for 5 minutes. Carefully decant the supernatant and resuspend the cell pellet in PBS.
- Viability Staining: Aliquot 100 µL of cell suspension. Add 20 µL of 7-AAD solution. Incubate for 15-20 minutes at room temperature in the dark.
- Immunophenotyping Staining: Aliquot another 100 µL of cell suspension. Add the predetermined optimal concentration of antibody mix. Incubate for 30 minutes at 4°C in the dark. Wash cells with buffer and resuspend in PBS for acquisition.
- Flow Cytometry Analysis: Analyze samples using a calibrated flow cytometer (e.g., CytoFLEX, Beckman Coulter).
- For viability: Use a 7-AAD vs. Side Scatter (SSC) dot plot. The 7-AAD negative population represents viable cells.
- For immunophenotyping: Gate on CD45+ lymphocytes, then identify T cells (CD3+), B cells (CD19+), and NK cells (CD56+) within this population.
- Data Interpretation: Calculate the percentage of viable cells. The sample passes QC if viability exceeds 80% and distinct lymphocyte populations are identifiable.
Protocol: Quality Control of Plasma Samples via miRNA Integrity
Principle: The integrity of plasma samples is assessed by measuring the consistent expression of specific, ubiquitous microRNAs (miRNAs). This confirms that the biomolecular content has not degraded during processing or storage.
Procedure:
- RNA Extraction: Isolve total RNA, including small RNAs, from a fixed volume of thawed plasma (e.g., 200 µL) using a commercial kit suitable for biofluids.
- Reverse Transcription: Convert the extracted RNA to cDNA using stem-loop primers specific for the target housekeeping miRNAs (e.g., miR-16-5p, miR-451a).
- Quantitative PCR (qPCR): Perform qPCR amplification using TaqMan or SYBR Green chemistry.
- Data Analysis: Use the comparative Cq (ΔΔCq) method. Compare the expression levels of target miRNAs in the test sample against positive and negative controls. High-quality samples will show robust and consistent miRNA expression [79].
Diagram 1: PBMC Quality Control Workflow.
Advanced Protocol: Assessing Organoid Viability and Function Post-Cryopreservation
Principle: This protocol evaluates the success of organoid cryopreservation by assessing viability, structural integrity, and functional capacity after thawing, which is critical for NGLBs [10] [80].
Materials and Reagents:
- Thawed organoids
- Viability stain (e.g., Calcein-AM / Propidium Iodide)
- Growth factor-reduced Matrigel
- Organoid-specific culture medium
- Fixative (e.g., 4% PFA)
- Antibodies for key lineage markers
- Organoid-specific drug compounds for functional assays
Procedure:
- Thawing and Recovery: Rapidly thaw cryovials and transfer organoids to pre-warmed medium. Centrifuge gently and resuspend in Matrigel for 3D culture. Culture for 24-48 hours to allow recovery.
- Viability Staining: Use a dual fluorescent stain (e.g., Calcein-AM for live cells, Propidium Iodide for dead cells). Image using a confocal microscope and quantify the live/dead cell ratio.
- Histological and Phenotypic Analysis: Fix recovered organoids, embed in paraffin, and section. Perform Hematoxylin and Eosin (H&E) staining to assess general architecture. Conduct immunofluorescence for organoid-specific markers to confirm retention of key cell types.
- Functional Drug Screening Assay: Dissociate organoids to form a single-cell suspension and re-plate in Matrigel. After 3-5 days, treat with a panel of relevant drugs (e.g., chemotherapeutics for tumor organoids). After 72-96 hours, assess viability using a cell titer-glo 3D assay. Compare the dose-response curves of organoids derived from frozen tissues against those from fresh tissues. A high-quality cryopreservation outcome is indicated by similar IC50 values [10].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of standardized QC protocols relies on specific, high-quality reagents and instruments.
Table 2: Key Research Reagent Solutions for Biobank QC
| Item | Function/Application | Example/Notes |
|---|---|---|
| 7-AAD Viability Dye | Distinguishes viable from non-viable cells for flow cytometry. | Beckman Coulter #IM3422; excludes viable cells [79]. |
| Flow Cytometry Antibody Panels | Immunophenotyping of specific cell subsets (e.g., T, B, NK cells). | CD45, CD3, CD19, CD56 combinations [79]. |
| NMR Metabolomics Platform | Standardized QC analysis of liquid biopsies; provides quantitative metabolic data. | Bruker B.I. BioBank Tool; analyzes >115 lipoprotein parameters & 26 metabolites in plasma [82]. |
| RNA Integrity Number (RIN) | Objective assessment of RNA quality for downstream genomic applications. | Assessed via Agilent Bioanalyzer; RIN >7.0 is typically required [81]. |
| Natural Cryoprotectants (CPAs) | Reduce cytotoxicity during organoid cryopreservation. | Antifreeze proteins, Deep Eutectic Solvents [80]. |
| Magnetic Nanoparticles (MNPs) | Enable uniform nanowarming to improve post-thaw survival of complex organoids. | Fe3O4 nanoparticles for magnetic nanowarming [80]. |
| Hydrogel Microencapsulation Matrix | Protects organoids from mechanical and osmotic stress during freezing. | Alginate or other biocompatible polymer matrices [80]. |
The standardization of quality control metrics is not a static goal but an evolving discipline that must adapt to new sample types and technological advancements. The integration of automated, non-destructive QC methods, such as NMR spectroscopy for liquid biopsies, represents a significant step forward [82]. For the specialized field of organoid biobanking, overcoming cryoinjury through novel CPAs, advanced nanowarming, and microencapsulation techniques is paramount to realizing the vision of "off-the-shelf" living resources for research and therapy [80]. Adherence to the detailed metrics and protocols outlined herein provides a robust foundation for ensuring that biobanked specimens, from simple plasma samples to complex organoids, are of consistently high quality, reproducible, and fully fit for their intended purpose in advancing biomedical science and personalized medicine.
Conclusion
Organoid biobanking represents a paradigm shift in biomedical research, moving from static tissue repositories to dynamic, functional living libraries. Successful implementation requires meticulous optimization of cryopreservation protocols to mitigate cryoinjury while preserving structural and functional integrity. The convergence of novel cryoprotectants, advanced rewarming technologies, and rigorous validation frameworks is paving the way for Next-Generation Living Biobanks. Future directions must focus on standardizing protocols across institutions, improving the preservation of complex tissue microenvironments, and establishing robust clinical correlation data to fully realize the potential of organoid biobanks in personalized medicine and drug development. As these technologies mature, they promise to transform organoids into 'off-the-shelf' resources, ultimately accelerating translational research and clinical application.
References
- 1. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOopgm14D6Y4e5ZD3DyBRz2MEmuiNxO43Faj9JXE8MahxVDhYFwAk]
- 2. Conditional Reprogramming for Patient-Derived Cancer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6912808/]
- 3. Next Generation Biobanking [https://labs.iqvia.com/biofortis-next-generation-biobanking]
- 4. Patient-Derived Organoid Biobanks for Translational ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12387782/]
- 5. Incorporating Cryopreservation Evaluations Into the Design of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9334677/]
- 6. Walking on thin ice: controlled freezing & thawing of ... [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/2677/Walking-on-thin-ice-controlled-freezing-thawing-of-pharmaceutical-active-molec]
- 7. Cell Cryopreservation: Unlocking Regenerative Medicine [https://www.bioprocessintl.com/cell-therapies/unlocking-regenerative-medicine-through-cell-cryopreservation]
- 8. Chemical approaches to cryopreservation [https://www.nature.com/articles/s41570-022-00407-4]
- 9. Decentralized biobanking platform for organoid research ... [https://www.frontiersin.org/journals/blockchain/articles/10.3389/fbloc.2025.1510429/full]
- 10. Establishing a cryopreserved biobank of living tumor ... [https://www.sciencedirect.com/science/article/pii/S2452199X24003955]
- 11. Protocol for the processing, cryopreservation, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11467654/]
- 12. How to Cryopreserve Intestinal Organoids [https://www.stemcell.com/how-to-cryopreserve-intestinal-organoids.html]
- 13. A pipeline for rapid, high-throughput imaging and quantitative ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0332418]
- 14. Protocol for cryopreserving neural organoids and human ... [https://www.sciencedirect.com/science/article/pii/S2666166724006312]
- 15. Tumor organoid biobank-new platform for medical research [https://www.nature.com/articles/s41598-023-29065-2]
- 16. A Living Organoid of Crohn's Disease Patients... | medRxiv Biobank [https://www.medrxiv.org/content/10.1101/2025.04.01.25325058v1]
- 17. Protocol for generation of and high-throughput drug testing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11166870/]
- 18. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOopnE2V_2DOWLURupTsw6a8ySDizI1ms1dHDRpPRzLHwUBVG-zhu]
- 19. ISO/WD 25630-1 - Biotechnology — Biobanking of Organoids [https://www.iso.org/standard/90957.html]
- 20. Introduction to the ISoOR-ISOB & Why This Standard Matters [https://www.linkedin.com/pulse/introduction-isoor-isob-why-standard-matters-isoor-tspwe]
- 21. How the world of biobanking is changing with artificial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12440935/]
- 22. Advancements, Strategies, and Challenges in Organoid- ... [https://www.sciencedirect.com/science/article/pii/S2095809925006083]
- 23. Application of nanomaterials in organoid culture and ... [https://pubs.rsc.org/en/content/articlehtml/2025/na/d5na00534e]
- 24. Breakthroughs and challenges of organoid models for ... [https://www.nature.com/articles/s41420-025-02505-w]
- 25. The current state of biobanking [https://www.ddw-online.com/the-current-state-of-biobanking-36067-202508/]
- 26. Biobanking Market Analysis and Forecast Report 2025-2035 [https://finance.yahoo.com/news/biobanking-market-analysis-forecast-report-082300816.html]
- 27. Comparative effectiveness of vitrification and slow freezing ... [https://bmcwomenshealth.biomedcentral.com/articles/10.1186/s12905-024-03505-1]
- 28. Comparison of the quality of ovarian tissue cryopreservation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11948982/]
- 29. Comparison of the quality of ovarian tissue ... [https://pubmed.ncbi.nlm.nih.gov/40140886/]
- 30. Equivalent outcomes of human oocytes after vitrification or ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11998440/]
- 31. A comparative analysis of vitrification and two slow freezing ... [https://link.springer.com/article/10.1007/s44164-025-00085-8]
- 32. Cryopreservation - Key Survey Findings from the ISCT ... [https://www.isctglobal.org/telegrafthub/blogs/ken-ip1/2025/05/28/cryopreservation-key-survey-findings-from-the-isct]
- 33. Comparison between two cryopreservation techniques of ... [https://www.sciencedirect.com/science/article/pii/S1355814523000391]
- 34. Organoid: Biomedical application, biobanking, and ... [https://www.sciencedirect.com/science/article/pii/S2405844025014094]
- 35. Organoid culture media formulated with growth factors of ... [https://www.nature.com/articles/s41598-019-42604-0]
- 36. Organoids in high-throughput and high-content screenings [https://www.frontiersin.org/journals/chemical-engineering/articles/10.3389/fceng.2023.1120348/full]
- 37. High-throughput solutions in tumor organoids: from culture ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11811636/]
- 38. Patient-Derived Organoid Biobanks for Translational ... [https://pubmed.ncbi.nlm.nih.gov/40863456/]
- 39. of Cryopreservation : Strategies, innovation, and future... organoids [https://pubmed.ncbi.nlm.nih.gov/38403430/]
- 40. Biobanking of human gut organoids for translational research [https://www.nature.com/articles/s12276-021-00606-x]
- 41. Organoid Biobanks: Living Predictive Models for Cancer ... [https://blog.crownbio.com/organoid-biobanks]
- 42. Protocol for Thawing Intestinal Organoids [https://www.stemcell.com/how-to-thaw-intestinal-organoids.html]
- 43. A Practical Guide to Developing and Troubleshooting Patient ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12566426/]
- 44. Establishment of an Organoid Culture Model Derived from ... [https://www.jcpjournal.org/journal/view.html?pn=lastest&uid=965&vmd=Full]
- 45. Application of nanomaterials in organoid culture and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12591057/]
- 46. Digitalized organoids: integrated pipeline for high-speed ... [https://www.nature.com/articles/s41592-025-02685-4]
- 47. Cardiac organoids: a new tool for disease modeling and ... [https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2025.1537730/full]
- 48. Brain Organoids and Assembloids-From Disease Modeling to ... [https://pubmed.ncbi.nlm.nih.gov/40498018/]
- 49. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOooU5Romj9AJwFCgoHU9_-Ssm9oqfkJ910TYhuQEBXnyOIJCNuIe]
- 50. Overcoming ice : cutting-edge materials and advanced strategies for... [https://jnanobiotechnology.biomedcentral.com/articles/10.1186/s12951-025-03265-6]
- 51. Cryopreservation of human kidney organoids - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11335230/]
- 52. and Their Usage in Cryoprotectants ... | IntechOpen Cryopreservation [https://www.intechopen.com/chapters/64165]
- 53. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOopjmUeFot-FBU8ORBFq0WILRxOiBElU7FS-6M4V1RK4-EyWfQUz]
- 54. Current State and Challenges of Tissue and Organ ... [https://www.mdpi.com/1422-0067/25/20/11124]
- 55. Cell Freezing Protocols | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/freezing-cells.html]
- 56. Protocols and Best Practices for Freezing Cells [https://www.stemcell.com/cryopreservation-basics-protocols-and-best-practices-for-freezing-cells]
- 57. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOoqPOBSkS14Yrg39KSpaqJQfXHpq2upJJtAeJJoFPxwsGZzPtS5B]
- 58. Cryopreservation of neuroectoderm on a pillar plate and in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11291134/]
- 59. Optimisation of cryopreservation conditions, including ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11441136/]
- 60. Quantitative and Functional Assessment of the Influence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8837123/]
- 61. Protocol for Immune Cell Isolation, Organoid Generation, ... [https://pubmed.ncbi.nlm.nih.gov/39803320/]
- 62. Protocol for Immune Cell Isolation, Organoid Generation, ... [https://bio-protocol.org/en/bpdetail?id=5157&type=0]
- 63. Cryopreservation as a versatile strategy for the construction ... [http://www.frigidzonemedicine.com.cn/en/article/doi/10.1515/fzm-2025-0013]
- 64. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOopbzwM-LGolcWgvrjo0d-Gs66_-pQI7gYvQNDmvzb6Ol3hKI1_G]
- 65. Impact-induced droplet release thawing enables ... [https://www.sciencedirect.com/science/article/pii/S0011224025003529]
- 66. New Cryopreservation Method Revolutionizes Brain ... [https://www.biobanking.com/new-cryopreservation-method-revolutionizes-brain-tissue-and-organoids-storage/]
- 67. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOorAEvf2dHFsNmhwMvkfDRLx6Tq1mJUMvQnJwO9cgMF2kJa7_3Yg]
- 68. Rise in the use of organoids to replace animal models [https://www.atelerix.co.uk/2025/07/rise-in-the-use-of-organoids-to-replace-animal-models-the-issue-with-storage-and-shipment/?srsltid=AfmBOormMGVAhypw9SLFuzRf68tTjH4NV9TLaoHybEHGi29yxUOF4OF7]
- 69. Organoids | Nature Reviews Methods Primers [https://www.nature.com/articles/s43586-022-00174-y]
- 70. Organoid assessment technologies - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10731122/]
- 71. Biomedical applications of organoids in genetic diseases - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11987506/]
- 72. Human cerebral organoids maintain integrity and viability ... [https://www.sciencedirect.com/science/article/pii/S2949921625000213]
- 73. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOoodFvZEQfzk9cmbVhklUgnDOGzrupZZq8CKyDunXZsgJxPkgnTq]
- 74. Implications for the use of biobanked tissues - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10100012/]
- 75. Drug response in organoids generated from frozen primary ... [https://www.nature.com/articles/srep18889]
- 76. Recovery of glioblastoma cells derived from patients after a ... [https://www.nature.com/articles/s41598-025-22908-0]
- 77. Data Management in Biobanking: Strategies, Challenges, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11417763/]
- 78. Biobanks in the Real World: 5 Uses You'll Actually See ... [https://www.linkedin.com/pulse/biobanks-real-world-5-uses-youll-actually-see-2025-zmmqe/]
- 79. An overview of Synlab SDN Biobank's quality control system [https://www.nature.com/articles/s41598-024-70263-3]
- 80. Cryopreservation as a versatile strategy for the construc... [https://www.degruyterbrill.com/document/doi/10.1515/fzm-2025-0013/html?srsltid=AfmBOopkJMqqUiWY0jbHrACXCk-texc41EJQrkTRfm2tBgnQk3HqQw8n]
- 81. Specimen Quality Control in Biobanking: A Systematic ... [https://www.thermofisher.com/blog/biobanking/specimen-quality-control-in-biobanking-a-systematic-review/]
- 82. BioBank Standardization | QC Process [https://www.bruker.com/en/products-and-solutions/mr/nmr-clinical-research-solutions/b-i-biobank-tool.html]