Organoid Generation from Pluripotent Stem Cells: A 2025 Guide for Advanced Disease Modeling and Drug Development
This article provides a comprehensive overview of the current state and future directions of organoid generation from human pluripotent stem cells (hPSCs).
Organoid Generation from Pluripotent Stem Cells: A 2025 Guide for Advanced Disease Modeling and Drug Development
Abstract
This article provides a comprehensive overview of the current state and future directions of organoid generation from human pluripotent stem cells (hPSCs). Tailored for researchers and drug development professionals, it explores the foundational biology of hPSCs, details advanced differentiation and 3D culture methodologies, and addresses key challenges in standardization and scalability. The content further examines the critical validation of these models against human physiology and their transformative applications in precision medicine, toxicology, and high-throughput drug screening, synthesizing the latest research and technological innovations shaping this rapidly evolving field.
The Biological Blueprint: Understanding Pluripotent Stem Cells and Organoid Self-Organization
Human pluripotent stem cells (hPSCs) represent a cornerstone of modern regenerative medicine and biological research, offering the unique ability to self-renew and differentiate into any cell type of the human body. This category primarily encompasses two distinct but related cell types: human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs). While hESCs are derived from the inner cell mass of pre-implantation blastocysts, hiPSCs are generated through the reprogramming of somatic cells back to a pluripotent state [1] [2] [3]. Understanding the fundamental similarities and differences between these cell types is crucial for selecting the appropriate starting material for organoid generation, disease modeling, and therapeutic development. This article provides a comprehensive comparison of hiPSCs and hESCs, focusing on their molecular profiles, functional characteristics, and practical applications within the context of organoid research.
Molecular and Functional Characterization
Proteomic and Metabolic Landscapes
Recent advances in proteomic technologies have revealed that while hiPSCs and hESCs express a nearly identical set of proteins, they exhibit consistent quantitative differences in protein abundance that have significant functional consequences [1] [4]. A comprehensive proteomic comparison using tandem mass tags (TMT) and MS3-based synchronous precursor selection demonstrated that hiPSCs possess >50% higher total protein content than hESCs while maintaining comparable cell cycle profiles [1]. These differences are often masked by standard median normalization methods in proteomic analyses, highlighting the importance of using absolute quantification approaches like the "proteomic ruler" method [1].
Table 1: Key Proteomic and Metabolic Differences Between hiPSCs and hESCs
| Parameter | hiPSCs | hESCs | Functional Consequences |
|---|---|---|---|
| Total Protein Content | >50% higher [1] | Baseline | Increased biomass and metabolic demand in hiPSCs |
| Mitochondrial Proteins | Significantly increased abundance [1] [4] | Lower levels | Enhanced mitochondrial potential and respiratory capacity |
| Nutrient Transporters | Elevated levels (e.g., glutamine transporters) [1] | Lower levels | Increased nutrient uptake (e.g., glutamine) |
| Metabolic Enzymes | Higher abundance, especially in lipid synthesis pathways [1] | Lower levels | Increased lipid droplet formation |
| Secreted Proteins | Elevated production (ECM components, growth factors) [1] [4] | Lower production | Enhanced paracrine signaling, potential tumorigenic properties |
| Immune Modulators | Higher levels of immune inhibitory proteins [4] | Lower levels | Potential immune evasion capabilities |
The data indicate that reprogramming of human fibroblasts to hiPSCs effectively restores the nuclear proteome to a state similar to hESCs but does not fully restore the profile of cytoplasmic and mitochondrial proteins [1]. This suggests that metabolic and cytoplasmic reprogramming lags behind nuclear reprogramming, resulting in hiPSCs that sustain higher growth rates through enhanced metabolic capacity.
Experimental Workflow for Proteomic Characterization
Protocol: Comparative Proteomic Analysis of hPSCs Using TMT-LC/MS-MS
Principle: This protocol enables quantitative comparison of protein expression between hiPSC and hESC lines using tandem mass tag (TMT) labeling and liquid chromatography-tandem mass spectrometry (LC/MS-MS) [1].
Materials:
- hiPSC and hESC lines (minimum 4 biologically independent lines per group)
- TMT 10-plex kit (Thermo Scientific)
- LC/MS-MS system with Orbitrap mass analyzer
- Synchronous Precursor Selection (SPS) capability
- Cell lysis buffer (8 M urea, 2% SDS, protease inhibitors)
Procedure:
- Cell Culture and Preparation: Maintain all hPSC lines under identical culture conditions (e.g., mTeSR1 medium on Matrigel-coated plates). Passage cells at 70-80% confluence using EDTA dissociation.
- Protein Extraction: Harvest 1×10⁷ cells per line, wash with PBS, and lyse in urea/SDS buffer. Sonicate and centrifuge at 16,000×g for 15 min. Collect supernatant.
- Protein Digestion: Reduce with 5 mM DTT (30 min, 55°C), alkylate with 15 mM iodoacetamide (30 min, dark), and digest with trypsin (1:50 ratio, overnight, 37°C).
- TMT Labeling: Desalt peptides and label with TMT reagents according to manufacturer's instructions. Use a randomized labeling scheme to minimize batch effects.
- LC/MS-MS Analysis: Pool labeled samples and analyze by LC/MS-MS with SPS-MS3 to improve quantification accuracy.
- Data Analysis: Process raw files using Proteome Discoverer or MaxQuant. Normalize using the "proteomic ruler" approach for absolute quantification [1]. Perform statistical analysis (ANOVA with multiple testing correction) to identify differentially expressed proteins.
Technical Notes: Allocate samples to specific isobaric tags to minimize cross-population reporter ion interference. Include at least three technical replicates per line. Validate key findings with orthogonal methods (e.g., Western blot, functional assays).
Figure 1: Experimental workflow for comparative proteomic analysis of hPSCs
hPSCs as Building Blocks for Organoid Generation
Derivation and Characteristics of Organoids
The advent of three-dimensional (3D) organoid culture systems has revolutionized the study of human development and disease by enabling the creation of miniature, simplified versions of organs in vitro [5] [6]. Organoids are 3D cell aggregates that recapitulate the cellular heterogeneity, structure, and functions of human organs to a remarkable degree [6]. Both hiPSCs and hESCs can serve as starting materials for organoid generation, leveraging their capacity to differentiate into cells of all three germ layers [5].
Table 2: Comparison of hiPSC-derived and ASC-derived Organoids
| Characteristic | hiPSC-derived Organoids | ASC-derived Organoids |
|---|---|---|
| Cell Source | Reprogrammed somatic cells (e.g., fibroblasts) [5] | Tissue-specific adult stem cells [7] |
| Developmental Stage | Embryonic/early development [6] | Adult tissue [6] |
| Cellular Complexity | High (multiple cell types, including mesenchymal/epithelial) [6] | Limited (mostly epithelial lineages) [6] |
| Differentiation Time | Prolonged (weeks to months) [7] | Shorter (days to weeks) [6] |
| Genetic Stability | Potential for genetic abnormalities during reprogramming [8] | Higher genetic stability [6] |
| Applications | Early development, genetic disorders, complex diseases [7] | Personalized medicine, drug screening, cancer modeling [7] |
| Tumorigenic Risk | Higher (teratoma formation) [8] | Lower [6] |
hiPSC-derived organoids demonstrate remarkable versatility, with successful generation of brain, retinal, kidney, liver, lung, gastrointestinal, cardiac, and vascularized organoids reported [5] [6]. The derivation process exploits the ability of hiPSCs to undergo self-organized differentiation and spatial patterning when provided with appropriate morphogenetic cues [5].
Protocol for Cerebral Organoid Generation from hPSCs
Principle: This protocol generates 3D cerebral organoids that mimic the developing human brain, containing multiple brain region-specific cell types and exhibiting primitive cortical layering [6].
Materials:
- hiPSC or hESC lines (quality-controlled, mycoplasma-free)
- mTeSR1 or equivalent hPSC maintenance medium
- Matrigel (Corning) or other extracellular matrix
- Neural induction medium (DMEM/F12, N2 supplement, non-essential amino acids)
- Differentiation medium (Neurobasal, B27 supplement, BDNF, GDNF)
- Low-adhesion 6-well plates and U-bottom 96-well plates
- Orbital shaker
Procedure:
- hPSC Maintenance: Culture hPSCs under standard conditions until 70-80% confluent. Ensure cells are in log-phase growth with minimal differentiation.
- Embryoid Body (EB) Formation: Dissociate hPSCs to single cells using Accutase. Seed 3×10⁶ cells per well in low-adhesion 6-well plates in mTeSR1 supplemented with 10 µM Y-27632 (ROCK inhibitor). Culture for 5 days with daily medium changes.
- Neural Induction: Transfer EBs to neural induction medium. Culture for 7 days with medium changes every other day.
- Matrix Embedding: On day 6, individually embed EBs in 10 µL Matrigel droplets. Transfer to 24-well plates and polymerize at 37°C for 30 min.
- Organoid Maturation: Add differentiation medium and culture on orbital shaker (60 rpm) for up to 3 months, with medium changes twice weekly.
- Quality Assessment: Analyze organoids for size uniformity, presence of neural rosettes (by immunostaining for PAX6, SOX2), and cortical layer formation (TBR1, CTIP2) [6].
Technical Notes: The SFEBq (serum-free floating culture of EB-like aggregates with quick aggregation) method enhances reproducibility [6]. For patterned brain region-specific organoids, add specific morphogens (e.g., SHH for ventral patterning, FGF8 for anterior patterning) during days 5-10 of differentiation.
Figure 2: Workflow for cerebral organoid generation from hPSCs
The Scientist's Toolkit: Essential Research Reagents
Successful hPSC culture and organoid generation require carefully selected reagents and materials. The following table summarizes essential solutions for establishing robust hPSC workflows.
Table 3: Essential Research Reagent Solutions for hPSC and Organoid Research
| Reagent Category | Specific Examples | Function & Application |
|---|---|---|
| Reprogramming Factors | OCT4, SOX2, KLF4, c-MYC (OSKM) [3] | Somatic cell reprogramming to generate hiPSCs |
| Reprogramming Delivery | Sendai virus, mRNA transfection, episomal plasmids [8] [9] | Non-integrating methods for clinical-grade hiPSC generation |
| hPSC Maintenance Media | mTeSR1, StemFlex, E8 medium [6] | Defined culture systems for hPSC expansion |
| Extracellular Matrices | Matrigel, Geltrex, laminin-521 [5] [6] | Substrates for hPSC attachment and 3D organoid culture |
| Pluripotency Markers | Antibodies against OCT4, NANOG, SOX2, TRA-1-60 [1] | Quality control of undifferentiated hPSCs |
| Neural Induction | N2 supplement, dual SMAD inhibitors [6] | Directed differentiation toward neural lineages |
| Organoid Maturation | B27 supplement, BDNF, GDNF [6] | Support long-term organoid culture and complexity |
| Gene Editing Tools | CRISPR/Cas9 systems, base editors [8] [9] | Genetic modification for disease modeling and correction |
The comprehensive characterization of hiPSCs and hESCs reveals that while these cell types share core pluripotency networks, they exhibit significant differences in their proteomic, metabolic, and functional properties. The enhanced metabolic capacity and protein content of hiPSCs [1], coupled with their ethical advantages and patient-specific nature [9], make them particularly valuable for organoid generation and disease modeling. However, researchers must remain cognizant of the persistent molecular differences between hiPSCs and the gold standard hESCs when interpreting experimental results.
The ongoing development of safer reprogramming methods [8] [9], improved differentiation protocols [6], and advanced gene editing technologies [8] [9] continues to enhance the utility of both hiPSCs and hESCs as versatile building blocks for organoid research. As these technologies mature, they promise to advance our understanding of human development and disease mechanisms, ultimately accelerating drug discovery and regenerative medicine applications.
The emergence of three-dimensional organoids represents a paradigm shift in our ability to model human development and disease in vitro. At the core of this technology lies the self-organization principle, an innate developmental program whereby stem cells undergo proliferation, differentiation, and spatial reorganization to form complex structures that mirror native organ architecture and function. This process recapitulates key aspects of in vivo organogenesis, including cellular polarization, tissue patterning, and the establishment of functional cellular networks. Framed within pluripotent stem cell research, self-organization provides an unprecedented window into human biology that was previously inaccessible, enabling researchers to move beyond oversimplified two-dimensional cultures and non-human model systems. The following application notes and protocols detail how this principle is harnessed to generate sophisticated tissue models, with a focus on quantitative assessments, standardized methodologies, and the essential tools that empower this transformative research.
Quantitative Profiling of Organoid Model Systems
The capacity of organoids to recapitulate in vivo physiology can be quantitatively assessed through morphological, molecular, and functional analyses. The table below summarizes key characteristics for a selection of advanced self-organizing organoid systems.
Table 1: Quantitative Profiling of Self-Organizing Organoid Systems
| Organoid Type | Key Cell Types Generated | Differentiation Timeline | Functional Metrics | Protocol Variants |
|---|---|---|---|---|
| Neuromusculoskeletal (NMS) Tri-tissue [10] [11] | Spinal motor neurons, skeletal muscle, bone cartilage | Not Specified | Spontaneous contraction; electrophysiological activity; altered activity in response to L-glutamate, NMDA/5-HT, and receptor blockers [11] | Co-development strategy for self-organized neural, muscular, skeletal domains [10] |
| Human Heart Organoids (hHO) [12] [13] | Atrial & ventricular cardiomyocytes, epicardium, endocardium, vascular cells | First contractile activity around day 6; complex tissues by day 15 [12] | Robust, synchronous beating; calcium transients; pharmacological response (e.g., to ondansetron) [12] [13] | Sequential Wnt activation/inhibition; retinoic acid signaling for patterning [12] [13] |
| Cerebral Organoids [14] [15] | Dorsal/ventral telencephalic progenitors, cortical neurons, retinal, choroid plexus identities | 1-2 months for developing cortex and other identities; maintainable >1 year [14] | Not Specified | Unguided (spontaneous) vs. Guided (morphogen-patterned) protocols [14] [15] |
| Human Neural Organoid Cell Atlas (HNOCA) [15] | >1.7 million cells representing diverse neuronal/glial trajectories from 26 protocols | 7 to 450 days in vitro [15] | Transcriptomic fidelity to primary fetal brain references; identification of under-represented primary cell types (e.g., Purkinje cells) [15] | Integrated analysis of 36 datasets and 26 distinct differentiation protocols [15] |
Experimental Protocols for Generating Self-Organizing Organoids
Protocol: Generation of Self-Organized Neuromusculoskeletal Tri-tissue Organoids (hNMSOs)
This protocol describes a co-development strategy to generate complex organoids containing neural, muscular, and skeletal tissues from human pluripotent stem cells (hPSCs) without physical assembly [10] [11].
Key Materials:
- hPSCs: Human embryonic stem cells (hESCs) or induced pluripotent stem cells (iPSCs).
- Basement Membrane Matrix: Matrigel or similar ECM for 3D embedding.
- Differentiation Media: Sequential media formulations to direct tri-lineage specification.
- Maestro MEA Platform: For non-invasive electrophysiological monitoring of functional neuromuscular circuits [11].
Methodology:
- Initial Aggregation: Dissociate hPSCs to single cells and aggregate them in ultra-low attachment 96-well plates to form embryoid bodies.
- Neural Induction: Transfer aggregates to neural induction medium. The protocol relies on a self-patterning approach where neural ectoderm uniformly forms on the aggregate's outer surface.
- Matrix Embedding and Maturation: Embed the neural-induced aggregates in a droplet of Basement Membrane Matrix to provide structural support. Transfer the embedded constructs to a differentiation medium that supports the concurrent development of neural, muscular, and skeletal lineages.
- Functional Validation:
- Immunostaining: Confirm the presence and spatial organization of distinct tissue domains using antibodies against lineage-specific markers (e.g., neuronal, muscle, and cartilage markers).
- Electrophysiology: Use a multi-electrode array (MEA) system like the Maestro to record spontaneous electrical activity and probe functional neuromuscular connectivity by applying neurotransmitters (e.g., L-glutamate) and receptor blockers [11].
- Calcium Imaging: Use Fluo-4 AM or similar dyes to visualize calcium flux in response to photostimulation, confirming the presence of functional connections.
The workflow for this self-organization process is outlined below.
Protocol: Generating Self-Assembling Human Heart Organoids (hHOs)
This protocol generates highly complex, patterned heart organoids through the self-organization of hPSCs using sequential Wnt pathway modulation [12] [13].
Key Materials:
- hPSCs: Maintained in feeder-free conditions (e.g., Essential 8 Flex Medium).
- Small Molecule Inhibitors/Activators: CHIR99021 (WNT activator), IWP2 (WNT inhibitor), Thiazovivin (ROCK inhibitor).
- Specialized Plates: 96-well round-bottom ultra-low attachment plates for suspension culture.
- Differentiation Media: RPMI 1640 supplemented with B-27 (with and without insulin).
Methodology:
- hPSC Culture: Maintain hPSCs in a pluripotent state for at least two passages before differentiation.
- Embryoid Body (EB) Formation: Dissociate hPSCs to single cells and seed them into ultra-low attachment 96-well plates to form uniform EBs by centrifugation.
- Cardiac Mesoderm Induction (Day 0): Activate the Wnt signaling pathway by treating EBs with CHIR99021 in RPMI/B-27 minus insulin medium.
- Cardiac Lineage Specification (Day 3): Inhibit Wnt signaling by adding IWP2 to direct cells toward definitive cardiac lineages.
- Proepicardial Induction and Maturation (Day 5): A second phase of Wnt activation is applied to induce proepicardial tissues. Subsequently, organoids are transferred to RPMI/B-27 complete medium for long-term maturation and self-organization.
- Analysis:
- Immunofluorescence: Analyze the organoids for the presence of atrial (MYL7) and ventricular (MYL2) cardiomyocytes, endocardial cells (PECAM1), and epicardial cells (WT1) to confirm spatial patterning [12] [13].
- Functional Assays: Record beating activity and characterize calcium transients using dyes like Fluo-4 AM.
- Disease Modeling: Test drug effects, such as the impact of ondansetron on ventricular cardiomyocyte development, to model congenital heart defects [13].
The critical signaling pathway manipulations are summarized in the following diagram.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful organoid generation is dependent on a foundational set of reagents and tools that support the self-organization process. The following table details key solutions used in the featured protocols.
Table 2: Essential Research Reagent Solutions for Organoid Generation
| Reagent/Tool | Function | Example Application |
|---|---|---|
| Basement Membrane Extract (e.g., Matrigel) | Provides a 3D extracellular matrix scaffold that supports polarized epithelial bud formation, structural integrity, and morphogenesis. | Essential for neuroepithelial expansion in cerebral organoids and for embedding hNMSOs [10] [14]. |
| Small Molecule Pathway Modulators | Precisely controls key developmental signaling pathways (e.g., WNT, BMP, TGF-β) in a time-dependent manner to direct cell fate. | CHIR99021 (GSK3β inhibitor) and IWP2 (WNT inhibitor) are used sequentially for heart organoid patterning [12]. |
| Pluripotent Stem Cell Media | Maintains hPSCs in a proliferative, undifferentiated state prior to the initiation of differentiation protocols. | Essential 8 Flex Medium is used for the culture of hPSCs for heart organoid generation [12]. |
| Specialized Cultureware | Enables the formation and maintenance of 3D aggregates. Ultra-low attachment surfaces prevent cell adhesion, forcing self-assembly. | 96-well round-bottom ultra-low attachment plates are critical for generating uniform EBs for heart and NMS organoids [10] [12]. |
| Defined Differentiation Media Kits | Provides a consistent, serum-free base medium supplemented with factors that promote the survival and maturation of specific cell lineages. | B-27 Supplement is a key component in neural and cardiac differentiation media [12] [14]. |
| Multi-Electrode Array (MEA) Systems | Allows for non-invasive, long-term monitoring of extracellular field potentials and spontaneous electrical activity in functional tissues. | The Maestro MEA platform is used to confirm functional neuromuscular circuits in hNMSOs [11]. |
The development of intestinal organoid technology represents a fundamental paradigm shift in biomedical research, moving from traditional two-dimensional (2D) cell cultures to complex three-dimensional (3D) models that faithfully recapitulate intestinal physiology. This breakthrough has its roots in 2009, when the Clevers laboratory established the first self-renewing, non-transformed mini-gut organoid culture from adult intestinal stem cells [16] [17]. This landmark achievement demonstrated that Lgr5+ intestinal stem cells could expand in vitro when provided with appropriate niche factors, specifically Epithelial Growth Factor (EGF), R-Spondin, and Noggin, thereby breaking the previous dependence on mesenchymal fibroblasts for propagation [18] [16]. The subsequent adaptation of this system for human intestinal tissue, combined with parallel advances in pluripotent stem cell (PSC) differentiation, has created unprecedented opportunities for studying human intestinal development, disease mechanisms, and therapeutic interventions [17].
These 3D miniaturized versions of the human intestinal epithelium conserve parental gene expression and mutation characteristics while maintaining the function and biological features of native tissue over extended periods in culture [18]. Unlike traditional 2D cultures where isolated tissue cells gradually lose their morphology and become flattened, organoids preserve the crypt-villus organization, epithelial polarization, and functional lumen that defines intestinal physiology [18] [19]. The rapid adoption of organoid technology across basic and clinical research domains underscores its transformative potential, particularly as researchers seek more physiologically relevant models that bridge the gap between conventional cell lines and in vivo experimentation [20].
Fundamental Milestones in Intestinal Organoid Development
Establishment of the Core Culture System
The foundational milestone in intestinal organoid technology was the development of a defined culture system that recapitulates the intestinal stem cell niche. This system employs a basement membrane extract (such as Matrigel) to provide structural support and key signaling molecules to maintain stemness and drive differentiation [16] [17]. The essential growth factors include:
- EGF (Epithelial Growth Factor): Supports epithelial cell survival and long-term culture [16].
- R-Spondin: Potentiates Wnt signaling by binding to the Lgr5 receptor, essential for maintaining intestinal stem cells [16] [17].
- Noggin: A BMP antagonist that prevents differentiation and maintains stem cell proliferation [16].
Subsequent adaptations for human intestinal organoids revealed important species-specific requirements, including the need for exogenous Wnt-3A, p38 inhibitors to suppress secretory lineage differentiation, and TGF-β inhibitors to permit WNT-driven proliferation [16]. These refinements enabled the establishment of robust, long-term cultures from human small intestine (enteroids) and colon (colonoids) [16] [17].
Progression from Epithelial to Multi-Tissue Systems
Recent advances have pushed beyond epithelial-only organoids to create complex multi-tissue systems. A landmark 2025 study demonstrated the derivation of human intestinal organoids with functional tissue-resident macrophages entirely from pluripotent stem cells [21]. This was achieved by generating HIOs and macrophages separately through directed differentiation of human PSCs, then combining them in vitro. Following two weeks of coculture, macrophages incorporated into the developing HIOs and persisted with a transcriptional signature resembling human fetal intestinal macrophages [21]. These incorporated macrophages demonstrated functional capacity for bacterial phagocytosis and inflammatory cytokine production in response to lipopolysaccharide, establishing a powerful new model for investigating inflammatory bowel disease mechanisms [21].
Table 1: Key Milestones in Intestinal Organoid Technology Development
| Year | Milestone Achievement | Significance | Reference |
|---|---|---|---|
| 2009 | First establishment of self-renewing intestinal organoids from mouse Lgr5+ stem cells | Broke dependence on mesenchymal niche; enabled long-term epithelial culture | [18] [16] [17] |
| 2011 | Generation of intestinal tissue from human pluripotent stem cells | Created model for human intestinal development | [17] [21] |
| 2014 | Development of human small intestine model using PSCs | Established patient-specific intestinal models without biopsy | [21] |
| 2017 | Adaptation for human colonic organoids (colonoids) | Expanded technology to model colonic physiology and disease | [16] |
| 2025 | Incorporation of functional tissue-resident macrophages into HIOs from PSCs | Created multi-tissue system with immune components for inflammation studies | [21] |
Experimental Protocols: From Stem Cells to Complex Organoids
Basic Protocol: Establishment of Human Intestinal Organoids from Pluripotent Stem Cells
The derivation of intestinal organoids from human pluripotent stem cells (hPSCs) follows a directed differentiation approach that mimics intestinal development [22] [17]:
Initial Differentiation to Definitive Endoderm (Days 1-4):
- Culture hPSCs in monolayer format with defined medium containing Activin A to induce definitive endoderm differentiation.
- Monitor for the emergence of CXCR4+ and c-KIT- cells indicating successful endoderm specification.
- Critical Step: Precise timing and growth factor concentration are essential for efficient endoderm formation.
Hindgut Specification and 3D Structure Formation (Days 5-14):
- Transition cells to medium containing FGF4 and WNT3A to promote hindgut specification.
- Spontaneous formation of 3D hindgut spheroids occurs typically between days 8-14.
- Isolate spheroids from culture supernatant for embedding in Matrigel.
- Technical Note: Spheroid size should be monitored; optimal diameter ranges from 100-300μm for uniform differentiation.
Intestinal Organoid Maturation (Days 15-30+):
- Embed hindgut spheroids in Matrigel domes and culture with intestinal growth medium.
- Base medium typically includes EGF, R-Spondin, Noggin, and WNT3A.
- Organoids develop crypt-like buds and intestinal epithelial characteristics within 7-14 days post-embedding.
- For enhanced maturation, consider extended culture (60+ days) or in vivo transplantation [17].
Advanced Protocol: Incorporating Tissue-Resident Macrophages
The generation of HIOs with functional macrophages requires separate differentiation of both lineages followed by integration [21]:
Macrophage Differentiation from hPSCs (21-28 days):
- Direct hPSCs through primitive hematopoiesis using defined cytokines (BMP4, VEGF, SCF, FLT3L).
- Collect hematopoietic progenitors and differentiate into macrophages using M-CSF.
- Validate macrophage identity by flow cytometry (CD14+, CD16+, CD115+, CD163+).
Integration with Intestinal Organoids:
- Gently dissociate HIOs to create small fragments while preserving 3D structure.
- Combine with macrophages at optimized ratio (recommended starting ratio: 1:10 macrophages to intestinal cells).
- Culture in modified intestinal organoid medium with reduced levels of inflammatory cytokines.
- Allow 14 days for macrophage incorporation and adaptation to tissue-resident phenotype.
- Quality Control: Assess macrophage incorporation by confocal microscopy and transcriptional profiling for tissue-resident markers.
Signaling Pathways Governing Organoid Development and Homeostasis
The development and maintenance of intestinal organoids requires precise recapitulation of key signaling pathways that define the intestinal stem cell niche. The following diagram illustrates the core signaling networks that must be activated or inhibited to successfully establish and maintain intestinal organoids:
This network diagram illustrates how the balance between Wnt/R-spondin signaling (activated for stem cell maintenance) and BMP signaling (inhibited by Noggin) controls the equilibrium between stem cell self-renewal and differentiation in intestinal organoids. EGF signaling independently promotes general epithelial cell proliferation, working synergistically with Wnt activation.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful intestinal organoid culture requires precise combination of extracellular matrix support, defined growth factors, and signaling modulators. The following table details essential components and their specific functions in organoid establishment and maintenance:
Table 2: Essential Research Reagents for Intestinal Organoid Culture
| Reagent Category | Specific Components | Function in Organoid Culture | Application Notes |
|---|---|---|---|
| Extracellular Matrix | Matrigel, Basement Membrane Extract | Provides structural support mimicking intestinal stroma; prevents anoikis | Protein concentration typically 8-12 mg/mL; keep liquid at 4°C during handling |
| Core Growth Factors | EGF, R-Spondin, Noggin | Mimics crypt niche signaling; maintains stem cell population | Human organoids require higher R-Spondin concentrations than mouse |
| Wnt Pathway Modulators | Wnt3A conditioned medium, GSK3β inhibitors | Potentiates Wnt signaling essential for Lgr5+ stem cells | Human organoids require exogenous Wnt; can use conditioned medium or recombinant protein |
| Signaling Inhibitors | p38 inhibitors (SB202190), TGF-β inhibitors (A83-01, SB431542) | Suppresses secretory differentiation; enhances proliferation | Particularly critical for human small intestinal organoid culture |
| Additional Maturation Factors | Prostaglandin E2, Nicotinamide | Enhances colonoid growth and maturation | Especially important for human colonic organoid culture |
Quantitative Assessment of Organoid Development and Maturation
Systematic evaluation of organoid quality and maturation state is essential for experimental reproducibility. Recent advances in quantitative profiling, particularly single-cell RNA sequencing, have enabled rigorous assessment of protocol efficiency and cellular diversity. A 2025 study of brain organoids introduced the NEST-Score as a quantitative metric to evaluate cell-line- and protocol-driven differentiation propensities, establishing a framework that could be adapted for intestinal organoid validation [23] [24]. Although developed for neural systems, this approach demonstrates the importance of standardized quantitative assessment in organoid technology.
Table 3: Quantitative Parameters for Intestinal Organoid Characterization
| Parameter Category | Specific Metrics | Typical Values/Ranges | Assessment Methods |
|---|---|---|---|
| Growth Dynamics | Organoid formation efficiency | 60-80% from crypts; 30-50% from single cells | Light microscopy, quantification of embedded structures |
| Cellular Composition | Stem cell percentage (Lgr5+ Olfm4+) | 5-15% of total epithelial cells | Flow cytometry, immunofluorescence, scRNA-seq |
| Differentiation Markers | Enterocyte (Villin+), Goblet (Muc2+), Enteroendocrine (Chromogranin A+) | Villin+: 60-70%; Muc2+: 10-15%; Chromogranin A+: 1-5% | Immunofluorescence, qPCR |
| Functional Maturation | CYP3A activity, P-gp transport, barrier formation (TEER) | CYP3A activity: 2-5 fold increase with maturation | Enzymatic assays, transporter studies, electrophysiology |
| Multilineage Capacity | Presence of all major epithelial lineages | ≥4 lineages: enterocytes, goblet, enteroendocrine, Paneth cells | scRNA-seq, immunohistochemistry |
Applications in Disease Modeling and Drug Development
Intestinal organoids have transformed pharmaceutical research by providing human-relevant models that improve predictive accuracy for drug efficacy and safety. Patient-derived organoids (PDOs) retain individual-specific genetic and phenotypic features, enabling personalized approaches to treatment selection and drug development [20]. In oncology, patient-derived tumor organoids (PDTOs) maintain histological and genomic features of original tumors, including intratumoral heterogeneity and drug resistance patterns [20]. These have been successfully deployed for medium-throughput drug screening to inform treatment decisions in colorectal, pancreatic, and lung cancers [20].
For infectious disease modeling, intestinal organoids provide superior platforms for studying host-pathogen interactions compared to traditional immortalized cell lines. They have been utilized to examine the pathologic effects of Clostridium difficile toxins on intestinal epithelium and to model rotavirus infection, where they support the complete viral life cycle with greater fidelity than previous systems [16]. The development of co-culture systems incorporating immune cells, such as the recently established macrophage-containing HIOs, further enhances the utility of these models for inflammatory bowel disease research and host-microbe interaction studies [21].
The evolution of intestinal organoids from simple epithelial structures to complex multi-tissue systems represents a remarkable scientific achievement that continues to transform gastrointestinal research. Current efforts focus on enhancing organoid complexity through incorporation of additional cell types, including neurons of the enteric nervous system, vascular components, and diverse immune populations [17] [21]. The integration of organoids with microfluidic organ-on-chip technologies promises to further improve physiological relevance by introducing mechanical forces, flow dynamics, and spatial organization that better mimic the in vivo intestine [20].
While challenges remain in standardization, scalability, and complete functional maturation, the rapid pace of innovation in intestinal organoid technology suggests these limitations will be addressed through interdisciplinary collaboration. The continued refinement of these models will undoubtedly accelerate drug discovery, enable personalized therapeutic approaches, and deepen our understanding of human intestinal development and disease pathogenesis. As these technologies mature, they will increasingly serve as bridges between traditional preclinical models and human clinical trials, potentially reducing both drug development costs and reliance on animal testing.
The advent of three-dimensional (3D) organoid systems represents a paradigm shift in biomedical research, providing an unprecedented in vitro platform that closely mimics human physiology. Organoids are 3D miniature structures cultured in vitro from human pluripotent stem cells (hPSCs), including induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs), or adult stem cells (ASCs) derived from healthy individuals and patients [18] [6]. These systems self-organize and differentiate into 3D cell masses that recapitulate the cellular heterogeneity, architecture, and specific functions of their in vivo counterparts [18]. Unlike traditional two-dimensional (2D) cultures where cells gradually lose their original morphology and hierarchical structure, organoids conserve parental gene expression and mutation characteristics while maintaining long-term function and biological stability in vitro [18]. This technological advancement bridges the critical gap between conventional cell culture models and human pathophysiology, offering exceptional prospects for modeling human genetic diversity, disease mechanisms, and therapeutic interventions in a laboratory setting [25].
Core Advantage 1: Faithful Recapitulation of Human Physiology
Structural and Functional Complexity
Organoid technology fundamentally surpasses the limitations of traditional 2D cultures by reproducing organ-specific microenvironments with remarkable fidelity. Cells cultured in 2D systems tend to become flattened and lose their original morphology, affecting cell-cell interactions and signal transduction [18]. In contrast, the 3D organoid culture system mimics the physicochemical microenvironments and cell-extracellular matrix interactions present in vivo, enabling cells to maintain genetic stability and chromatin heterogeneity while exhibiting specific cellular functions [18]. This preservation of structural integrity is crucial for modeling organ functionality, as demonstrated by various organoid systems:
- Brain organoids can form cortical-like structures with apical-basal polarity, generating cortical progenitors and functional neurons that mimic the six-layer structure of cortical tissue [6].
- Thymic organoids recently developed from iPSCs recapitulate thymic epithelial development, producing FOXN1+ TEC progenitor-like cells and diverse mature MHCII+ populations resembling cortical and medullary thymic epithelial cells (TECs) [26].
- Intestinal organoids exhibit crypt-villus structures containing stem cells, progenitor cells, and terminally differentiated cell types, including enterocytes, goblet cells, Paneth cells, and neuroendocrine cells [27].
Signaling Pathway Fidelity
The physiological relevance of organoids extends to their ability to recapitulate complex signaling pathways that direct organ development and function. The directed differentiation of PSCs into organoids relies on precisely manipulating developmental signaling pathways—including WNT, BMP, TGF-β, FGF, and retinoic acid—to establish proper regional identity [18]. For instance, in the generation of thymic organoids, controlled retinoic acid signaling directly promotes HOXA3 expression, specifying the positional identity of the third pharyngeal pouch from which the thymus arises [26]. Similarly, brain organoid patterning requires careful modulation of WNT and TGF-β signaling to establish dorsal-ventral and anterior-posterior axes resembling the developing neural tube [6].
Table 1: Quantitative Profiling of Brain Organoid Cell Diversity Across Protocols
| Organoid Protocol Type | Cell Types Identified | Similarity to In Vivo Counterparts | Key Patterning Factors |
|---|---|---|---|
| Dorsal Forebrain Organoids | Neural progenitors, glutamatergic neurons, astrocytes, oligodendrocytes | Recapitulates 6-layer cortical structure | WNT inhibition, TGF-β inhibition |
| Ventral Forebrain Organoids | GABAergic neurons, medial ganglionic eminence progenitors | Mimics developing striatum | SHH activation, WNT modulation |
| Midbrain Organoids | Dopaminergic neurons, substant nigra-like cells | Expresses characteristic markers (TH, FOXA2) | FGF8, SHH activation |
| Thalamic Organoids | Glutamatergic thalamic projection neurons | Recapitulates thalamic nuclear organization | WNT activation, SHH modulation |
Core Advantage 2: Modeling Human Genetic Diversity
Addressing Ancestral Diversity in Biomedical Research
The imperative for genetically diverse experimental models represents a critical advancement in organoid technology. Despite tremendous growth in human genetics, a well-documented issue persists: the inadequate representation of samples with non-European ancestries [28]. In genome-wide association studies (GWAS), approximately 95.76% of participants are of European ancestry, with those of Asian, African, and Hispanic/Latin American ancestries collectively representing less than 5% [28]. This disparity produces polygenic risk scores that are less effective in underrepresented populations and can exacerbate clinical misdiagnosis, thereby deepening existing health care disparities [28]. Organoid technology directly addresses this challenge through:
- Diverse iPSC Biobanks: Establishing iPSC collections from donors of diverse genetic backgrounds, ancestries, and geographical origins [28].
- Population-Specific Disease Modeling: Generating organoids that capture population-specific disease susceptibilities and drug responses, such as the increased Warfarin sensitivity in individuals of North Asian ancestry [28].
- HLA-Matched Therapeutic Development: Creating iPSC libraries homozygous for human leukocyte antigen (HLA) haplotypes common in specific populations to facilitate future cell replacement therapies with reduced immune rejection [28].
Genetic Diversity for Enhanced Discovery
Incorporating genetic diversity into organoid models accelerates biological discovery through multiple mechanisms. Populations of African ancestry, for instance, possess more genetic variation with shorter haploblocks and less linkage disequilibrium, allowing causal alleles to be mapped at higher spatial resolution [28]. Furthermore, certain disease-associated risk variants demonstrate significant population-specific prevalence, such as the COVID-19 respiratory failure risk variant present in 60% of individuals of South Asian ancestry compared to 15% of those of European ancestry [28]. Studies confirm that genetic differences between individual donors affect most cellular traits in iPSCs and their differentiated progeny, including differentiation capacity, cellular morphology, and drug responses [28]. The strategic inclusion of diverse genetic backgrounds in organoid studies thus enables:
- Identification of genetic variants underlying specific traits and disorders with enhanced resolution
- Functional characterization of population-specific risk variants
- Development of more effective and equitable therapeutic strategies
- Improved understanding of how genetic background influences disease presentation and treatment response
Table 2: Applications of Genetically Diverse Organoid Models in Disease Research
| Disease Category | Specific Condition | Genetic Background | Research Application |
|---|---|---|---|
| Neurological Disorders | Primary microcephaly | Gene-edited iPSCs carrying CPAP/CENPJ mutations [25] | Study of neurodevelopmental defects |
| Hereditary Syndromes | DiGeorge syndrome (22q11.2 deletion) | Patient-derived iPSCs with 22q11.2 deletion [25] | Modeling neural crest migration |
| Liver Diseases | α1-antitrypsin deficiency | Patient-derived organoids [25] | Pathophysiology and drug testing |
| Retinal Disorders | Retinitis pigmentosa | Patient-derived iPSCs with RP2 or USH2A mutations [25] | Disease mechanisms and gene therapy |
Applications in Disease Modeling and Drug Development
Genetic Disease Modeling
Organoids have revolutionized the study of genetic disorders by providing human-specific models that faithfully recapitulate disease pathophysiology. The integration of organoid technology with CRISPR-Cas9 gene editing enables precise introduction of patient-specific genetic mutations while maintaining isogenic controls [27] [25]. This approach allows researchers to systematically investigate genotype-phenotype relationships in a controlled experimental environment. Notable applications include:
- Neurological Disorders: Brain organoids model conditions including primary microcephaly, Rett syndrome, AUTS2 syndrome, tuberous sclerosis complex, and Down syndrome, revealing disease-specific alterations in neurogenesis, neuronal migration, and synaptic function [25].
- Hereditary Metabolic Diseases: Liver organoids from patients with α1-antitrypsin deficiency accumulate A1AT protein, mirroring the clinical and pathophysiological characteristics of the disease [25].
- Multisystem Genetic Syndromes: Thymic organoids enable study of thymic disorders associated with immunodeficiencies and autoimmunity, which have been challenging to model in vitro [26].
Drug Screening and Personalized Medicine
Organoids provide powerful platforms for drug discovery and personalized therapeutic testing. Patient-derived organoids (PDOs) maintain the genetic fingerprint of their tissue of origin, making them ideal for predicting individual drug responses [18] [29]. This application is particularly valuable in oncology, where tumor organoids constructed from preoperative biopsies or tumor resections play important roles in individualized drug sensitivity prediction [18]. The utility of organoids in pharmaceutical applications includes:
- High-Throughput Compound Screening: Intestinal organoids from cystic fibrosis patients quantify CFTR function to identify and validate CFTR-modulating drugs [27] [29].
- Pharmacogenomics: Hepatocyte organoids from diverse genetic backgrounds assess population-specific variability in drug metabolism and toxicity [28].
- Cancer Immunotherapy: Co-culture of epithelial tumor organoids with peripheral blood lymphocytes expands tumor-reactive T-cells and characterizes sensitivity to T cell-mediated killing at a personalized level [27].
- Precision Medicine: Organoid biobanks derived from multiple patients enable in vitro drug sensitivity testing before treatment administration, directing care and predicting therapeutic outcomes for cancer and genetic disease patients [29].
Experimental Protocols
Protocol 1: Cerebral Organoid Generation from Human PSCs
Workflow Overview: This protocol generates whole-brain organoids with diverse regional identities through minimal exogenous patterning [30].
Materials:
- Human PSCs (iPSCs or ESCs)
- Matrigel or other extracellular matrix
- Neural induction medium
- Differentiation medium
- Growth factors (BDNF, GDNF, etc.)
Procedure:
- Embryoid Body (EB) Formation: Harvest PSCs and transfer to low-attachment plates in PSC medium to form EBs (Day 0).
- Neural Induction: Change to neural induction medium containing SMAD inhibitors (Days 1-7).
- Matrix Embedding: Transfer EBs to Matrigel droplets (Day 7).
- Organoid Maturation: Culture in differentiation medium with BDNF, GDNF, TGF-β, and cAMP on orbital shaker (Days 7-90).
- Media Refreshing: Partially replace medium every 3-4 days.
Key Considerations:
- EB size uniformity affects reproducibility
- Matrix concentration influences nutrient penetration
- Orbital shaking enhances viability and reduces central necrosis
Protocol 2: Thymic Epithelial Organoid Differentiation from iPSCs
Workflow Overview: This recently published protocol generates thymic epithelial cells through retinoic acid-based endodermal patterning [26].
Materials:
- Human iPSCs
- Definitive endoderm induction medium
- Anterior foregut endoderm (AFE) medium
- Pharyngeal endoderm (PE) medium
- Retinoic acid, FGF8
Procedure:
- Definitive Endoderm Induction: Culture iPSCs in definitive endoderm induction medium (Days 0-3).
- Anterior Foregut Patterning: Switch to AFE medium with BMP, TGF-β, and WNT inhibitors (Days 3-7).
- Pharyngeal Endoderm Specification: Transfer to PE medium with retinoic acid and FGF8 (Days 7-18).
- Self-Directed Differentiation: Maintain in basal medium without exogenous patterning factors (Days 18-28).
- Maturation and Analysis: Characterize FOXN1 expression and TEC markers (Day 28+).
Key Considerations:
- Retinoic acid concentration critically specifies 3rd pharyngeal pouch identity
- FGF8 enhances PE marker expression and FOXN1 induction
- WNT activation after day 18 abolishes FOXN1 expression
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for Organoid Culture
| Reagent Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| Stem Cell Sources | iPSCs, ESCs, ASCs | Self-renewal and differentiation potential | iPSCs avoid ethical concerns; ASCs yield more mature organoids [18] [6] |
| Extracellular Matrices | Matrigel, Collagen, Synthetic hydrogels | Provide 3D structural support | Matrigel most common but batch variability concerns [29] |
| Patterning Molecules | Retinoic acid, BMP4, FGFs, WNT agonists/antagonists | Direct regional specification | Concentration and timing critically affect identity [26] |
| Media Supplements | N2, B27, B27 without vitamin A | Support survival and differentiation | Vitamin A exclusion important for neural patterning [30] |
| Gene Editing Tools | CRISPR-Cas9, PiggyBac transposons | Introduce disease mutations or reporters | Non-integrating methods preferred for clinical applications [27] |
Visualization of Signaling Pathways and Workflows
Signaling Pathway Diagram: Thymic Organoid Differentiation
Diagram Title: Signaling Pathway for Thymic Organoid Differentiation
Experimental Workflow Diagram: Cerebral Organoid Generation
Diagram Title: Cerebral Organoid Generation Workflow
Organoid technology represents a transformative advancement in biomedical research by uniquely recapitulating human physiology and genetic diversity in vitro. These 3D models faithfully mimic the structural complexity, cellular heterogeneity, and functional characteristics of human organs while encompassing the genetic variability of human populations. The core advantages of organoids—including their physiological relevance, genetic stability, and compatibility with genome editing—position them as indispensable tools for modeling human development, disease pathogenesis, and therapeutic interventions. As the field advances, addressing current challenges related to reproducibility, maturation, and cellular complexity will further enhance the utility of organoid systems. The integration of organoid technology with cutting-edge genomic, bioengineering, and analytical approaches promises to accelerate biological discovery and pave the way for personalized regenerative medicine applications.
From Cells to Complex Systems: Protocols, Advanced Co-Cultures, and Pharmaceutical Applications
The generation of brain organoids from pluripotent stem cells represents a paradigm shift in modeling human brain development and disease. These three-dimensional (3D) structures self-organize to recapitulate aspects of the complex cellular diversity and spatial architecture of the developing human brain [30]. The core protocol hinges on the precise interplay of three fundamental components: soluble patterning factors that direct regional cell fate, extracellular matrix (ECM) scaffolds that provide structural and biochemical support, and defined differentiation pathways that guide the transition from pluripotency to specialized neural lineages [31]. This protocol breakdown details the essential elements required for the successful generation of region-specific brain organoids, providing a standardized yet adaptable framework for researchers in developmental biology and drug discovery.
The Scientist's Toolkit: Essential Research Reagents
Successful brain organoid generation requires a suite of specialized reagents and equipment. The table below catalogs the core components of the protocol, each playing a critical role in directing stem cell fate and supporting 3D tissue development.
Table 1: Key Research Reagent Solutions for Brain Organoid Generation
| Reagent Category | Specific Examples | Primary Function |
|---|---|---|
| Patterning Factors | SB431542 (TGF-β inhibitor), LDN193189 (BMP inhibitor), CHIR99021 (Wnt agonist), Purmorphamine (Shh agonist) | Instructs regional neural fate by modulating key developmental signaling pathways [30] [32]. |
| Basal Media | Neural Induction Medium (NIM), Serum-free Culture Media | Provides the foundational nutrients and environment for neural differentiation and tissue growth [33] [30]. |
| Media Supplements | B-27 Supplement (with/without Vitamin A), N-2 Supplement, Ascorbic Acid, Brain-Derived Neurotrophic Factor (BDNF) | Supports neural progenitor survival, maturation, and long-term health of neuronal cultures [30] [32]. |
| Extracellular Matrix | Matrigel, Laminin-rich ECMs | Acts as a scaffold to support epithelial polarization, lumen formation, and tissue morphogenesis [33] [31]. |
| Specialized Equipment | SpinΩ Bioreactor, Ultra-Low Attachment Plates, Light-Sheet Microscope | Enables suspension culture, improves nutrient diffusion, and allows long-term live imaging [33] [32]. |
| Cell Lines | Fluorescently-tagged iPSC lines (e.g., membrane-CAAX, actin-ACTB, nucleus-H2BJ) | Facilitates live-cell tracking and segmentation of cellular and subcellular dynamics [33]. |
Quantitative Breakdown of Soluble Patterning Factors
Soluble factors, including small molecule inhibitors and agonists, are precisely timed to mimic the signaling gradients of the embryonic environment. They direct the differentiation of pluripotent stem cells toward specific regional neural fates. The following table summarizes the key factors used to generate forebrain, midbrain, and hypothalamic organoids.
Table 2: Soluble Factors for Regional Brain Organoid Patterning
| Brain Region | Key Patterning Factors | Protocol Timing | Targeted Signaling Pathways |
|---|---|---|---|
| Forebrain | Dual-SMAD inhibition (SB431542, LDN193189) [30] [32] | Early neural induction (Days 1-7) | Inhibits TGF-β and BMP signaling to promote default neuroectoderm fate [32]. |
| Midbrain | SHH Agonist (Purmorphamine/SAG), FGF8b, CHIR99021 [30] | Patterning phase (e.g., Days 7-28) | Activates Shh and Wnt signaling to induce midbrain dopaminergic progenitor identity [30]. |
| Hypothalamus | SHH Agonist (high concentration), BMP, WNT inhibitors [32] | Patterning phase | Activates strong Shh signaling while modulating other pathways to specify ventral diencephalon fates [32]. |
| General Patterning | Retinoic Acid (RA), BMP4, FGFs, WNTs [31] | Variable, depending on target region | Establishes anteroposterior and dorsoventral axes in a concentration-dependent manner [31]. |
The Role of Extracellular Matrix Scaffolds
The ECM is not a passive scaffold but an active instructor of morphogenesis. Natural matrices like Matrigel, a laminin-rich basement membrane extract, are critical for establishing tissue polarity and enabling complex structural organization.
- Function in Neuroepithelial Formation: Embedding embryoid bodies in Matrigel droplets is a critical step in many protocols, as it promotes the formation and expansion of a polarized neuroepithelium surrounding large luminal regions [33]. This process is essential for creating the ventricle-like structures that characterize brain organoids.
- Mechanosensing and Patterning: Research shows that the ECM enhances lumen expansion and influences global tissue patterning. For instance, the presence of an extrinsic matrix promotes telencephalon formation, while its absence can lead to altered morphologies and caudalized tissue identities. This mechanosensing is linked to YAP and WNT signaling pathways, with matrix-induced regional guidance involving the WNT ligand secretion mediator (WLS) [33].
- Customization: While Matrigel is widely used, it is not fully defined, leading to batch variability. Innovative bioengineering approaches are developing synthetic, customizable hydrogel matrices to fine-control critical biophysical cues such as stiffness and degradability for more reproducible organoid generation [31].
Stage-by-Stage Experimental Protocol & Workflow
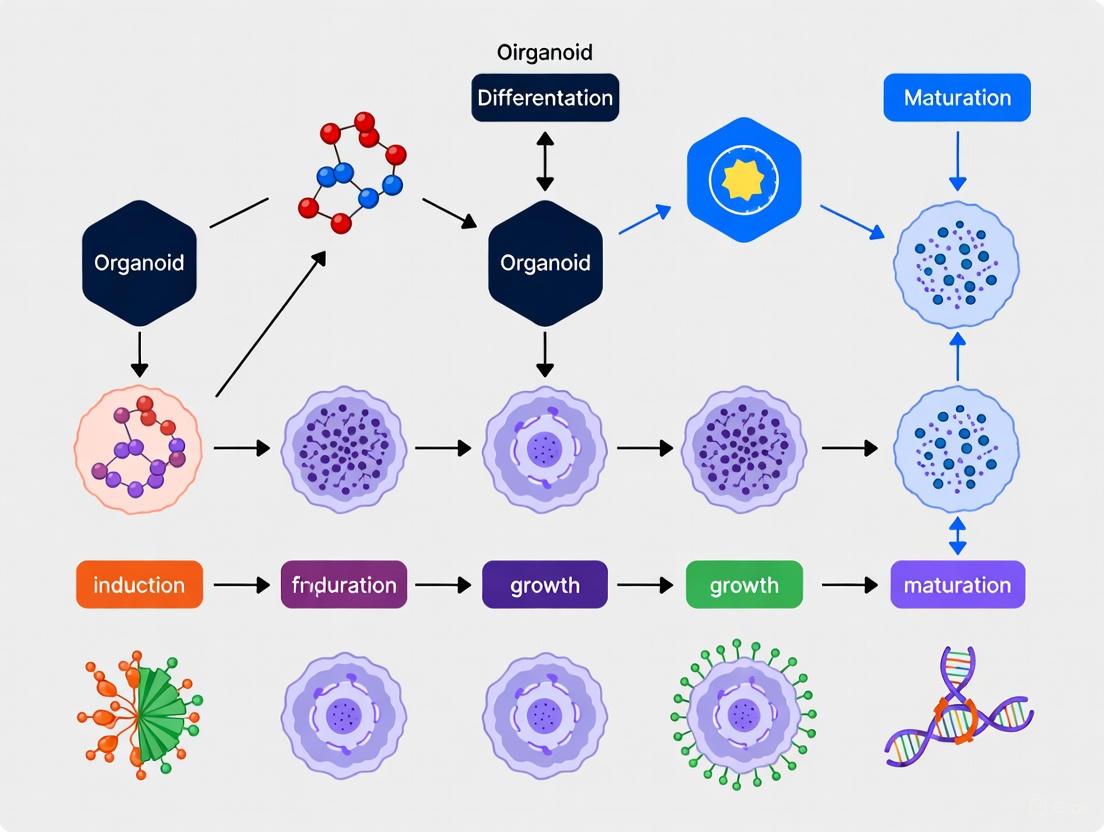
A standard protocol for generating brain region-specific organoids progresses through defined stages, from pluripotent cell aggregation to mature organoid culture. The workflow below outlines the key stages and their duration.
Figure 1. A generalized experimental workflow for generating brain region-specific organoids, highlighting key stages and typical timeframes from pluripotent stem cells to mature organoids [33] [32].
Stage 1: Embryoid Body (EB) Formation & Neural Induction (Days 1-5)
- Procedure: Dissociate human iPSCs or ESCs into single cells and aggregate them (approximately 500-10,000 cells) in ultra-low attachment (ULA) plates to form EBs in suspension. Culture in medium maintaining proliferation and multipotency until day 4-5 [33] [32].
- Key Reagents: Serum-free media, ROCK inhibitor to enhance cell survival after dissociation.
Stage 2: Regional Patterning (Days 5-7+)
- Procedure: Transition EBs into neural induction medium (NIM) supplemented with specific combinations of small molecule inhibitors and growth factors (see Table 2) to direct differentiation toward the desired brain region identity (e.g., forebrain, midbrain) [30] [32].
- Key Reagents: Dual-SMAD inhibitors for forebrain fate; SHH agonists and Wnt modulators for midbrain or hypothalamic fates.
Stage 3: ECM Embedding & 3D Culture (Day 7+)
- Procedure: Individual EBs are embedded in droplets of ECM, typically Matrigel, to provide a scaffold for 3D expansion. The embedded organoids are then transferred to spinning bioreactors for long-term culture [33] [32].
- Key Reagents: Matrigel or other defined ECMs; spinning bioreactors like the SpinΩ to enhance nutrient and oxygen perfusion [32].
Stage 4: Maturation & Differentiation (Days 10-15+)
- Procedure: After initial neuroepithelial structures form, organoids are cultured in differentiation media containing factors that support neuronal maturation, synaptogenesis, and glial differentiation. Media are refreshed regularly, and organoids can be maintained for months [33] [30].
- Key Reagents: Media supplements like B-27 with Vitamin A, BDNF, GDNF, and ascorbic acid [30].
Signaling Pathways Governing Differentiation
The sequential activation and inhibition of key developmental pathways are the cornerstone of directed organoid differentiation. The following diagram and breakdown illustrate the core signaling pathways manipulated in these protocols.
Figure 2. Core signaling pathways and their manipulation to direct regional fate from neuroectoderm. Soluble factors are used to activate or inhibit these pathways in a spatially and temporally controlled manner [31] [32].
- TGF-β/Activin/NODAL and BMP Inhibition: The initial step in neural induction is "Dual-SMAD inhibition," which blocks both TGF-β/Activin/NODAL (using SB431542) and BMP (using LDN193189) signaling pathways. This inhibition pushes pluripotent stem cells toward a default neuroectodermal fate [30] [32].
- WNT/β-catenin Signaling: The spatiotemporal manipulation of WNT signaling is crucial for anteroposterior patterning. Activation of WNT signaling (using CHIR99021) promotes caudal and midbrain fates. Furthermore, the ECM influences regionalization through the WNT ligand secretion mediator (WLS), linking mechanosensing to patterning [33] [31].
- Sonic Hedgehog (SHH) Signaling: The dorsoventral axis is patterned by SHH signaling. Low or absent SHH activity favors dorsal telencephalic fates (cerebral cortex), while progressively higher concentrations of SHH agonists (e.g., Purmorphamine) ventralize the tissue toward midbrain or hypothalamic identities [30] [32].
- Hippo Pathway (YAP/TAZ): This pathway is a key mediator of mechanotransduction. ECM-derived signals regulate YAP/TAZ activity, which in turn influences cell proliferation, lumen morphogenesis, and gene expression programs, including the upregulation of WLS [33].
The advent of three-dimensional (3D) organoid technology represents a paradigm shift in biomedical research, offering in vitro models that recapitulate the structural and functional complexity of human organs. Derived from pluripotent stem cells (PSCs), including embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), these self-organizing structures provide an unprecedented platform for studying human development, disease modeling, drug screening, and regenerative medicine [34] [20]. Unlike traditional two-dimensional (2D) cell cultures that lack tissue context and animal models that suffer from species-specific differences, organoids mimic the cellular heterogeneity, spatial organization, and cell-cell interactions of native tissues [34] [35]. This article provides a comprehensive overview of the protocols and applications for generating brain, liver, kidney, and pancreatic organoids, framed within the broader context of pluripotent stem cell research.
Core Principles of Organoid Generation
Organoid formation leverages the innate self-organization capacity of stem cells when provided with appropriate biochemical and biomechanical cues. The process typically involves directing PSCs through stages that mirror embryonic development via sequential activation and inhibition of key signaling pathways [36]. The initial aggregation of stem cells into 3D aggregates is often followed by fate specification using small molecules and growth factors, long-term maturation in 3D culture conditions, and finally, analysis or application [34] [36]. The success of this approach relies on optimizing the culture format, media composition, and extracellular matrix (ECM) support to ensure proper morphogenesis and functional maturation [37] [33].
Table 1: Key Signaling Pathways in Organoid Development
| Pathway | Role in Organogenesis | Common Modulators |
|---|---|---|
| WNT/β-catenin | Anterior-posterior patterning, nephron induction, pancreatic growth | CHIR99021 (activator), IWP2 (inhibitor) [38] [36] |
| FGF | Branching morphogenesis, mesoderm induction, progenitor maintenance | FGF2, FGF9, FGF10 [36] |
| BMP | Cell fate specification, dorsal-ventral patterning | BMP4, BMP7 [36] |
| RA (Retinoic Acid) | Anterior-posterior patterning, pancreatic differentiation | Retinoic acid [37] |
| Hippo/YAP | Mechanosensing, tissue growth, brain regionalization | YAP1 [33] |
| Notch | Nephron segmentation, cell fate decisions | DAPT (inhibitor) [36] |
Brain Organoids
Protocols and Regional Specification
Brain organoids model the complexity of the human brain, providing insights into neurodevelopment and disease. Two primary approaches exist: unguided (self-patterned) and guided (region-specific) protocols. The pioneering unguided protocol from the Knoblich/Lancaster lab involves embedding iPSC aggregates in Matrigel and using rotating bioreactors to generate whole-brain organoids containing multiple brain region identities [34]. In contrast, guided protocols from labs like Pasca's utilize small molecule morphogens (e.g., SMAD inhibitors, WNT agonists) to direct differentiation toward specific brain regions like the dorsal or ventral forebrain [34]. Recent advances include the "Hi-Q brain organoid" culture method that bypasses the traditional embryoid body stage, enabling generation of hundreds of high-quality organoids per batch with minimal cellular stress [34]. For studying circuit formation, assembloids can be created by fusing region-specific organoids (e.g., cortical-striatal) to model inter-regional connectivity [34].
Applications and Morphodynamic Insights
Brain organoids have been extensively applied to model neurodevelopmental disorders (e.g., autism, microcephaly), neurodegenerative diseases (e.g., Alzheimer's, Parkinson's), and psychiatric disorders [34]. They enable the study of human-specific aspects of brain development and function that cannot be adequately modeled in animals. Recent morphodynamic studies using long-term live light-sheet microscopy have revealed that extracellular matrix components enhance lumen expansion and influence regional patterning, particularly through WNT and Hippo (YAP1) signaling pathways [33]. These findings highlight the role of matrix-linked mechanosensing in brain regionalization.
Liver Organoids
Generation and Disease Modeling
Liver organoids can be generated from either pluripotent stem cells (PSCs) or adult stem cells (ASCs), each with distinct advantages. PSC-derived organoids offer the ability to model early developmental processes and can be created with less invasive methods, while ASC-derived organoids better maintain the characteristics of adult tissue and demonstrate greater genetic stability during long-term expansion [35]. A key advancement is the development of protocols that generate organoids with proper branched architecture and correct patterning of cell domains without relying on animal-derived matrices [37]. These organoids contain hepatocyte-like cells capable of synthesizing proteins, metabolizing drugs, and exhibiting polarized morphology with functional bile canaliculi [35].
Liver organoids have proven particularly valuable for modeling a wide spectrum of liver pathologies. For alcoholic liver disease (ALD), organoids replicate inflammatory responses and mitochondrial dysfunction in response to ethanol exposure [35]. For metabolic dysfunction-associated steatotic liver disease (MASLD) and its progressive form MASH, organoids accumulate lipids and exhibit inflammatory and fibrotic responses when treated with free fatty acids (FFA) [35]. Additionally, organoids support hepatitis B and E virus infection, enabling study of viral entry, replication, and host responses [35]. Patient-derived tumor organoids retain genomic features of original hepatocellular carcinoma, cholangiocarcinoma, and combined tumors, serving as personalized drug testing platforms [35].
Table 2: Liver Organoid Disease Modeling Applications
| Disease Model | Cellular Source | Induction Method | Key Features Recapitulated |
|---|---|---|---|
| Alcoholic Liver Disease (ALD) | Human ESC-derived liver organoid | 100 mM ethanol treatment for 7 days | Inflammatory response, mitochondrial depolarization [35] |
| MASLD | Human PSC-derived liver organoid | FFA treatment for 3-9 days | Lipid accumulation, inflammatory responses [35] |
| MASH | Human iPSC-derived liver organoid | FFA treatment; patient-derived organoids with steatosis-associated SNPs | Steatosis, inflammation, fibrosis [35] |
| Viral Hepatitis | Human ductal organoid | HBV or HEV infection | Viral entry, replication, host response [35] |
| Liver Cancer | Primary liver cancer-derived organoid | Patient-derived tumor cells | Genomic features, drug resistance [35] |
Future Directions in Liver Modeling
Current innovations focus on creating more physiologically relevant systems through multicellular assembloids, organoid-on-chip platforms with perfusable vasculature, and 3D bioprinting [35]. These advanced models better mimic the liver microenvironment and complex tissue interactions, enhancing their predictive value for drug screening and disease modeling. Integration with microfluidic systems allows for dynamic flow conditions that improve hepatocyte function and enable real-time monitoring of drug responses [35].
Kidney Organoids
Differentiation Protocols
Kidney organoid generation follows a stepwise protocol that recapitulates kidney development by directing PSCs through primitive streak, intermediate mesoderm, and metanephric mesenchyme stages [36]. The process begins with WNT activation using GSK3β inhibitors like CHIR99021 to induce posterior primitive streak, followed by treatment with FGF9 and sometimes BMP7 to pattern intermediate mesoderm [36]. Cells are then aggregated into 3D spheroids that self-organize into nephron-like structures containing glomerular, proximal tubule, loop of Henle, and distal tubule segments [36]. These structures express characteristic markers including NPHS1 (podocytes), LTL (proximal tubules), and ECAD (distal tubules) [36].
Applications and Limitations
Kidney organoids have been successfully applied to model genetic kidney diseases such as polycystic kidney disease (PKD) and congenital anomalies of the kidney and urinary tract (CAKUT) [36]. They also serve as predictive platforms for nephrotoxicity screening, as proximal tubule-like structures in organoids demonstrate functional uptake capabilities including albumin endocytosis [36]. However, current kidney organoid systems face limitations including incomplete maturation, lack of full nephron segmentation, limited vascularization, and variability between protocols [36]. To address these challenges, researchers are implementing dynamic culture systems like spinning bioreactors, organ-on-a-chip platforms, and co-culture with endothelial cells to improve functional maturation and vascular integration [36].
Pancreatic Organoids
Protocol Optimization
The generation of pancreatic organoids from human iPSCs has been challenging due to difficulties in achieving proper branched architecture with correct patterning of cell domains. Recent protocol optimizations have successfully promoted acinar cell differentiation and generated organoids with branches patterned into central trunk and peripheral tip domains without relying on animal-derived matrices [37]. This represents a significant advancement as it more accurately recapitulates the most important aspects of pancreatic tissue architecture, opening the door to high-throughput investigations of human pancreatic development and disease [37].
Disease Modeling and Future Directions
Pancreatic organoids provide a human-relevant system for studying pancreatic diseases including diabetes and pancreatic cancer [37]. These models are particularly valuable for investigating acinar-to-ductal metaplasia (ADM), a key step in the development of pancreatic intraepithelial neoplasia (PanIN) and pancreatic cancer [37]. The ability to generate patient-specific organoids from iPSCs enables personalized approaches to drug screening and disease modeling, potentially reducing reliance on animal models [37]. Future directions include enhancing functional maturation of endocrine components, particularly insulin-producing β-cells, and integrating pancreatic organoids with other cell types to model the complex interactions within the pancreatic microenvironment.
Table 3: Research Reagent Solutions for Organoid Generation
| Reagent/Category | Specific Examples | Function in Organoid Culture |
|---|---|---|
| Extracellular Matrices | Matrigel, Collagen I | Provide structural support and biochemical cues for 3D growth and polarization [34] [33] |
| WNT Pathway Modulators | CHIR99021 (activator), IWP2 (inhibitor) | Direct lineage specification and progenitor maintenance [38] [36] |
| Growth Factors | FGF2, FGF9, FGF10, BMP4, BMP7, VEGF, HGF, EGF | Pattern specific tissue identities and support proliferation [36] |
| Neural Induction Media | NIM (Neural Induction Medium) | Promote neuroectodermal commitment [34] [33] |
| Metabolic Inducers | Free Fatty Acids (FFA), Ethanol | Induce disease phenotypes like steatosis or alcoholic injury [35] |
| Cell Culture Platforms | Low-adhesion plates, Spinning bioreactors, Microfluidic chips | Support 3D aggregation and improve nutrient/waste exchange [34] [36] |
Tissue-specific organoids derived from pluripotent stem cells have revolutionized our approach to studying human development, disease mechanisms, and drug responses. While significant progress has been made in generating brain, liver, kidney, and pancreatic organoids with remarkable architectural and functional complexity, challenges remain in achieving full maturation, reducing variability, and incorporating missing components such as vasculature and immune cells. Future advances will likely come from interdisciplinary approaches combining bioengineering, microfluidics, and single-cell technologies to enhance the physiological relevance and translational potential of these models. As the field continues to evolve, organoid technology is poised to become an indispensable tool for personalized medicine, drug discovery, and ultimately, regenerative therapies.
Organoids generated from human pluripotent stem cells (hPSCs), including both embryonic and induced pluripotent stem cells, have emerged as a transformative technology in biomedical research, offering unprecedented opportunities for studying human development, disease modeling, and drug development [20]. These three-dimensional, self-organizing structures mimic the cytoarchitecture and functional characteristics of native human organs, providing a more physiologically relevant platform compared to traditional two-dimensional cultures [20] [39]. However, conventional organoid systems face significant limitations in replicating the complexity of in vivo microenvironments, particularly regarding two crucial components: the immune system and vascular networks.
The absence of immune cells in standard organoid cultures limits their utility for studying immunobiology, tumor-immune interactions, and immunotherapeutic interventions [40] [41]. Similarly, the lack of functional vasculature restricts organoid size, maturity, and physiological relevance due to inadequate oxygen and nutrient delivery [42] [43]. This application note addresses these limitations by providing detailed protocols and strategies for integrating immune co-cultures and vascularization systems into hPSC-derived organoid research, enabling the generation of more complete and physiologically accurate models for basic research and drug development applications.
Immune Co-Culture Strategies for hPSC-Derived Organoids
Fundamentals of Organoid-Immune Interactions
The tumor immune microenvironment plays a critical role in cancer progression and therapy response, comprising diverse immune cell populations including T cells, B cells, natural killer (NK) cells, macrophages, dendritic cells, and myeloid-derived suppressor cells [40] [41]. These cells engage in complex interactions with tumor cells through direct contact and cytokine signaling, influencing immune surveillance, evasion, and therapeutic outcomes. Recapitulating these interactions in hPSC-derived organoids requires sophisticated co-culture systems that maintain functional immune populations alongside organoid structures.
Immune organoids are three-dimensional culture systems derived from immune cells or containing immune cell components designed to mimic the structure and function of the immune system or its specific parts [41]. In cancer research, these systems enable the investigation of how immune cells interact with cancer cells within the tumor microenvironment, providing insights into mechanisms of immune evasion and potential therapeutic targets. For hPSC-derived organoids, integrating immune components typically involves co-culturing with autologous or allogeneic immune cells, including peripheral blood lymphocytes, tumor-infiltrating lymphocytes, or macrophages [40] [44].
Established Immune Co-Culture Methodologies
Three primary methods have been established for co-culturing immune cells with organoids, each offering distinct advantages and limitations for specific research applications:
Method 1: Indirect Co-culture System This approach involves mixing organoids with extracellular matrix (typically Matrigel or BME-2) and adding exogenous immune cells to the culture medium, allowing interaction through soluble factors without direct cell contact [41]. The system facilitates immune cell infiltration and paracrine signaling while maintaining organoid integrity. This method is particularly suitable for studying cytokine-mediated responses and immune cell migration.
Method 2: Direct Co-culture with Dissociated Organoids This protocol begins with established organoids that are dissociated into single cells or small clusters, then mixed with immune cells (typically lymphocytes from peripheral blood) and re-embedded in matrix for continued culture [41]. This approach maximizes direct cell-cell contacts and is ideal for studying immune synapse formation and direct cytotoxicity assays.
Method 3: Combined Matrix Embedding This technique involves directly mixing intact organoids and immune cells within the extracellular matrix simultaneously at the initiation of culture [41]. This method preserves organoid structure while enabling both direct and indirect interactions, serving as a balanced approach for general tumor-immune interaction studies.
Table 1: Comparison of Immune Co-culture Methods for hPSC-Derived Organoids
| Method | Key Features | Optimal Applications | Limitations |
|---|---|---|---|
| Indirect Co-culture | Immune cells in medium; indirect interactions via soluble factors | Study of cytokine signaling, immune cell migration, chemotaxis assays | Limited direct cell contact, potential loss of spatial organization |
| Direct Co-culture with Dissociated Organoids | Organoids dissociated and mixed with immune cells before embedding | T cell cytotoxicity assays, immune synapse studies, high-throughput screening | Disruption of native organoid architecture, potential loss of heterogeneity |
| Combined Matrix Embedding | Intact organoids and immune cells mixed together in matrix | General tumor-immune interactions, preserving organoid structure | Variable immune cell distribution, potential for inadequate infiltration |
Detailed Protocol: Establishing Immune Co-cultures with hPSC-Derived Organoids
Materials Required:
- hPSC-derived organoids (20-30 organoids per condition)
- Immune cell source (autologous peripheral blood mononuclear cells, tumor-infiltrating lymphocytes, or specific immune cell subsets)
- Advanced DMEM/F12 culture medium
- Growth factor-reduced Matrigel or similar extracellular matrix
- Organoid culture medium (specific to organoid type)
- Immune cell culture supplements (IL-2, IL-15, etc., as required)
- 24-well or 96-well culture plates
- Centrifuge tubes and pipettes
Procedure for Combined Matrix Embedding (Method 3):
Preparation of Organoids:
- Harvest hPSC-derived organoids from maintenance culture using appropriate dissociation methods.
- Wash organoids twice with advanced DMEM/F12 medium to remove residual matrix.
- Count organoids and adjust concentration to 50-100 organoids per mL in cold medium.
Preparation of Immune Cells:
- Isolate immune cells from desired source (peripheral blood, tumor tissue, or in vitro differentiation).
- For T cells, activate with anti-CD3/CD28 beads or cytokines as required for experimental goals.
- Count cells and adjust concentration to 1-5×10^6 cells/mL in appropriate immune cell medium.
Co-culture Establishment:
- Combine organoids and immune cells in a 1:1 ratio in a cold tube.
- Add cold Matrigel to achieve final concentration of 50-70% matrix.
- Plate 30-50 μL drops in pre-warmed culture plates.
- Polymerize matrix for 20-30 minutes at 37°C.
- Carefully add pre-warmed organoid-immune co-culture medium.
Culture Maintenance:
- Refresh medium every 2-3 days, maintaining appropriate cytokine supplements.
- Monitor organoid-immune interactions daily using microscopy.
- Harvest for analysis at predetermined timepoints (typically 3-14 days).
Critical Considerations:
- Maintain appropriate immune cell to organoid ratios (typically 10:1 to 100:1 depending on cell types)
- Include controls (organoids alone, immune cells alone) for proper experimental interpretation
- Optimize cytokine combinations for specific immune cell survival and function
- Monitor for overgrowth of either component that might imbalance the system
Vascularization Strategies for hPSC-Derived Organoids
The Importance of Vascular Networks in Organoid Maturation
Vascularization represents a critical advancement in organoid technology, enabling enhanced oxygen and nutrient delivery, metabolic waste removal, and improved organoid growth and maturation [42] [43]. The development of vascular networks within organoids not only supports larger and more complex structures but also better recapitulates native tissue interactions and promotes tissue regeneration potential [42]. For hPSC-derived organoids, vascularization strategies can be broadly categorized into in vivo and in vitro approaches, each with distinct mechanisms and applications.
Recent studies demonstrate that vascularized organoid models provide valuable insights into tissue interactions and promote tissue regeneration [42]. The creation of vascular networks within heart and liver organoids using hPSCs has been achieved through novel combinations of growth factors and differentiation protocols, resulting in organoids that closely model human organ development [43]. These advances highlight the potential of vascularized organoids to bridge the gap between traditional in vitro models and in vivo physiology.
Vascularization Techniques for hPSC-Derived Organoids
In Vivo Vascularization Strategies: In vivo approaches involve transplanting organoids into host organisms (typically immunocompromised mice) where they become vascularized by the host circulatory system [42]. This method leverages the host's natural angiogenic capabilities but limits experimental accessibility and introduces host variables.
In Vitro Self-Assembly Vascularization: This technique utilizes the innate capacity of hPSCs to differentiate into endothelial and perivascular cells when exposed to specific cytokine combinations [43]. Protocols typically involve sequential differentiation with VEGF, FGF, and BMP signaling pathway modulators to promote vasculogenesis.
Bioengineering Approaches: Advanced bioengineering techniques including 3D bioprinting, microfluidic organ-on-chip systems, and scaffold-based strategies provide precise control over vascular architecture [42] [45]. These methods enable the creation of perfusable networks but require specialized equipment and expertise.
Table 2: Vascularization Strategies for hPSC-Derived Organoids
| Strategy | Mechanism | Key Advantages | Limitations |
|---|---|---|---|
| In Vivo Transplantation | Host-mediated angiogenesis after implantation | Robust, functional anastomosis with host circulation, high survival rates | Limited experimental access, host variability, immunocompatibility issues |
| In Vitro Self-Assembly | Directed differentiation of hPSCs to vascular lineages | Preserves entirely human system, enables study of human-specific vasculogenesis | Limited structural organization, immature vessel characteristics, variability |
| Bioengineering Approaches | 3D bioprinting, microfluidics, scaffold design | Precise control over architecture, potential for perfusion, high reproducibility | Specialized equipment required, complex protocols, potential biocompatibility issues |
Detailed Protocol: Generating Vascularized Heart Organoids from hPSCs
Materials Required:
- hPSCs (maintained in feeder-free conditions)
- Essential 8 or mTeSR medium for hPSC maintenance
- RPMI 1640 medium
- B27 supplements (with and without insulin)
- CHIR99021 (GSK3 inhibitor)
- IWP2 or IWP4 (Wnt inhibitor)
- VEGF165
- FGF2
- BMP4
- Y-27632 (ROCK inhibitor)
- Low-growth factor Matrigel
- Ultra-low attachment plates
- Differentiation basal medium
Procedure for Vascularized Cardiac Organoid Generation:
hPSC Maintenance and Quality Control:
- Culture hPSCs in Essential 8 medium on recombinant vitronectin-coated plates.
- Passage cells at 70-80% confluence using EDTA dissociation.
- Ensure >90% viability and pluripotency marker expression before differentiation.
Mesoderm Induction and Cardiac Progenitor Specification:
- Dissociate hPSCs to single cells using Accutase.
- Seed 2,000-4,000 cells per well in 96-well ultra-low attachment plates in medium containing Y-27632.
- Day 0-1: Activate Wnt signaling with 6-12 μM CHIR99021 in RPMI/B27 minus insulin.
- Day 2-4: Inhibit Wnt signaling with 2-5 μM IWP2 in RPMI/B27 minus insulin.
Vascular Co-differentiation:
- Day 5-7: Add VEGF165 (50 ng/mL) and FGF2 (20 ng/mL) to promote endothelial differentiation.
- Day 8-14: Continue culture with VEGF165 (25 ng/mL) in RPMI/B27 complete medium.
- Refresh medium every 2-3 days, monitoring organoid formation.
Cardiac and Vascular Maturation:
- Day 15-30: Transfer organoids to orbital shaking platform (60-80 rpm) to enhance nutrient exchange.
- Maintain in RPMI/B27 complete medium with reduced VEGF (10 ng/mL).
- Spontaneous contraction typically appears between days 10-15, with vascular network formation evident by day 20.
Characterization and Quality Assessment:
- Monitor beating activity and rate as functional cardiac marker
- Immunostaining for cardiac troponin T (cardiomyocytes), CD31 (endothelial cells), and α-smooth muscle actin (perivascular cells)
- Assess vascular network formation using lectin perfusion or dextran uptake assays
- Evaluate structural organization using confocal microscopy and 3D reconstruction
Integrated Workflow and Signaling Pathways
The successful integration of immune components and vascular networks into hPSC-derived organoids requires careful coordination of multiple signaling pathways and culture parameters. The following diagrams illustrate key workflows and molecular pathways involved in these advanced organoid systems.
Integrated Workflow for Advanced Organoid Generation
Diagram 1: Integrated Organoid Generation Workflow. This diagram illustrates the sequential process for generating advanced organoids with immune and vascular components from hPSCs.
Key Signaling Pathways in Organoid Vascularization and Immune Interaction
Diagram 2: Key Signaling Pathways. This diagram shows the principal molecular pathways coordinating vascular development and immune interactions in advanced organoid systems.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of immune co-cultures and vascularization strategies requires careful selection of research reagents and materials. The following table details essential components for establishing these advanced organoid systems.
Table 3: Essential Research Reagents for Advanced Organoid Models
| Reagent Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| Extracellular Matrices | Growth factor-reduced Matrigel, Collagen I, Fibrin | Structural support, biomechanical cues, reservoir for signaling factors | Matrigel concentration typically 50-70%; collagen concentration 2-4 mg/mL; fibrin 5-10 mg/mL |
| Cytokines & Growth Factors | VEGF165, FGF2, BMP4, SCF, IL-2, IL-15, IFN-γ | Direct differentiation, support cell survival and function | VEGF: 10-50 ng/mL; FGF2: 10-25 ng/mL; IL-2: 10-100 IU/mL depending on application |
| Small Molecule Inhibitors/Activators | CHIR99021, IWP2, IWP4, SB431542, Y-27632 | Modulate signaling pathways, enhance survival | CHIR99021: 3-12 μM; IWP2: 2-5 μM; Y-27632: 10 μM (first 24-48h after passaging) |
| Cell Surface Markers for Characterization | Anti-CD31, Anti-CD34, Anti-CD45, Anti-CD3, Anti-CD56 | Identify and isolate specific cell populations | Use validated antibodies for flow cytometry (1:100-1:500 dilution) and immunofluorescence (1:50-1:200) |
| Culture Media & Supplements | Advanced DMEM/F12, RPMI 1640, B27, N2, GlutaMAX | Nutritional support, defined culture conditions | B27 typically used at 1X or 2X concentration; GlutaMAX at 1-2 mM |
Applications and Future Directions
The integration of immune components and vascular networks into hPSC-derived organoids significantly expands their applications in basic research and drug development. These advanced models enable more physiologically relevant studies of human development, disease mechanisms, and therapeutic interventions.
In cancer research, patient-derived tumor organoids with autologous immune cells have demonstrated remarkable clinical correlation for predicting immunotherapy responses [40] [41]. These systems allow for personalized therapeutic testing and biomarker discovery. Similarly, vascularized organoids provide enhanced models for studying neurovascular interactions, liver metabolism, and cardiac function under conditions that better mimic in vivo physiology [42] [43] [45].
Future developments in this field will likely focus on further enhancing organoid complexity through integration of additional tissue components, improving maturation states, and developing standardized protocols for reproducibility and scalability. The convergence of organoid technology with advanced bioengineering, microfluidics, and high-content screening platforms will continue to bridge the gap between traditional in vitro models and in vivo physiology, accelerating drug discovery and development pipelines.
Organoids generated from human pluripotent stem cells (hPSCs), including embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), represent a paradigm shift in preclinical research. These self-organizing three-dimensional (3D) structures mimic the architectural and functional complexity of native human organs more faithfully than traditional two-dimensional (2D) cultures or animal models [20] [46]. Their ability to recapitulate human physiology, genetic variability, and disease mechanisms positions them as powerful platforms for translational applications. This document details specific application notes and experimental protocols for using hPSC-derived organoids in disease modeling, personalized therapy screening, and toxicology, providing a practical resource for researchers and drug development professionals.
Disease Modeling with Patient-Derived Organoids
Application Note: Patient-derived cancer organoids (PDCOs) have revolutionized the modeling of human cancers, particularly those of the digestive system (e.g., gastric, liver, colorectal, pancreatic). These models faithfully preserve the histological features, genetic landscapes, and intratumoral heterogeneity of the original patient tumors, enabling the study of disease mechanisms and therapy resistance [47]. A key breakthrough was the establishment of long-term culture systems for intestinal organoids, which was later extended to generate tumor organoids from colorectal adenomas and adenocarcinomas [47]. Similarly, brain organoids derived from hiPSCs provide a unique model for studying neurodegenerative diseases like Alzheimer's and Parkinson's, offering insights into early cellular and molecular events that are difficult to capture in post-mortem tissue or animal models [46].
- Protocol 2.1: Establishing Patient-Derived Cancer Organoids (PDCOs)
- Objective: To generate and culture 3D patient-derived cancer organoids from a tumor biopsy for disease modeling and drug testing.
- Materials:
- Tumor tissue sample (from surgical resection or biopsy)
- Gentle Cell Dissociation Reagent (e.g., from STEMCELL Technologies)
- Phosphate-buffered saline (PBS)
- Growth factor-reduced Matrigel
- Organoid culture medium (Advanced DMEM/F12), supplemented with specific growth factors tailored to the cancer type (e.g., EGF, Noggin, R-spondin) [47]
- 70 μm cell strainer
- Centrifuge
- Method:
- Tissue Processing: Mechanically mince the tumor tissue into small fragments (2-4 mm) and wash extensively with cold PBS.
- Digestion: Incubate tissue fragments with Gentle Cell Dissociation Reagent for 15-30 minutes at room temperature to dissociate the tissue into single cells and small clusters.
- Crypt/Organoid Isolation: Remove the dissociation reagent and wash the fragments with PBS by pipetting to release crypts or tumor cell clusters. Collect the supernatant containing the isolated structures.
- Filtration and Centrifugation: Pass the supernatant through a 70 μm cell strainer to remove debris. Centrifuge the filtrate at 300 g for 5 minutes to pellet the cells/organoids.
- Embedding in Matrix: Resuspend the pellet in Matrigel and plate as droplets in a pre-warmed culture dish. Allow the Matrigel to polymerize at 37°C for 20-30 minutes.
- Culture: Overlay the polymerized Matrigel droplets with organoid culture medium. Refresh the medium every 2-3 days.
- Passaging: Once organoids reach a sufficient size (typically after 7-14 days), dissociate them mechanically or enzymatically and re-embed in fresh Matrigel for expansion [47] [48].
The following workflow outlines the key steps for establishing and utilizing patient-derived organoids for translational research.
Personalized Therapy Screening
Application Note: PDCOs and hiPSC-derived disease models are transforming precision oncology and personalized medicine. These models serve as "avatars" of a patient's tumor, allowing for ex vivo testing of chemotherapies, targeted therapies, and immunotherapies to predict clinical response [20] [47]. Studies have demonstrated that the genetic alterations in organoids can predict their dependencies and drug sensitivities; for instance, gastric cancer organoids with RNF43 mutations are sensitive to Wnt inhibitors, while those with ERBB amplifications respond to HER2-targeted therapies [47]. The integration of organoids with microfluidic systems and AI-driven image analysis (e.g., OrBITS platform) further enhances the throughput and predictive power of these drug screens [47].
- Protocol 3.1: High-Throughput Drug Screening on Cancer Organoids
- Objective: To test the efficacy and IC₅₀ of multiple therapeutic compounds on patient-derived cancer organoids in a medium-throughput format.
- Materials:
- Expanded PDCOs
- 96- or 384-well cell culture plates
- Dilution series of therapeutic compounds (e.g., chemotherapy, targeted therapy)
- Cell viability assay kit (e.g., ATP-based luminescence)
- Automated liquid handler (optional)
- Microplate reader
- AI-powered image analysis software (e.g., for organoid morphology) [47]
- Method:
- Organoid Preparation: Harvest and dissociate expanded PDCOs into single cells or small fragments. Seed them uniformly into 96- or 384-well plates pre-coated with Matrigel.
- Drug Addition: After 24-48 hours, add a dilution series of each drug candidate to the wells. Include negative (DMSO) and positive (maximum cell death) controls.
- Incubation: Incubate the plates for 3-7 days, depending on the organoid growth rate and drug mechanism.
- Viability Assessment: Measure cell viability using an ATP-based luminescence assay according to the manufacturer's instructions. Alternatively, use high-content imaging to quantify organoid size, morphology, and viability in real-time.
- Data Analysis: Calculate the percentage of viability relative to controls and determine the half-maximal inhibitory concentration (IC₅₀) for each drug. AI platforms can automate the analysis of imaging data to classify responders and non-responders [47].
Table 1: Quantitative Drug Screening Data from Digestive System Cancer Organoids
| Cancer Type | Drug Tested | Organoid Response Correlation with Patient | Key Genetic Biomarker Linked to Response | Reference |
|---|---|---|---|---|
| Colorectal Cancer | Anti-EGFR | ~90% Predictive Accuracy | KRAS mutations confer resistance | [47] |
| Gastric Cancer | Trastuzumab | High correlation | HER2 amplification predicts response | [47] |
| Pancreatic Cancer | Chemotherapy (e.g., Gemcitabine) | Significant heterogeneity observed | Subpopulations with inherent resistance | [47] |
| Liver Cancer | Multi-kinase Inhibitors | Recapitulated clinical response spectra | CTNNB1 mutations influence sensitivity | [47] |
Toxicology and Safety Assessment
Application Note: Organoids offer a physiologically relevant human-based platform for toxicology, overcoming the species-specific limitations of animal models [20] [49]. Liver organoids, for example, can be used to assess drug-induced hepatotoxicity, a major cause of drug attrition [20]. Similarly, brain organoids provide a model for neurotoxicity screening [46]. The 3D architecture and cellular heterogeneity of organoids allow for the evaluation of complex toxicological endpoints, including barrier function in gut organoids and metabolic function in liver organoids. The recent FDA Modernization Act 2.0, which no longer mandates animal testing for drug approval, has further accelerated the adoption of these human-relevant models [50].
- Protocol 4.1: Hepatotoxicity Assessment using Liver Organoids
- Objective: To evaluate the toxic effects of a drug candidate on hPSC-derived liver organoids by measuring functional and viability endpoints.
- Materials:
- hPSC-derived liver organoids
- Test compound
- Albumin ELISA kit
- Urea colorimetric assay kit
- CYP450 activity assay (e.g., P450-Glo Assay)
- Lactate Dehydrogenase (LDH) release cytotoxicity assay kit
- Method:
- Organoid Treatment: Culture liver organoids in 96-well plates and expose them to a range of concentrations of the test compound for 24-72 hours.
- Functional Assessment:
- Albumin Secretion: Collect culture supernatant and quantify albumin secretion using an ELISA kit. A decrease indicates impaired hepatocyte function.
- Urea Production: Measure urea concentration in the supernatant using a colorimetric assay. Reduced production suggests metabolic dysfunction.
- CYP450 Activity: Assess the activity of key cytochrome P450 enzymes using a luminogenic substrate assay to evaluate potential for drug-drug interactions.
- Cytotoxicity Assessment: Measure the release of LDH into the culture medium using a colorimetric or fluorometric assay, which indicates cell membrane damage and cell death.
- Data Integration: Combine data from functional and cytotoxicity assays to determine a compound's therapeutic index and predict its potential human hepatotoxicity [49].
Table 2: Functional Toxicity Endpoints in Organoid Models
| Organoid Type | Toxicity Endpoint | Assay Method | Physiological Relevance |
|---|---|---|---|
| Liver Organoids | Hepatocyte Function | Albumin/Urea Production | Measures synthetic and metabolic capacity of hepatocytes [49] |
| Metabolic Competence | CYP450 Activity | Indicates potential for drug metabolism and interaction [49] | |
| Cytotoxicity | LDH Release | Quantifies compound-induced cell death [49] | |
| Gut Organoids | Barrier Integrity | Transepithelial Electrical Resistance (TEER) | Models damage to the gut lining and increased permeability [49] |
| Absorptive Function | Nutrient Uptake Assays | Assesses impact of toxins on digestive function [49] | |
| Brain Organoids | Neurotoxicity | Neuronal Viability/Morphology Staining | Identifies damage to neurons and glial cells [46] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents for hPSC-Derived Organoid Culture and Assays
| Item | Function | Example/Note |
|---|---|---|
| Matrigel | Extracellular matrix (ECM) substitute providing structural support and biochemical cues for 3D growth. | Animal-derived, batch variability is a challenge [47]. |
| Defined Media Kits | Culture medium optimized for specific organoid types (e.g., intestinal, hepatic, cerebral). | Contains essential growth factors like EGF, Noggin, R-spondin [38]. |
| Growth Factors (Noggin, R-spondin) | Key signaling molecules that inhibit differentiation and promote stemness in gut and other organoids. | Often added as recombinant proteins or produced by feeder cells [47]. |
| Gentle Cell Dissociation Reagent | Enzymatic solution for dissociating organoids into single cells for passaging or analysis. | Prevents excessive damage to cell surface proteins [48]. |
| Y-27632 (ROCK inhibitor) | Small molecule that inhibits apoptosis, increasing cell survival after dissociation. | Crucial for improving plating efficiency after passaging. |
| CRISPR/Cas9 Systems | Genome-editing tool for introducing disease-specific mutations or correcting genetic defects in hiPSCs. | Enables precise disease modeling [47]. |
| ATP-based Viability Assays | Sensitive luminescent method to quantify cell viability and proliferation in high-throughput screens. | Standard for drug screening applications [47]. |
| Microfluidic "Organ-on-Chip" Devices | Platforms for perfusing organoids, enabling vascularization and more complex tissue modeling. | Improves nutrient delivery and maturity [20] [51]. |
The translational applications of hPSC-derived organoids in disease modeling, personalized therapy screening, and toxicology are rapidly advancing. By providing human-relevant, physiologically complex models, organoids significantly enhance the predictive power of preclinical research, align with the 3Rs principles (replacement, reduction, and refinement of animal testing), and accelerate the development of safer, more effective personalized therapies. While challenges in standardization, vascularization, and scalability remain, ongoing interdisciplinary innovations in bioengineering, AI, and protocol harmonization are poised to fully integrate organoid technology into the mainstream drug development pipeline and clinical decision-making.
Navigating Technical Hurdles: Solutions for Standardization, Scalability, and Maturation
Addressing Batch-to-Batch Variability and Reproducibility Issues
The field of pluripotent stem cell-derived organoid research represents a paradigm shift in biomedical science, offering unprecedented opportunities for modeling human development and disease. However, the transformative potential of this technology is constrained by significant challenges in batch-to-batch variability and experimental reproducibility. These issues span biological, technical, and analytical domains, creating barriers to translational application and reliable data interpretation [52] [53]. The inherent complexity of three-dimensional (3D) culture systems, combined with the dynamic nature of stem cell differentiation, introduces multiple sources of variation that must be systematically addressed to advance the field.
Recent comprehensive analyses reveal that reproducibility concerns are particularly pronounced in neural differentiation models. A review of 58 research articles utilizing iPSC-derived neural cells to investigate intellectual and developmental disabilities (IDDs) demonstrated a troubling decline in reported quality control measures over time, despite increased use of patient-specific lines [53]. This trend highlights the critical need for standardized reporting practices and rigorous quality assessment protocols throughout the organoid generation workflow. Furthermore, systematic profiling of brain organoids across multiple cell lines and protocols indicates that both protocol choices and pluripotent cell line characteristics significantly influence organoid variability and cell-type representation [23]. This application note provides a comprehensive framework for identifying, quantifying, and mitigating sources of variability to enhance reproducibility in organoid research.
Stratified Variability Metrics in Organoid Systems
Table 1: Classification and impact of major variability sources in organoid generation
| Variability Category | Specific Sources | Impact Level | Detection Methods |
|---|---|---|---|
| Biological Variability | Genetic background diversity [53] | High | SNP microarray, RNA sequencing [53] |
| Donor-specific effects [53] | Medium-High | Multi-donor line comparison | |
| Cell line differentiation propensity [23] | High | NEST-Score, transcriptional profiling [23] | |
| Technical Variability | Extracellular matrix batch effects [52] | High | Proteomic analysis, rheological testing |
| Reprogramming method differences [53] | Medium | STR profiling, pluripotency verification [53] | |
| Passaging method inconsistencies [53] | Medium | Morphological analysis, growth rate tracking | |
| Protocol Variability | Neural induction timing [52] | High | Single-cell RNA sequencing [23] |
| Morphogen concentration gradients [52] | High | Immunostaining, spatial transcriptomics | |
| Embedding methodology [52] [33] | Medium-High | Light-sheet microscopy, morphometrics [33] | |
| Environmental Variability | Media composition fluctuations [53] | Medium | Metabolomic profiling, pH monitoring |
| Seeding density variations [53] | Medium | Automated cell counting, size distribution analysis |
The quantitative impact of these variability sources has been systematically investigated through multi-protocol comparisons. Research demonstrates that protocol and pluripotent cell line choices significantly influence organoid variability and cell-type representation, necessitating careful validation of these parameters for each experimental application [23]. The introduction of quantitative assessment tools like the NEST-Score provides a standardized metric for evaluating cell-line- and protocol-driven differentiation propensities through comparison to in vivo references [23]. Furthermore, longitudinal tracking of organoid morphodynamics has revealed that subtle differences in initial conditions can profoundly influence subsequent patterning and regionalization, highlighting the need for precise control over early developmental stages [33].
Standardized Protocols for Enhanced Reproducibility
Quality Control Framework for Pluripotent Stem Cells
Rigorous quality control of starting materials is fundamental to reducing batch-to-batch variability. The following protocol outlines essential characterization steps for pluripotent stem cells prior to organoid differentiation:
Molecular Characterization: Perform G-banded karyotyping or KaryoStat assays to identify chromosomal abnormalities. Conduct STR profiling to verify cell line identity and hPSC genetic analysis to assess pluripotency status [53].
Functional Assessment: Validate pluripotency through embryoid body formation followed by trilineage differentiation analysis via qPCR. Optional teratoma formation assays can provide additional confirmation of differentiation potential [53].
Culture Monitoring: Document passage number, morphology assessment (including presence of spontaneously differentiating cells), and mycoplasma/sterility testing. Record media composition and extracellular matrix details for each culture condition [53].
Implementation of this comprehensive QC framework significantly enhances cross-study reproducibility and enables meaningful comparison of results across different laboratories and experimental platforms.
Standardized Cerebral Organoid Generation with Reduced Variability
Table 2: Key reagents and quality checkpoints in cerebral organoid generation
| Stage | Critical Reagents | Quality Checkpoints | Acceptance Criteria |
|---|---|---|---|
| EB Formation | Stem cell lines (characterized) [53] | Embryoid body size uniformity | Diameter: 450-550 µm [33] |
| Neural Induction | SMAD inhibitors (e.g., LDN, SB) [52] | Neuroepithelium formation | Rosette structures by day 7-10 [33] |
| Patterning | Morphogens (e.g., SHH, FGF, RA) [52] | Regional identity verification | Immunostaining for region-specific markers |
| Maturation | BDNF, GDNF, vitamin A [33] | Neuronal functionality | Multi-electrode array, patch clamp [53] |
This protocol leverages both guided and unguided approaches, with specific modifications to enhance reproducibility:
3D Embryoid Body Formation: Aggregate 450-550 pluripotent stem cells per EB using forced-aggregation plates to ensure uniform size distribution. Culture in EB medium for 4-5 days with daily medium changes [33].
Neural Induction Phase: Transfer EBs to neural induction medium containing dual SMAD inhibitors (LDN-193189 100 nM, SB-431542 1-10 µM) for 10-12 days. For unguided protocols, omit additional patterning factors [52].
ECM Embedding Considerations: At day 4-6, carefully embed organoids in ECM. Acknowledge that Matrigel, while common, introduces batch variability due to its complex, undefined composition [52]. Document batch numbers and consider defined synthetic hydrogels as alternatives for critical applications [52].
Regional Patterning (Guided Protocols): For dorsal forebrain specification, maintain SMAD inhibition and add WNT inhibitors (IWR-1 3 µM) from days 10-25. For ventral identities, utilize SHH pathway agonists (SAG 100-500 nM) during this critical window [52].
Long-term Maturation: After 30 days, transfer organoids to spinning bioreactors or orbital shakers to enhance nutrient delivery. Culture for up to 120 days with bi-weekly medium changes, monitoring for excessive necrotic core formation [54].
Experimental Design and Analytical Frameworks
Signaling Pathways in Regional Patterning
The following diagram illustrates key signaling pathways manipulated during regional patterning of brain organoids and their cross-inhibitory relationships:
Signaling Pathways in Brain Organoid Patterning
This signaling network illustrates how coordinated manipulation of these pathways enables generation of region-specific organoids, with dorsal identities promoted by WNT inhibition and ventral fates induced through SHH activation [52]. The precise timing, concentration, and duration of morphogen exposure critically influence patterning outcomes and represent a significant source of protocol variability between laboratories.
Experimental Workflow for Reproducibility Assessment
The integrated workflow for systematic variability assessment throughout the organoid generation process includes both quality control checkpoints and functional validation:
Organoid Generation and Quality Control Workflow
This workflow emphasizes critical checkpoints for minimizing technical variability, including comprehensive stem cell characterization before differentiation initiation, morphological assessment during neuroepithelium formation, and functional validation of mature neuronal populations [53] [33]. Integration of multi-omic profiling at terminal timepoints provides definitive assessment of cell-type composition and transcriptional states relative to in vivo benchmarks [23].
Research Reagent Solutions for Variability Reduction
Table 3: Essential research reagents and their applications in reproducibility enhancement
| Reagent Category | Specific Examples | Function | Variability Considerations |
|---|---|---|---|
| Extracellular Matrices | Matrigel [52] | Support 3D architecture | High batch variability; document lot numbers |
| Synthetic hydrogels [52] | Defined alternative | Increased reproducibility, reduced complexity | |
| Morphogenetic Factors | SMAD inhibitors (LDN) [52] | Neural induction | Concentration-critical; prepare aliquots |
| SHH pathway agonists (SAG) [52] | Ventral patterning | Titrate for optimal response | |
| WNT inhibitors (IWR) [52] | Dorsal patterning | Stability concerns; fresh preparation | |
| Cell Lines | Commercially available iPSCs [23] | Protocol standardization | Characterize differentiation propensity |
| Isogenic controls [53] | Genetic background control | CRISPR-edited matched lines | |
| Characterization Tools | NEST-Score [23] | Protocol performance | Benchmark against in vivo references |
| Mass cytometry panels [55] | Single-cell signaling | 40+ parameters for deep phenotyping |
Strategic selection and quality control of critical reagents significantly enhances experimental reproducibility. Particularly for extracellular matrices, which exhibit substantial batch-to-batch variability, meticulous documentation of lot numbers and performance validation is essential [52]. For morphogenetic factors, preparation of single-use aliquots at optimized concentrations prevents activity loss and ensures consistent patterning outcomes across experimental batches.
Emerging Technologies and Future Perspectives
Advanced technologies offer promising approaches for addressing persistent reproducibility challenges in organoid research. Automation and artificial intelligence are being increasingly integrated into organoid workflows to standardize protocols and remove human technical variability [54]. These systems enable standardized protocols that reduce variability and remove human bias from decision-making, ensuring cells receive consistent treatment throughout the differentiation process [54].
Multi-omics integration provides unprecedented resolution for characterizing organoid systems and identifying sources of heterogeneity. Recent advances in single-cell mass cytometry enable measurement of 28 signaling post-translational modifications across multiple cell types in millions of single organoid cells, revealing cell-type-specific signaling networks that underlie phenotypic variability [55]. Similarly, long-term live light-sheet microscopy of developing brain organoids enables tracking of tissue morphology, cell behaviors, and subcellular features over weeks of development, providing dynamic insights into morphogenetic processes that contribute to batch variations [33].
The emerging "Organoid plus and minus" framework combines technological augmentation with culture system refinement to improve screening accuracy, throughput, and physiological relevance [56]. This approach, coupled with advanced biofabrication strategies such as organ-on-chip integration and 3D bioprinting, addresses fundamental limitations in organoid vascularization, maturation, and physiological mimicry that contribute to experimental variability [20] [57]. As these technologies mature and become more widely adopted, they are expected to significantly enhance the reproducibility and translational relevance of organoid-based research platforms.
Addressing batch-to-batch variability and reproducibility challenges requires a systematic, multi-faceted approach spanning stem cell biology, protocol standardization, quality control implementation, and advanced analytical methodologies. By adopting the frameworks and recommendations presented in this application note, researchers can significantly enhance the reliability and translational potential of organoid technologies. Continued development of standardized protocols, defined culture systems, and robust characterization benchmarks will further advance the field toward consistent generation of high-fidelity organoid models that faithfully recapitulate in vivo biology while minimizing technical artifacts and inter-experimental variability.
Organoid technology, derived from human pluripotent stem cells (hPSCs), has revolutionized the study of human development and disease. However, widespread adoption in research and drug development is hampered by several persistent technical limitations. This Application Note addresses three critical challenges—necrotic core formation, incomplete functional maturation, and the retention of fetal-like phenotypes—by providing validated protocols and analytical frameworks to enhance organoid reproducibility and physiological relevance for pharmaceutical applications.
Section 1: Addressing Necrotic Core Formation
A major limitation in 3D organoid culture is the development of necrotic cores due to insufficient nutrient and oxygen diffusion into larger structures [58] [59]. This spontaneous necrosis complicates data interpretation, particularly in disease modeling where distinguishing experimental from technical artifacts is crucial [58].
Protocol 1.1: Generation of Necrotic Core-Free Human Spinal Cord Organoids (nf-hSCOs)
This protocol demonstrates how controlling initial aggregation size prevents necrotic core formation, enabling cleaner ischemia modeling [58].
Materials
- H9 hPSCs or equivalent pluripotent stem cell line
- DMEM/F12 and Neurobasal Medium (1:4 ratio for maturation)
- Neural Induction Supplements: 10 µM SB431542 (TGF-β inhibitor), 3 µM CHIR99021 (WNT activator)
- Matrigel-coated plates for initial cell culture
- 96-well low attachment plates for 3D aggregation
- Differentiation Factors: 20 ng/mL bFGF, 0.1 µM retinoic acid (RA)
- Rock inhibitor (Y-27632) for initial plating survival
Methodology
- hPSC Culture: Maintain H9 hPSCs on matrigel-coated plates in mTeSR1 medium, passaging every 5 days using ReLeSR [58].
- Caudal Neural Stem Cell Induction: Treat high-density hPSC clumps with 10 µM SB431542 and 3 µM CHIR99021 in differentiation medium for 3 days [58].
- Cell Dissociation and Aggregation: Dissociate cNSCs to single cells using Accutase. Seed exactly 75,000 cells per well in 96-well low attachment plates in differentiation medium supplemented with 20 ng/mL bFGF and 10 µM Rock inhibitor (day 1 only) [58].
- Initial Patterning: Culture aggregates for 4 days with daily bFGF treatment to promote neural fate [58].
- Regional Specification: Transfer organoids to medium containing 0.1 µM retinoic acid without bFGF for 6 days to pattern spinal cord identity, changing medium every other day [58].
- Organoid Maturation: Maintain nf-hSCOs in DMEM/F12:Neurobasal (1:4) mixture with 2% B27, 1% Penicillin/Streptomycin, 1% Glutamax, and 0.1 µM RA, changing medium every 4 days [58].
Validation Assessment
- Live/Dead Staining: Assess viability using propidium iodide (dead cells) and acridine orange (live cells) staining for 1 hour at 37°C. Calculate viability ratio from fluorescence intensities [58].
- Immunohistochemistry: Confirm regional identity with antibodies against caudal neural markers [58].
- Functional Testing: Plate organoids on matrigel-coated coverslips to assess axonal outgrowth capacity under control and experimental conditions [58].
Table 1: Impact of Initial Cell Number on Organoid Quality
| Initial Cell Number | Necrotic Core Presence | Viability Ratio (Live/Dead) | Modeling Applications |
|---|---|---|---|
| High (>100,000) | Frequent | 0.65 ± 0.15 | Limited - high background |
| 75,000 (optimized) | Absent | 0.92 ± 0.08 | Ischemia, drug screening |
| Low (<50,000) | Absent | 0.95 ± 0.05 | Limited - small size |
Section 2: Promoting Functional Maturation
Even after extended culture (≥6 months), brain organoids typically remain at fetal-to-early postnatal stages, limiting their utility for modeling adult-onset disorders [59]. Incomplete maturation affects multiple cellular domains, including synaptic refinement, gliogenesis, and network-level plasticity [59].
Protocol 2.1: Multidimensional Assessment of Organoid Maturity
Implement this comprehensive benchmarking framework to systematically evaluate maturation progress across multiple dimensions [59].
Structural Architecture Assessment
- Cortical Lamination Analysis: Validate layer-specific neurons using antibodies against SATB2 (upper layers II-IV), TBR1 and CTIP2 (deep layers V-VI) [59].
- Barrier Formation: Assess glia limitans development via aquaporin-4 expressing astrocyte endfeet at organoid periphery [59].
- Synaptic Ultrastructure: Confirm presynaptic (synaptobrevin-2) and postsynaptic (PSD-95) maturation using electron microscopy [59].
Functional Maturation Assessment
- Electrophysiological Recording: Utilize multielectrode arrays (MEAs) to detect synchronized network activity, γ-band oscillations, and spontaneous action potentials [59].
- Calcium Imaging: Map spatial activity patterns using GCaMP reporters expressed in neurons and astrocytes [59].
- Neurotransmitter Profiling: Validate neurotransmitter systems through immunostaining for VGLUT1 (glutamatergic), GAD65/67 (GABAergic), and HPLC detection of dopamine in midbrain organoids [60].
Molecular Profiling
- Single-Cell RNA Sequencing: Resolve cellular heterogeneity and identify maturation-associated transcriptional programs [59].
- Metabolic Analysis: Assess transition from glycolytic to oxidative metabolism using Seahorse extracellular flux analyzers [59].
Table 2: Multidimensional Benchmarking of Organoid Maturity
| Assessment Dimension | Key Markers/Parameters | Fetal Stage | Mature Stage |
|---|---|---|---|
| Cellular Diversity | Neurons (MAP2), Astrocytes (GFAP, S100β), Oligodendrocytes (MBP) | Limited progenitor populations | Diverse, subtype-specific populations |
| Synaptic Maturity | SYB2/PSD-95 clustering, Spine density, EM ultrastructure | Immature synapses, few spines | Mature synaptic densities, spine diversity |
| Network Function | MEA: Synchronized bursting, Oscillation patterns | Sporadic, uncoordinated activity | Synchronized network oscillations |
| Regional Identity | Forebrain (FOXG1), Midbrain (OTX2), Hindbrain (HOX genes) | Mixed regional identities | Distinct, region-appropriate markers |
Protocol 2.2: Bioengineering Strategies to Accelerate Maturation
Incorporate these bioengineering interventions to promote advanced maturation beyond developmental timelines [59].
Materials
- Microfluidic perfusion systems for enhanced nutrient/waste exchange
- Electrical stimulation platforms for activity-dependent maturation
- Synthetic hydrogel matrices with tunable mechanical properties
- Vascularization components: hPSC-derived endothelial cells and pericytes
Methodology
- Dynamic Culture Conditions: Implement microfluidic perfusion with controlled flow rates (0.1-1 mL/hour) to improve nutrient delivery and mimic circulatory systems [59].
- Activity-Dependent Maturation: Apply patterned electrical stimulation (0.1-2 Hz, 1-2 hours daily) to promote synaptic strengthening and network integration [59].
- Mechanotransduction Modulation: Culture organoids in synthetic hydrogels with brain-mimetic stiffness (0.1-0.5 kPa) to provide appropriate mechanical cues [59].
- Vascular Co-culture: Incorporate hPSC-derived endothelial cells and pericytes at day 20-30 of differentiation to promote vascular network formation [59].
Section 3: Overcoming Fetal Phenotypes
Organoids derived from hPSCs typically exhibit fetal-like characteristics even after prolonged culture, limiting their utility for modeling adult-onset diseases [61]. Quantitative assessment tools are essential for evaluating and improving organoid maturity.
Protocol 3.1: Quantitative Organ-Specific Similarity Analysis
The Web-based Similarity Analytics System (W-SAS) provides quantitative evaluation of organoid maturity through organ-specific gene expression panels [61].
Materials
- RNA-seq data from organoids (TPM, FPKM/RPKM values)
- W-SAS platform (https://www.kobic.re.kr/wsas/)
- Reference transcriptomes from target human organs
Methodology
- Transcriptome Profiling: Isolve high-quality RNA from organoids and perform RNA-seq analysis [61].
- Data Formatting: Normalize expression data to TPM or FPKM/RPKM values and prepare according to W-SAS specifications [61].
- Similarity Calculation: Input data into W-SAS to calculate organ-specific similarity percentages using pre-defined gene expression panels [61].
- Interpretation: Use similarity scores to guide protocol optimization, with scores >70% indicating advanced maturation [61].
Organ-Specific Gene Panels
- Heart-specific Panel (HtGEP): 144 genes
- Lung-specific Panel (LuGEP): 149 genes
- Stomach-specific Panel (StGEP): 73 genes
- Liver-specific Panel (LiGEP): Previously validated [61]
Table 3: Research Reagent Solutions for Advanced Organoid Generation
| Reagent/Category | Specific Examples | Function in Protocol | Considerations |
|---|---|---|---|
| Patterning Molecules | SB431542 (TGF-β inhibitor), CHIR99021 (WNT activator), Retinoic Acid | Regional specification, dorsal-ventral patterning | Concentration and timing critical for precise patterning |
| Extracellular Matrix | Matrigel, Synthetic hydrogels (GelMA) | 3D structural support, mechanical signaling | Batch variability in matrigel; synthetic alternatives improve reproducibility |
| Maturation Factors | BDNF, GDNF, NT-3 | Neurotrophic support, neuronal survival, synaptic maturation | Required for long-term maintenance and functionality |
| Metabolic Modulators | Galactose/FAO-promoting media | Shift from glycolytic to oxidative metabolism | Promotes adult-like metabolic profiles |
| Analysis Tools | W-SAS algorithm, scRNA-seq, MEA | Quantitative maturity assessment, functional characterization | Multimodal validation essential for comprehensive assessment |
Integrated Workflow for Enhanced Organoid Generation
This integrated protocol combines solutions for necrosis, maturation, and fetal phenotypes into a unified workflow for generating high-quality organoids suitable for drug development applications.
Temporal Workflow
- Days 0-7: Initiate controlled aggregation with optimized cell numbers (Protocol 1.1) and early patterning [58]
- Days 7-30: Implement bioengineering interventions (Protocol 2.2) while continuing regional specification [59]
- Days 30-90: Apply chronic maturation protocols with metabolic modulation and functional assessment [59] [61]
- Day 90+: Perform comprehensive multidimensional analysis including W-SAS scoring (Protocol 3.1) and functional validation [59] [61]
Quality Control Checkpoints
- Day 10-14: Assess initial patterning and absence of necrosis via live/dead staining [58]
- Day 30-40: Evaluate early network formation and cellular diversity via scRNA-seq [59]
- Day 60-90: Perform intermediate functional assessment using MEA and calcium imaging [59]
- Endpoint: Conduct comprehensive maturity benchmarking including organ-specific similarity scoring [61]
The protocols presented here provide systematic approaches to overcome the three major limitations in organoid technology. Through controlled aggregation, bioengineering maturation strategies, and quantitative assessment tools, researchers can generate more reproducible, physiologically relevant organoids that better recapitulate adult human tissue properties. These advances will enhance the predictive validity of organoid-based models in drug discovery and disease modeling applications.
Organoid technology has emerged as a transformative tool in biomedical research, enabling the in vitro modeling of human tissues with remarkable architectural and functional complexity. Derived from pluripotent stem cells (PSCs), including both embryonic and induced pluripotent stem cells, organoids self-organize into three-dimensional structures that recapitulate key aspects of their in vivo counterparts [62]. This technology has advanced studies in developmental biology, disease modeling, drug screening, and regenerative medicine [63] [62]. However, traditional immersion culture methods often face significant challenges, including lack of standardization, organoid fusion, hypoxia-induced necrosis, and high media consumption [63].
To address these limitations, innovative culture platforms have been developed, with air-liquid interface (ALI) and bioreactor systems at the forefront. The ALI method, which exposes the apical cell surface to air while the basal side interacts with culture medium, optimizes gas exchange and promotes enhanced cellular differentiation [62] [64]. Bioreactor systems, through controlled agitation and aeration, improve nutrient distribution and support the growth of larger, more complex organoid structures [62]. This application note details the implementation, advantages, and protocols of these advanced culture technologies within the context of PSC-derived organoid research.
Air-Liquid Interface (ALI) Culture
The ALI culture system mimics the physiological environment of hollow organs and epithelial barriers. In this setup, cells are typically seeded on a semi-permeable membrane that separates an apical air-exposed compartment from a basolateral medium-filled compartment [62] [64]. This configuration is particularly advantageous for modeling tissue-specific functions and studying barrier properties. For instance, when hiPSC-derived alveolar epithelial cells (iAEC2) are cultured at ALI, they adopt a type 2 alveolar cell-like phenotype, expressing surfactant proteins and developing lamellar bodies, which are essential for studying alveolar repair mechanisms [64].
A specific ALI adaptation, the AirLiwell technology, utilizes non-adhesive microwells on a semi-permeable membrane to generate and maintain individualized organoids at an air-liquid interface [63]. This method prevents organoid fusion, eliminates the need for continuous agitation, reduces media volume requirements, and is compatible with Good Manufacturing Practices (GMP), making it suitable for therapeutic applications [63].
Bioreactor Culture
Bioreactors provide a dynamic culture environment by precisely controlling parameters such as fluid flow, gas exchange, and nutrient delivery. Systems like the SpinΩ miniaturized rotating bioreactor enhance nutrient uptake and oxygen availability, supporting the derivation of iPSCs into various brain organoids (e.g., forebrain, midbrain, and hypothalamus) in a more compact design with reduced media costs [62]. These systems are instrumental in maintaining organoids in a highly undifferentiated state during long-term culture and enabling large-scale expansion of stem cells while preserving their pluripotency [62].
Table 1: Quantitative Comparison of Organoid Culture Technologies
| Parameter | Traditional Immersion (3D-i) | Air-Liquid Interface (3D-ALi) | Bioreactor Culture |
|---|---|---|---|
| Standardization | Low (high fusion rate) [63] | High (individualized organoids) [63] | Moderate to High [62] |
| Oxygenation | Limited, leading to hypoxia/necrosis [63] | Optimized via direct air exposure [63] | Enhanced via controlled aeration [62] |
| Agitation Requirement | Constant agitation needed [63] | No agitation required [63] | Continuous, controlled mixing [62] |
| Media Volume | High (3-4 mL/well) [63] | Reduced [63] | Variable, often optimized for scale [62] |
| Cellular Composition (Midbrain Organoid Example) | 61% Neural (49% Neurons), 39% Non-neural [63] | 99% Neural (86% Neurons) [63] | Data not provided in search results |
| Key Functional Readout | Heterogeneous electrophysiological activity [63] | Striking electrophysiological synchronization [63] | Supports complex structure formation [62] |
| Scalability & GMP Compatibility | Challenges in standardization [63] | High, GMP-compatible [63] | High, suitable for scale-up [62] [54] |
Application Notes & Experimental Protocols
Protocol: Generating Midbrain Organoids Using AirLiwell ALI Technology
This protocol adapts the AirLiwell method for generating standardized midbrain organoids from human pluripotent stem cells (hPSCs), based on demonstrated research [63].
Initial Cell Culture and Seeding
- hPSC Culture: Maintain hPSCs (e.g., HS420 cell line) in StemFlex medium on laminin 521-coated tissue culture flasks.
- Preparation for Differentiation: At 70% confluency, passage cells using Accutase and prepare for 3D culture.
- Seeding in AirLiwell Plates:
- Seed 2,000 hPSCs per microwell in an AirLiwell plate (~800 microwells per well of a 6-well plate) in supplemented X-VIVO medium.
- Gently shake the plate and place it on a stable, flat support for 15 minutes to ensure even cell distribution across microwells.
- Add the appropriate volume of supplemented X-VIVO medium to the reservoir beneath the insert to establish the air-liquid interface.
- Culture the plate at 37°C and 5% CO₂.
Differentiation and Maturation
- Neural Induction: The initial culture in X-VIVO medium is supplemented with dual-SMAD inhibitors (LDN193189 and SB431542) to promote neural induction.
- Midbrain Patterning: To direct midbrain fate, add specific morphogens including SHH (100 ng/mL), Purmorphamine (2 µM), and FGF-8 (100 ng/mL).
- Maturation: After several days, transition to a neurobasal-based medium supplemented with growth factors such as GDNF (20 ng/mL), BDNF (20 ng/mL), and cAMP (0.5 mM) to support neuronal maturation and dopaminergic specification.
- Maintenance: Culture the organoids for the desired duration (e.g., over 60 days), with periodic medium changes. Organoids remain individualized within the microwells without the need for transfer or agitation.
Protocol: Generating Cerebral Organoids Using a Bioreactor
This protocol outlines the key steps for generating cerebral organoids using a spinning bioreactor system, synthesizing information from established procedures [65] [62].
Embryoid Body (EB) Formation
- hPSC Dissociation: Dissociate hPSC colonies into a single-cell suspension using an enzyme such as TrypLE.
- Aggregation: Seed cells into an Aggrewell plate (3 million cells per well) in mTeSR medium supplemented with ROCK inhibitor (Y27632) and other small molecules (e.g., Trans-ISRIB, Chroman 1, Emricasan) to promote cell survival and inhibit differentiation.
- Centrifugation: Centrifuge the Aggrewell plate at 100 × g for 3 minutes to aggregate cells at the bottom of the microwells.
- EB Formation: Culture the plate for 24-48 hours to allow for EB formation.
Neural Induction and Differentiation
- EB Transfer: Carefully transfer the formed EBs to a bioreactor system containing neural induction medium (e.g., M1 medium containing DMEM, KOSR, and small molecule inhibitors like LDN and SB).
- Dynamic Culture: Maintain the EBs in the bioreactor under constant, gentle rotation to prevent aggregation and ensure even nutrient distribution.
- Matrigel Embedding (Optional): Around day 7, embed EBs in Matrigel droplets to provide structural support and enhance neuroepithelial budding, a step critical for forming complex cerebral organoid structures [65].
- Long-term Maturation: Culture the organoids in differentiation medium (e.g., a mix of DMEM/F12 and Neurobasal medium supplemented with N2 and B27) for extended periods (≥ 60 days), with medium changes performed periodically.
Key Signaling Pathways for Organoid Patterning
The directed differentiation of PSCs into region-specific organoids requires precise manipulation of key developmental signaling pathways. The diagram below summarizes the core signaling interactions for patterning neural organoids.
Figure 1: Signaling Pathway Control for Neural Organoid Patterning. Directed differentiation of PSCs into region-specific neural organoids is achieved by sequential modulation of key developmental signaling pathways. Initial neural induction is primarily driven by dual-SMAD inhibition. Subsequent dorsalization is promoted by BMP and WNT inhibition, ventralization by SHH activation, and caudalization by WNT, Retinoic Acid (RA), and FGF activation [62] [52].
The Scientist's Toolkit: Essential Research Reagents
Successful organoid generation relies on a carefully selected set of reagents and materials to support cell survival, direct differentiation, and mimic the native extracellular environment.
Table 2: Key Research Reagent Solutions for Organoid Culture
| Reagent Category | Specific Examples | Function & Application |
|---|---|---|
| Extracellular Matrices | Matrigel, Laminin-521, Collagen | Provides a 3D scaffold that supports cell attachment, polarization, and self-organization. Critical for embedding organoids in many protocols [65] [52]. |
| Small Molecule Inhibitors/Activators | LDN193189 (BMP inhibitor), SB431542 (TGF-β inhibitor), CHIR99021 (WNT activator), Purmorphamine (SHH agonist) | Precisely controls cell fate decisions by modulating key developmental signaling pathways (e.g., SMAD, WNT, SHH) for neural and regional specification [63] [65] [52]. |
| Growth Factors & Cytokines | SHH, FGF8, GDNF, BDNF, Activin A | Provides mitogenic and patterning cues to guide differentiation toward specific lineages (e.g., SHH/FGF8 for midbrain, GDNF/BDNF for neuronal maturation) [63] [66]. |
| Basal Media & Supplements | Neurobasal Medium, DMEM/F12, B-27 Supplement, N-2 Supplement, N-Acetylcysteine | Provides essential nutrients, hormones, and survival factors tailored to support the metabolic needs of neural and other specialized tissues [63] [65]. |
| Specialized Culture Platforms | Aggrewell Plates, AirLiwell Plates, SpinΩ Bioreactor | Enables forced aggregation of cells to form uniform EBs, and provides advanced culture conditions (ALI, dynamic flow) for improved organoid growth and standardization [63] [65] [62]. |
The integration of air-liquid interface and bioreactor technologies represents a significant leap forward in organoid research. The ALI method addresses critical limitations of traditional immersion culture by providing enhanced standardization, improved gas exchange, and reduced operational complexity [63]. Bioreactor systems facilitate the scalable production of more complex and mature organoid models through superior nutrient distribution and environmental control [62]. As these technologies continue to evolve—through integration with organs-on-chips, advanced vascularization strategies, and automated high-throughput systems—they are poised to further bridge the gap between in vitro models and in vivo physiology. This progress will undoubtedly accelerate drug discovery, enhance the fidelity of disease models, and advance the therapeutic application of PSC-derived organoids.
Leveraging AI and Automation for High-Throughput, Reproducible Production
The field of organoid research is undergoing a transformative shift, moving from manual, low-throughput protocols to fully automated, AI-driven production systems. This evolution addresses one of the most significant challenges in the field: the reproducibility and scalability of complex 3D biological models derived from pluripotent stem cells (PSCs) [67]. Organoids generated from human induced pluripotent stem cells (hiPSCs) offer unprecedented opportunities for modeling human development, disease pathology, and drug response in a physiologically relevant context [68] [39]. However, traditional manual culture methods are labor-intensive, time-consuming, and prone to technical variability, creating a critical bottleneck in pharmaceutical research and personalized medicine applications [67].
The integration of artificial intelligence (AI) and automation technologies is now revolutionizing organoid workflows by standardizing the entire process from stem cell differentiation to functional analysis. These advanced systems enable researchers to maintain precise control over culture conditions, timing, and differentiation cues while generating the large-scale, consistent organoid populations required for high-throughput drug screening and toxicology testing [69] [70]. This paradigm shift is particularly crucial for PSC-derived organoids, which require extended culture periods and complex, multi-stage differentiation protocols that are difficult to standardize across laboratories and operators [71].
The Need for Automation in PSC-Derived Organoid Research
Limitations of Traditional Organoid Culture Methods
Conventional organoid culture systems face several fundamental challenges that impede their widespread adoption in industrial-scale research and clinical applications. The technical complexity of culturing PSC-derived organoids includes labor-intensive processes such as seeding, feeding, passaging, and monitoring that demand significant expertise and manual handling [67]. These processes are particularly challenging for hiPSC-derived models that require precise temporal control of differentiation signals and careful quality assessment at multiple developmental stages [68] [72].
Reproducibility remains a substantial hurdle, with difficulties in standardizing organoid formation, size, cellular composition, and maturity across multiple wells or plates [67]. Studies have demonstrated that the same organoid protocol can generate different results across laboratories, and even experienced technicians struggle to maintain consistency batch-to-batch [71]. This variability directly impacts experimental outcomes and the reliability of data generated for drug discovery pipelines.
Scalability presents another critical limitation, as adapting complex 3D organoid models for high-throughput screening has proven challenging with manual methods [67]. The pharmaceutical industry requires hundreds to thousands of consistent organoids for comprehensive compound screening, a scale that is impractical with traditional culture techniques. Furthermore, the lack of standardization complicates comparative analyses and the establishment of quality control benchmarks essential for regulatory acceptance [71].
Market Drivers and Growth Projections
The global market for organoids and related technologies is experiencing rapid growth, driven by increasing demand for physiologically relevant models that bridge the gap between traditional 2D cell cultures and animal models [73] [74]. This expansion is particularly evident in drug discovery applications, where the high failure rate of drug candidates in clinical trials (approximately 90%) has created an urgent need for more predictive human-relevant models [67].
Table 1: Organoids and Spheroids Market Projections (2024-2034)
| Metric | 2024 Value | 2025 Value | 2034 Projected Value | CAGR (2025-2034) |
|---|---|---|---|---|
| Market Size | USD 1.5 billion [74] | USD 1.8 billion [74] | USD 4.0-9.6 billion [73] [74] | 10.7%-20.3% [73] [74] |
| Leading Segment | Organoids (76.2% share) [74] | - | - | 20.2% (forecasted) [74] |
| Key Growth Region | North America [74] | - | Asia Pacific (fastest growing) [74] | - |
| Primary End Users | Pharmaceutical & Biotechnology Companies (42.3% share) [74] | - | - | - |
This substantial market growth is fueled by several key factors, including advancements in stem cell research, 3D culture systems, and regenerative medicine [73]. The increasing use of organoids in disease modeling, personalized medicine, and drug discovery applications has accelerated adoption across academic, pharmaceutical, and biotechnology sectors [73] [74]. particularly for patient-derived organoid models that enable personalized treatment strategies and biomarker discovery [74].
Automated Platforms for Organoid Production
Integrated Automated Culture Systems
The CellXpress.ai Automated Cell Culture System represents a comprehensive approach to addressing the scalability and reproducibility challenges in PSC-derived organoid research. This integrated platform combines an automated incubator, liquid handling system, high-content imager, and robotic elevator for seamless plate movement [67] [69]. The system enables full automation of complex processes including cell seeding, feeding, passaging, imaging, and data analysis specifically designed for delicate stem cell-derived organoids [67].
A key advantage of this integrated system is its ability to maintain a 24/7 schedule for growing and scaling multiple stem cell lines, spheroids, or organoids without requiring manual intervention [69]. This continuous operation is particularly valuable for hiPSC-derived organoids that have demanding feeding and passaging schedules and can require extended differentiation periods spanning several weeks to months [68] [72]. The platform's capacity to process over 100 plates in parallel significantly increases the production of complex cell models, with reported gains of up to 25x compared to manual methods [69].
The system incorporates machine learning-assisted decision making that standardizes the development process to deliver consistent, unbiased, and biologically relevant results at scale [69]. This AI-driven approach allows the software to guide experimental timing based on image analysis, ensuring that critical maturation steps or feeding events occur at optimal timepoints [67]. The unified software environment provides workflow repeatability through automation and applies imaging-guided AI decision-making to ensure cell culture feeding and passaging events happen at the right time [69].
Table 2: Technical Specifications of the CellXpress.ai Automated Culture System
| Component | Specifications | Options |
|---|---|---|
| Incubator | Capacity | 44 plates (2 rack capacity), 154 plates (7 rack capacity), or 132 plates static/60 plates rocking (6-rack capacity) [69] |
| Environmental Control | CO₂, temperature, and humidity control [69] | |
| Decontamination | Automated hydrogen peroxide decontamination [69] | |
| Imager | Objectives | 2X through 40X objectives available [69] |
| Imaging Modes | Brightfield (TL) and up to 6 fluorescence (FL) channels [69] | |
| Acquisition Modes | Time lapse, Z-stack, optional Digital Confocal [69] | |
| Camera | 24 megapixel [69] | |
| Liquid Handler | Pipette Head | Span 8 [69] |
| Deck Locations | 9 positions with plate cooling, heating, and tilting capabilities [69] | |
| Cell Media Handling | Heated and cooled positions for low-volume and bulk media [69] | |
| System | Transfer Ports | 2 plate transfer ports [69] |
| Enclosure | HEPA filtered with anti-clog sensor [69] | |
| Dimensions | 2,250 mm W x 954 mm D x 2,155 mm H [69] |
AI-Powered Imaging and Analysis Systems
High-content imaging is essential for studying organoids because it delivers detailed 3D insights that 2D imaging methods cannot provide [67]. Platforms like the ImageXpress HCS.ai incorporate confocal imaging capabilities that allow researchers to penetrate organoid structures and visualize internal features, enabling the generation of 3D image stacks for volumetric analysis [67] [70]. This technology facilitates characterization of organoid shape, size, and density over time, as well as analysis of individual cell populations within an organoid to monitor viability, proliferation, and phenotypic changes [67].
These imaging systems are coupled with sophisticated AI algorithms, such as the SINAP (Single-Cell Image-Based Analysis of Organoids) deep learning model, which enables researchers to analyze large datasets generated from 3D organoid cultures rapidly and with remarkable precision [70]. This AI model helps identify subtle cellular changes that might otherwise be missed in manual analysis, a critical advantage when studying diseases with complex pathologies or assessing delicate differentiation patterns in PSC-derived organoids [70].
The integration of automated microscopy with AI-powered analysis creates a closed-loop system where imaging data can inform culture decisions in real-time. For example, machine learning-based decision-making allows users to distinguish between culture wells that are progressing as intended and those that aren't, enabling early intervention or removal of aberrant organoids from downstream processing [69]. This capability is particularly valuable for quality control in large-scale organoid production where manual assessment of every well would be impractical.
Experimental Protocols for Automated Organoid Generation
Protocol 1: Generation of Planar Hair-Bearing Skin Organoids from hiPSCs
This protocol outlines the automated generation of human induced pluripotent stem cell-derived planar skin organoids (SOs) containing hair follicles, providing a sophisticated in vitro model for investigating intercellular interactions within the skin microenvironment [68].
Detailed Methodology
Initial Cystic Skin Organoid Formation (Days 0-20)
- hiPSC Preparation: Culture hiPSCs in feeder-free conditions using defined mTeSR1 medium until 70-80% confluent in 6-well plates.
- Neural Ectoderm Induction: Replace medium with neural induction medium containing SB431542 (10 μM) and Noggin (100 ng/mL) for 5 days with daily medium changes using automated liquid handling.
- Surface Ectoderm Specification: Switch to defined keratinocyte medium containing BMP4 (25 ng/mL) and retinoic acid (10 μM) for 7 days with automated feeding every 48 hours.
- Cystic Organoid Formation: Transfer cells to low-adhesion 96-well plates in organoid formation medium containing FGF2 (20 ng/mL) and EGF (50 ng/mL). The CellXpress.ai system automatically performs medium exchanges every 3 days.
- Quality Checkpoint 1 (Day 20): Image each organoid using the 10X objective on the automated imaging system. Select only cystic organoids with smooth, spherical morphology and diameter between 300-500 μm for further processing.
Transition to Planar Configuration (Days 21-27)
- Air-Liquid Interface Transfer: Using automated liquid handling, transfer selected cystic SOs to 24-well transwell inserts pre-coated with collagen IV (10 μg/mL).
- Flattening Procedure: Carefully dissect cystic SOs using automated microsurgical tools or enzymatic digestion (collagenase IV, 1 mg/mL for 15 minutes at 37°C) to create planar configurations.
- ALI Culture Conditions: Maintain organoids at the air-liquid interface with specialized dermal-epidermal interaction medium in the basal chamber. The automated system replenishes medium from the basal side every 48 hours while maintaining the apical surface at air interface.
- Maturation: Culture planar SOs for minimum 27 total days to allow development of mature skin structures including stratified epithelium and hair follicle primordia.
Endpoint Analysis (Day 27+)
- Immunofluorescence Staining: Fix organoids with 4% PFA for 30 minutes at room temperature using automated processing.
- Permeabilize with 0.3% Triton X-100 for 15 minutes
- Block with 5% normal goat serum for 1 hour
- Incubate with primary antibodies (anti-KRT10, anti-LOR, anti-KRT15, anti-HOXC13) overnight at 4°C
- Apply fluorescent secondary antibodies for 2 hours at room temperature
- Image using high-content confocal imaging system with 20X objective
- Functional Assessment: Evaluate barrier function through dye exclusion assays and measure transepithelial electrical resistance (TEER) using automated electrode systems.
Quality Control Parameters
- Day 20 Selection Criteria: Cystic organoids must exhibit spherical morphology, diameter 300-500 μm, and smooth surface texture.
- Day 27 Success Metrics: Presence of pluristratified pigmented epithelium with skin appendages, including hair follicles and sebaceous gland-like structures confirmed by immunofluorescence.
- Exclusion Criteria: Organoids showing necrotic centers, irregular morphology, or diameter outside specified range at any checkpoint should be automatically flagged and removed from further culture.
Protocol 2: Generation of Vascularized Brain Organoids via Organoid Fusion
This protocol describes an automated approach for generating vascularized brain organoids by fusing separately induced brain organoids (BOrs) and vessel organoids (VOrs), creating an advanced model that incorporates neurovascular interactions and microglial cells [75].
Detailed Methodology
Brain Organoid (BOr) Generation (Days 0-20)
- hPSC Aggregation: Dissociate hPSCs to single cells and aggregate 9,000 cells per well in 96-well V-bottom ultra-low attachment plates in neural induction medium with ROCK inhibitor Y-27632 (10 μM).
- Neural Induction: Treat aggregates with dual-SMAD inhibitors (SB431542 at 10 μM and LDN-193189 at 100 nM) for 5 days with automated medium exchange every other day.
- Neural Patternning: Transfer aggregates to Matrigel droplets and culture in neural differentiation medium containing FGF2 (20 ng/mL) and EGF (20 ng/mL) for 15 days with automated feeding every 48 hours.
- Maturation: Maintain BOrs in neural maturation medium with BDNF (20 ng/mL) and GDNF (20 ng/mL) for extended culture periods up to 90 days with twice-weekly automated medium exchanges.
Vessel Organoid (VOr) Generation (Days 0-40)
- Mesoderm Induction: Aggregate hPSCs (9,000 cells per well) in V-bottom plates and treat with GSK3 inhibitor CHIR99021 (3 μM) for 2 days to induce mesoderm using automated liquid handling.
- Endothelial Differentiation: Treat embryonic bodies (EBs) with bFGF (20 ng/mL), VEGF (50 ng/mL), and BMP4 (25 ng/mL) for 3 days to promote vascular progenitor differentiation.
- Endothelial Maturation: Culture in endothelial cell growth medium (ECGM-MV2) containing VEGF (50 ng/mL) for 5 days using automated feeding.
- Matrix Embedding and Neural Conditioning: Embed VOrs in Matrigel droplets and culture in vessel maturation medium with N2 and B27 supplements to induce brain-specific vascular characteristics for 30 days with automated medium exchanges every 72 hours.
Organoid Fusion and Maturation (Days 21-60)
- Fusion Protocol: Select mature BOrs and VOrs (approximately 500 μm diameter) and combine in 1:1 ratio in fresh Matrigel droplets using automated picking and transfer systems.
- Fused Culture Conditions: Maintain fused organoids in combined neural-vascular medium (1:1 ratio of neural maturation and endothelial maturation media) for 30+ days with automated feeding every 48 hours.
- Quality Monitoring: Image fused organoids weekly using 10X confocal imaging to assess vascular integration and structural development.
Functional Assessment
- Calcium Imaging: Load organoids with Fluo-4 AM calcium indicator (5 μM) for 30 minutes at 37°C and image spontaneous calcium oscillations using high-speed confocal microscopy.
- Barrier Function Assays: Perfuse fused organoids with 10 kDa dextran-Texas Red conjugate and measure extravasation over time using automated image analysis.
- Immune Challenge: Treat organoids with lipopolysaccharide (LPS, 1 μg/mL) for 24 hours and monitor microglial activation and cytokine secretion using automated immunoassay systems.
Key Signaling Pathways and Molecular Regulation
The vascularized brain organoid protocol requires precise temporal control of several key developmental signaling pathways:
Wnt/β-catenin Signaling: Activated during initial mesoderm induction in VOr differentiation using CHIR99021 [75]. This pathway specifies mesodermal progenitors that give rise to vascular lineages.
BMP Signaling: Regulates both neural and vascular patterning. BMP4 is utilized during vascular progenitor differentiation and must be carefully titrated to avoid inhibition of neural fate specification [75] [72].
VEGF Signaling: Critical for endothelial differentiation and vascular network formation. VEGF concentration and timing must be optimized to promote robust vascularization without causing aberrant angiogenesis [75].
FGF Signaling: Supports both neural progenitor maintenance and endothelial cell proliferation. FGF2 is included in both neural and vascular culture media at specific developmental timepoints [75].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of automated organoid generation requires carefully selected reagents and specialized materials. The following table details essential components for PSC-derived organoid research, particularly focusing on the protocols described in this application note.
Table 3: Essential Research Reagents and Materials for Automated Organoid Generation
| Category | Specific Product/Reagent | Function/Application | Protocol Specificity |
|---|---|---|---|
| Stem Cell Maintenance | mTeSR1 or equivalent defined PSC medium | Maintenance of pluripotent stem cells in feeder-free conditions | General PSC culture [68] [72] |
| ROCK inhibitor (Y-27632) | Enhances single-cell survival after passaging | All protocols during initial cell aggregation [72] | |
| Induction Factors | CHIR99021 (GSK3 inhibitor) | Wnt pathway activation for mesoderm induction | Vessel organoid differentiation [75] [72] |
| SB431542 (TGF-β inhibitor) | Promotes neural crest and neural differentiation | Brain organoids, skin organoids [68] [72] | |
| BMP4 | Dorsal patterning, epidermal specification | Skin organoids (specific concentrations) [68] [72] | |
| VEGF | Endothelial differentiation and vascular maturation | Vessel organoids [75] | |
| FGF2 (bFGF) | Neural progenitor expansion, endothelial support | Brain and vessel organoids [75] | |
| Extracellular Matrices | Matrigel or similar ECM | 3D scaffold for organoid formation and fusion | All organoid protocols [75] |
| Collagen IV | Coating for planar culture configurations | Skin organoid ALI culture [68] | |
| Specialized Media | Neural induction media | Dual-SMAD inhibition for neural specification | Brain organoid protocol [75] |
| Endothelial cell media (ECGM-MV2) | Vascular maturation and maintenance | Vessel organoid protocol [75] | |
| Air-liquid interface media | Supports epidermal stratification and maturation | Skin organoid planar culture [68] | |
| Analysis Reagents | Lineage-specific antibodies (KRT10, LOR, CD31) | Immunofluorescence characterization of differentiation | All protocols for endpoint analysis [68] [75] |
| Calcium indicators (Fluo-4 AM) | Functional assessment of neuronal and cardiac activity | Brain and cardiac organoids [67] [75] | |
| Barrier function assays (dextran conjugates) | Measurement of blood-brain barrier integrity | Vascularized brain organoids [75] |
Applications and Future Perspectives
Current Research Applications
Automated production of PSC-derived organoids is already transforming several areas of biomedical research. In cancer research, patient-derived tumoroids are being used to evaluate drug responses in a personalized medicine context, with automated systems enabling high-throughput screening of therapeutic compounds [67]. Similarly, neurodegenerative disease modeling using neurospheroids is advancing the study of potential treatments for conditions such as Alzheimer's and Parkinson's disease [67]. The scalability afforded by automation makes it feasible to generate the large numbers of consistent neural organoids required for compound screening.
In toxicology assessment, high-throughput assays using cardiac organoids are improving the evaluation of drug safety for conditions like heart failure and arrhythmia [67]. Automated systems can simultaneously monitor beat rate, amplitude, and rhythmicity in hundreds of cardiac organoids, providing comprehensive cardiotoxicity profiles early in drug development. Likewise, liver organoids are gaining traction as alternative models for pharmaceutical toxicology, reducing reliance on animal testing while providing human-relevant data [67].
The integration of functional assays with automated organoid production is particularly valuable for capturing dynamic physiological responses. Calcium flux analysis in specific organoid models provides real-time insights into drug effects [67]. In cardiac organoids, this approach can measure beat rate, amplitude, and rhythmicity, while in neurospheroid assays, it can detect neuronal oscillations offering insights into the effects of neuroactive compounds [67].
Emerging Trends and Future Directions
The future of automated organoid production lies in the development of increasingly complex multi-tissue systems and the deeper integration of AI across the entire workflow. Several key trends are shaping this evolution:
Multi-Organoid Systems and Organ-on-Chip Integration: The emergence of multi-organoid systems and organ-on-chip platforms is enhancing the simulation of complex tissue interactions [74]. These advanced models are improving outcomes in drug screening, toxicity testing, and disease progression studies by capturing inter-organ communication and systemic effects [74]. The combination of organoid technology with microfluidic systems creates more physiologically relevant microenvironments while enabling controlled exposure to test compounds [71].
Organoid Atlases and Standardization Frameworks: Large-scale collaborative projects are addressing the critical challenge of standardization through the development of organoid atlases. Researchers from Roche's Institute of Human Biology, ETH Zurich, and Helmholtz Munich have created computational tools to integrate datasets from different protocols and labs into comprehensive "organoid atlases" [71]. These resources allow researchers to compare organoid cells across labs, protocols, and patient samples, establishing benchmarks for quality assessment and protocol optimization.
AI-Driven Predictive Modeling and Personalized Medicine: The integration of cutting-edge AI tools with organoid technology is creating unprecedented opportunities for personalized medicine. As noted by Professor Fabian Theis, "Fine-tuning the used protocols to individual needs via generative AI will bring us closer to personalized medicine, enabling the creation of organoids tailored to individual patients and specific therapeutic needs" [71]. This approach is particularly valuable for rare diseases and conditions with significant patient-to-patient variability.
Advanced Vascularization and Immune System Integration: Next-generation organoid protocols are increasingly incorporating non-parenchymal cell types such as endothelial cells and immune cells to create more complete tissue models [75]. The development of vascularized brain organoids containing functional blood-brain barrier-like structures and microglial cells represents a significant advancement [75]. Similar approaches are being applied to other organ systems to enhance maturity, functionality, and physiological relevance.
The convergence of automation, AI, and advanced bioengineering is poised to accelerate the adoption of organoid technologies across basic research, drug discovery, and clinical applications. As these systems become more sophisticated and accessible, they have the potential to transform how we model human biology, develop new therapeutics, and implement personalized medicine approaches.
Benchmarking Fidelity: How Organoids Compare to Traditional Models and Human Tissues
The transition from traditional two-dimensional (2D) cell cultures to three-dimensional organoids represents a paradigm shift in biomedical research. Organoids, which are self-organizing 3D structures derived from pluripotent stem cells (PSCs), closely mimic the architecture and functionality of human organs. This advancement addresses critical limitations of 2D cultures, particularly their lack of physiological context, which often leads to poor translation of preclinical findings to clinical outcomes [76] [77]. For researchers focused on PSC-derived organoid generation, understanding the comparative advantages and applications of these models is essential for designing biologically relevant experiments.
This application note provides a structured comparison of organoids and 2D cultures, detailing their experimental workflows, key signaling pathways, and reagent solutions. It aims to equip scientists with the tools to implement organoid models effectively in studies of development, disease modeling, and drug discovery.
Comparative Analysis of Physiological Relevance
Table 1: Quantitative and Qualitative Comparison of 2D vs. Organoid Cultures
| Feature | 2D Cell Cultures | Organoid Cultures |
|---|---|---|
| Spatial Architecture | Monolayer; forced polarity | 3D structure; self-organized tissue-like organization [76] |
| Cell-Cell Interactions | Limited to horizontal contacts | Multidirectional; includes stem/progenitor niches [76] |
| Gene Expression | Altered due to plastic substrate | In vivo-like profiles; better functional differentiation [76] [20] |
| Drug Response | Often overestimates efficacy; misses resistance | Predicts clinical response; models resistance mechanisms [76] [20] |
| Physiological Gradients | Absent | Oxygen, nutrient, and pH gradients present (e.g., hypoxic cores) [76] |
| Throughput & Cost | High-throughput; low cost per sample [76] | Medium-throughput; higher cost and technical demand [78] [54] |
| Applications | Primary screening, genetic manipulation [76] | Disease modeling, personalized therapy, developmental studies [20] [78] [79] |
Table 2: Functional Outcomes in Disease Modeling
| Disease Area | 2D Culture Limitations | Organoid Advantages |
|---|---|---|
| Cancer | Fails to model tumor microenvironment [76] | Recapitulates hypoxic cores, drug penetration barriers [76] [20] |
| Neurodegenerative Disorders | Limited cellular diversity | Models blood-brain barrier, neuronal networks [79] |
| Genetic Diseases | Poor phenotype representation | Patient-derived organoids mirror disease pathophysiology [72] [79] |
| Infectious Diseases | Altered host-pathogen interactions | Authentic modeling of viral infection (e.g., SARS-CoV-2) [79] |
Experimental Protocols for Organoid Generation from PSCs
Protocol 1: Directed Differentiation of PSCs into Jawbone-like Organoids
Background: This protocol models mandibular bone development using iPSCs, demonstrating the capacity to recapitulate craniofacial structures and disease phenotypes like osteogenesis imperfecta [72].
Steps:
- 3D Aggregation of iPSCs:
- Seed dissociated iPSCs into V-bottom ultra-low-adhesion 96-well plates.
- Use medium supplemented with ROCK inhibitor (Y-27632) to enhance cell survival.
Neural Crest Cell (NCC) Induction:
- Treat aggregates with BMP4 (10 ng/mL) for 24 hours to suppress neuroectoderm fate.
- Replace medium with TGF-β inhibitor (SB431542) and GSK3β inhibitor (CHIR99021) in shaking culture for 4 days.
- Validate NCC induction via flow cytometry for CD271 (≥95% purity) and immunostaining for SOX10/TFAP2A [72].
Mandibular Ectomesenchyme (mdEM) Patterning:
- Stimulate NCCs with FGF8 and endothelin-1 (Edn1) to drive mdEM specification.
- Confirm proximal-distal patterning using markers (DLX5, HAND2).
Osteogenic Differentiation and Organoid Maturation:
- Transfer mdEM to osteogenic medium (ascorbic acid, β-glycerophosphate, dexamethasone).
- Culture for 21–28 days to form mineralized bone matrices with embedded osteocytes.
- Analyze via Alizarin Red staining and 3D confocal imaging of osteocyte networks.
Validation:
- RNA sequencing to confirm HOX-negative NCC identity.
- Transplantation into jawbone defects to assess regenerative capacity [72].
Protocol 2: Cerebral Organoid Generation for Blood-Brain Barrier (BBB) Modeling
Background: BBB assembloids enable study of neurovascular interactions and drug penetration [79].
Steps:
- Dual Differentiation of iPSCs:
- Differentiate iPSCs into neural progenitor cells (NPCs) and endothelial cells (ECs) in parallel.
- NPCs: Use dual-SMAD inhibition (SB431542, LDN-193189).
- ECs: Activate WNT signaling with CHIR99021.
3D Assembly and Fusion:
- Combine NPC and EC aggregates in low-adhesion plates.
- Culture in VEGF-supplemented medium to promote vascular invasion.
Maturation and Functional Assay:
- Maintain assembloids under dynamic culture (orbital shaking) for 30 days.
- Assess barrier integrity via TEER measurements and immunostaining for claudin-5/ZO-1 [79].
Applications:
- Modeling stroke, neurodegenerative diseases, and metastatic brain invasion.
- High-content screening for CNS drug candidates.
Signaling Pathways in Organoid Self-Organization
Diagram 1: Key Signaling Pathways in Neural Crest-Derived Organoid Patterning
Short Title: Jawbone Organoid Patterning Pathway
Diagram 2: Workflow for Organoid Generation from PSCs
Short Title: PSC to Organoid Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for PSC-Derived Organoid Generation
| Reagent Category | Example Products | Function |
|---|---|---|
| Stem Cell Maintenance | mTeSR1, StemFlex Medium | Supports pluripotency and expansion of PSCs [72] |
| Induction Molecules | BMP4, SB431542 (TGF-β inhibitor), CHIR99021 (GSK3β inhibitor) | Directs lineage specification (e.g., neural crest) [72] |
| 3D Scaffolds | Matrigel, synthetic PEG hydrogels | Provides ECM-like structure for self-organization [76] [78] |
| Patterning Factors | FGF8, endothelin-1, retinoic acid | Establishes anterior-posterior and proximal-distal axes [72] |
| Validation Tools | CD271 antibodies, SOX10 reporters, TEER instruments | Quality control for differentiation efficiency and barrier function [72] [78] |
| Maturation Supplements | Ascorbic acid, β-glycerophosphate | Promotes mineralization in bone organoids [72] |
Organoids derived from pluripotent stem cells offer a physiologically relevant alternative to 2D cultures by preserving tissue architecture, cellular heterogeneity, and functional gradients. While 2D models remain useful for high-throughput screening, organoids excel in disease modeling, drug testing, and personalized medicine. The protocols and tools outlined here provide a foundation for integrating organoid technology into research workflows, enabling more predictive and human-relevant outcomes in biomedical science.
Advanced Note: Emerging technologies such as organoids-on-chips and automated AI-driven culture systems are poised to address current challenges in vascularization, scalability, and functional maturation [54] [80].
The Role of Organoid Atlases for Standardized Characterization and Quality Control
The advent of three-dimensional (3D) organoid technology represents a paradigm shift in biomedical research, providing in vitro models that recapitulate the cellular complexity and functional properties of native human organs. These self-organizing structures, derived from human pluripotent stem cells (hPSCs), enable studies of human development, disease modeling, and drug response in a human-specific context [20]. However, the translational potential of organoids has been hampered by significant challenges in protocol standardization and quality control. Different laboratories employing varying differentiation protocols generate organoids with considerable compositional heterogeneity, making cross-study comparisons difficult and impeding reproducible research outcomes [71].
To address these challenges, the scientific community has initiated the development of comprehensive organoid atlases—integrated reference frameworks that systematically characterize the cellular composition and molecular states of organoids across diverse protocols. These atlases, built primarily through single-cell RNA sequencing (scRNA-seq) technologies, provide quantitative benchmarks for assessing organoid fidelity relative to primary human tissues [15] [81]. By mapping organoid cell types to their in vivo counterparts, these resources establish much-needed standards for evaluating the strengths and limitations of different organoid protocols, thereby enabling more rigorous quality control measures and accelerating the adoption of organoid technologies in drug discovery and personalized medicine [71].
Quantitative Characterization of Organoid Atlases
Scale and Composition of Existing Atlas Initiatives
Recent large-scale collaborative efforts have yielded organoid atlases of unprecedented scale and resolution. The Human Neural Organoid Cell Atlas (HNOCA) integrates 36 single-cell transcriptomic datasets spanning 26 distinct protocols, encompassing more than 1.7 million cells from organoids ranging from 7 to 450 days in culture [15]. Similarly, the Human Endoderm-Derived Organoid Cell Atlas (HEOCA) incorporates data from 218 samples across 9 different tissues, totaling nearly one million cells [82]. These integrated resources capture the remarkable heterogeneity of cell types generated across laboratories and differentiation methods, enabling systematic comparison of protocol efficacy.
Table 1: Scale and Scope of Major Organoid Atlas Initiatives
| Atlas Initiative | Biological System | Number of Cells | Number of Protocols | Number of Datasets | Reference Tissues |
|---|---|---|---|---|---|
| HNOCA [15] | Neural system | >1.7 million | 26 | 36 | Developing human brain |
| HEOCA [82] | Endoderm-derived organs | ~1 million | 55 publications | 218 samples | Fetal and adult endodermal tissues |
| HCA|Organoid Pilot [81] | Colon and brain | Population-scale | Multiple | 200+ individuals | Primary colon and brain tissue |
Analytical Framework for Quality Assessment
Organoid atlases employ sophisticated computational pipelines to quantify organoid fidelity and composition. The analytical framework typically involves:
- Data integration and batch correction: Advanced computational methods such as scPoli are used to harmonize data across different laboratories and protocols, effectively removing technical artifacts while preserving biological signals [15] [82].
- Reference mapping: Organoid cells are projected into reference spaces defined by primary tissue atlases (e.g., developing human brain, fetal and adult organs) to enable direct comparison between in vitro and in vivo cell states [15].
- Quantitative fidelity metrics: Algorithms calculate transcriptomic similarity scores and presence scores to determine how closely organoid cell types resemble their primary counterparts and which primary cell types are adequately represented across protocols [15].
This analytical framework allows researchers to move beyond qualitative assessments to quantitative evaluations of organoid quality, identifying specific cell types that are consistently well-represented or problematic across different differentiation methods.
Protocol for Atlas-Based Organoid Characterization
Experimental Workflow for Single-Cell Profiling
Implementing quality control using organoid atlases requires a standardized workflow for sample processing and data generation:
- Organoid generation: Generate neural organoids from hPSCs using either guided (e.g., dorsal telencephalic, ventral telencephalic, midbrain patterns) or unguided protocols. Maintain careful documentation of differentiation timeline and culture conditions [15].
- Sample preparation: At appropriate time points (typically spanning early development to maturation stages), dissociate organoids into single-cell suspensions using enzymatic and mechanical methods.
- Library preparation and sequencing: Process cells using standard scRNA-seq protocols (e.g., 10x Genomics, Smart-seq2). Target a sequencing depth of 50,000-100,000 reads per cell to ensure adequate transcript detection.
- Data preprocessing: Perform quality control to remove low-quality cells (high mitochondrial content, low unique gene counts) and doublets using tools such as CellRanger or Seurat.
- Atlas integration: Upload processed data to the Organoid Cell Atlas Portal (when publicly available) or implement computational pipelines to project new data into existing atlas frameworks [81].
Diagram 1: Experimental workflow for organoid characterization
Computational Analysis Pipeline
The computational assessment of organoid quality involves multiple steps that compare new datasets to established atlas references:
- Data integration: Use scArches or similar algorithms to map new organoid scRNA-seq data into the pre-integrated atlas space, effectively comparing new samples to the collective reference [15].
- Cell type annotation: Transfer labels from the reference atlas to new cells based on transcriptional similarity, identifying both expected target cell types and off-target populations.
- Fidelity assessment: Calculate similarity metrics between organoid cells and their primary tissue counterparts, identifying potential aberrant metabolic states or stress responses that commonly distinguish in vitro systems [15].
- Compositional analysis: Quantify the proportions of different cell types present and compare to reference organoids or primary tissues to identify significant deviations that may indicate protocol-specific biases.
Table 2: Key Computational Tools for Atlas-Based Quality Assessment
| Tool Name | Function | Application in Quality Control | Reference |
|---|---|---|---|
| scPoli | Label-aware data integration | Harmonizes data across batches and protocols | [15] |
| scArches | Reference mapping | Projects new data into established atlas frameworks | [15] |
| RSS | Reference similarity spectrum | Quantifies similarity to primary tissue references | [15] |
| Neighborhood graph correlation | Cell state similarity | Compares organoid cell states to primary counterparts | [82] |
Application in Quality Control and Protocol Development
Assessing Protocol Efficacy and Reproducibility
Organoid atlases enable systematic evaluation of differentiation protocol performance across multiple dimensions:
- Target specificity: Atlas analyses reveal that while guided protocols generally enrich for targeted brain regions, they frequently show increased proportions of cells from neighboring regions. For example, midbrain organoid protocols often generate substantial populations of hindbrain neurons, indicating imperfect regional specification [15].
- Protocol precision: Comparative studies demonstrate that different protocols can generate similar cell types but in dramatically different proportions, explaining much of the variability observed across laboratories [71].
- Temporal maturation: Mapping organoids to developing human brain references shows that they typically recapitulate early to mid-fetal developmental stages, with limited progression to later maturation states observed in some long-term cultures [15].
Identification of Systematic Limitations
The atlas approach has revealed consistent challenges across neural organoid protocols:
- Missing cell types: Certain primary cell populations remain substantially under-represented across most protocols, including thalamic reticular nucleus GABAergic neurons, dorsal midbrain GABAergic neurons, and cerebellar Purkinje cells [15].
- Non-neural lineages: Organoids generally lack vascular endothelial cells, immune cells, and other non-neuroectodermal lineages that play crucial roles in brain development and function [15].
- Metabolic disturbances: A universal feature distinguishing organoid neurons from their primary counterparts is altered metabolic signatures, particularly features associated with glycolysis, likely reflecting adaptation to in vitro culture conditions [15].
Diagram 2: Quality control cycle for protocol optimization
Research Reagent Solutions for Organoid Characterization
Successful implementation of organoid quality assessment requires specific research tools and reagents. The following table details essential solutions for generating and characterizing organoids within an atlas framework:
Table 3: Essential Research Reagents for Organoid Quality Assessment
| Reagent Category | Specific Examples | Function in Workflow | Considerations |
|---|---|---|---|
| Stem Cell Media | mTeSR, StemFlex | Maintenance of hPSCs | Choose xeno-free formulations for clinical relevance [72] |
| Neural Induction Factors | SB431542, CHIR99021, BMP4 | Directed differentiation of neural lineages | Concentration and timing critically impact regional patterning [72] |
| Patterning Morphogens | FGF8, EDN1, RA | Regional specification (e.g., midbrain, mandibular) | Combinatorial signals often required for precise patterning [72] |
| Dissociation Enzymes | Accutase, TrypLE | Generation of single-cell suspensions | Optimization needed to maximize viability and minimize stress responses |
| scRNA-seq Kits | 10x Genomics Chromium | Single-cell library preparation | Consider cell throughput and sequencing depth requirements |
| Cell Surface Markers | CD271 (NGFR) | Assessment of neural crest induction efficiency | Enables FACS purification of specific progenitor populations [72] |
Future Directions and Implementation Recommendations
The development of organoid atlases represents a transformative advancement in stem cell research, but several challenges remain for widespread implementation. Current atlases primarily focus on transcriptomic characterization, while future iterations will need to incorporate epigenomic, proteomic, and spatial profiling data to provide more comprehensive quality metrics [81]. Additionally, as organoid protocols evolve to include more complex features such as vascularization, immune cell integration, and multi-tissue interfaces, atlases must correspondingly expand to encompass these advancements.
For research groups implementing organoid quality control based on existing atlases, we recommend:
- Regular benchmarking: New organoid batches should be routinely profiled and compared to atlas references to monitor protocol drift and maintain experimental consistency.
- Multi-level assessment: Combine molecular profiling (scRNA-seq) with functional assays and histological validation to ensure comprehensive quality evaluation.
- Community engagement: Contribute high-quality datasets to public atlas initiatives to expand their diversity and utility, promoting standardized quality metrics across the field.
As these resources mature and become more accessible through platforms like the Organoid Cell Atlas Portal [81], they will increasingly serve as essential tools for ensuring reproducibility, validating disease models, and ultimately strengthening the translational potential of organoid technology in drug development and personalized medicine.
Patient-derived organoids (PDOs) have emerged as a transformative model in precision oncology, demonstrating a remarkable ability to recapitulate patient-specific drug responses. These self-organizing, three-dimensional structures preserve the genetic and histological characteristics of original tumors, providing a powerful platform for in vitro drug screening. This application note details the robust correlation between PDO drug sensitivity and clinical outcomes, with a specific focus on methodologies derived from pluripotent stem cell (PSC) research. We provide validated experimental protocols for establishing PSCs-derived organoid models, quantitative data linking in vitro results to patient survival, and essential guidelines for implementing this technology in drug development and personalized treatment planning.
The high failure rate of cancer therapies, often attributable to inter-patient heterogeneity, underscores the critical need for predictive preclinical models. While traditional 2D cell lines have contributed extensively to pharmacogenomic databases, they often fail to capture the complexity of the tumor microenvironment [83]. Organoids generated from pluripotent stem cells (PSCs)—including embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs)—represent a physiologically relevant alternative. By applying developmental biology principles, such as the timed manipulation of Wnt, FGF, TGF-β/BMP, and retinoic acid signaling pathways, PSCs can be directed to self-organize into complex, three-dimensional tissues that mimic their in vivo counterparts [84] [39]. This capacity to model human physiology and disease positions PSC-derived organoids as a premier tool for predicting clinical drug efficacy and advancing personalized medicine.
Key Evidence: Quantitative Correlation with Clinical Outcomes
Substantial evidence now confirms that drug sensitivity profiles from PDOs can predict patient responses. The following table summarizes key quantitative findings from recent studies that correlate organoid responses with clinical outcomes.
Table 1: Correlation Between Organoid Drug Responses and Clinical Outcomes
| Cancer Type | Therapeutic Agent(s) | Organoid Response Metric | Clinical Correlation Finding | Source / Model |
|---|---|---|---|---|
| Colorectal Cancer | 5-Fluorouracil, Oxaliplatin | Predicted Sensitivity Score | Fine-tuned model hazard ratios (HR) for sensitive vs resistant: 5-FU: HR=3.91; Oxaliplatin: HR=4.49 [83] | PharmaFormer AI Model (Cell line + Organoid data) [83] |
| Colorectal Cancer | 5-FU, Oxaliplatin, Irinotecan, Cetuximab | IC50 / Inhibition Rate | PDO responses correlated with clinical outcomes in most cases; RAS-mutant organoids were resistant to cetuximab [85] | Patient-Derived Organoids (PDOs) [85] |
| Bladder Cancer | Gemcitabine, Cisplatin | Predicted Sensitivity Score | Fine-tuned model hazard ratio (HR) for Gemcitabine: HR=4.91 [83] | PharmaFormer AI Model (Cell line + Organoid data) [83] |
| Ovarian Cancer (HGSOC) | Carboplatin, PARP inhibitors, 19 other FDA drugs | Drug Sensitivity | PDTO sensitivity correlated with clinical outcomes; BRCA1 mutant PDTO showed expected resistance [86] | Patient-Derived Tumor Organoids (PDTOs) [86] |
Experimental Protocols
Protocol: Directed Differentiation of PSCs into Colonic Organoids
This protocol outlines the key developmental steps for generating colonic organoids from human PSCs, leveraging conserved signaling pathways that guide embryonic gut development [84].
Principle: Sequential activation and inhibition of key morphogenetic signaling pathways (Wnt, FGF, BMP, EGF) direct PSCs through definitive endoderm, posterior gut, and ultimately colonic organoid fate.
Materials:
- Source Cells: Human ESCs or iPSCs (maintained in a pluripotent state under feeder-free conditions).
- Key Signaling Molecules:
- Activin A: A Nodal mimetic for inducing definitive endoderm.
- Wnt3a (Recombinant) & CHIR99021 (GSK3β inhibitor): To activate Wnt signaling and posteriorize endoderm.
- FGF4 & FGF2: For posterior pattern specification and proliferation.
- EGF: Promoves epithelial growth and survival.
- BMP Inhibitors (e.g., Noggin, LDN-193189): To suppress ventral/anterior fates and promote intestinal specification.
- R-spondin 1: Potentiates Wnt signaling and supports intestinal stem cell maintenance.
- A83-01 (TGF-β inhibitor): Inhibits mesenchyme differentiation and supports epithelial growth.
Methodology:
- Definitive Endoderm Induction:
- Culture PSCs as single cells and aggregate into suspension.
- Treat with Activin A (100 ng/mL) in a low-serum medium for 3 days to induce definitive endoderm. Confirm by flow cytometry for SOX17 and FOXA2 (>80% positive cells).
Posterior Gut Patterning:
- Transfer aggregates to a 3D extracellular matrix (e.g., Matrigel).
- Culture for 4 days in medium containing CHIR99021 (3 µM) and FGF4 (500 ng/mL) to specify mid/hindgut fate. Spheroids will form and express CDX2.
Colonic Organoid Maturation:
- Maintain hindgut spheroids in Matrigel domes.
- Culture for 6-8 weeks in expansion medium containing EGF (50 ng/mL), Noggin (100 ng/mL), R-spondin 1 (conditioned medium or recombinant), FGF2 (100 ng/mL), and A83-01 (500 nM).
- Change medium every 3-4 days. Passage organoids every 2-3 weeks by mechanical dissociation and re-embedding in fresh Matrigel.
QC Checkpoint: Mature organoids should exhibit a cystic or dense morphology with a central lumen and express colonic epithelial markers (e.g., CK20, SATB2) by immunohistochemistry.
Protocol: Drug Sensitivity Assay in PSC-Derived Organoids
This standardized protocol assesses drug efficacy in established organoids, generating IC50 values for correlation with clinical data.
Materials:
- Mature PSC-derived organoids (as from Protocol 3.1)
- Test Compounds: Chemotherapeutics (e.g., 5-FU, Oxaliplatin) and targeted agents (e.g., Cetuximab)
- Extracellular Matrix: Matrigel, Growth Factor Reduced
- Organoid Culture Medium: Advanced DMEM/F12 supplemented with essential factors (see Table 2)
- Cell Viability Assay Kit: e.g., CellTiter-Glo 3D
Methodology:
- Organoid Preparation and Seeding:
- Harvest mature organoids and dissociate into small fragments or single cells using a gentle dissociation reagent.
- Resuspend the cell/organoid fragment pellet in cold Matrigel and seed 10-20 µL drops into a multi-well plate. Allow to polymerize at 37°C for 20-30 minutes.
- Overlay with warm organoid culture medium. Culture for 3-5 days to allow re-formation.
Drug Treatment:
- Prepare a serial dilution of the drug(s) of interest in culture medium (typically a 6-8 point, 1:10 dilution series).
- Remove the existing medium and add the drug-containing medium to the organoid cultures.
- Include a DMSO vehicle control (0.1% v/v).
- Incubate organoids with drugs for 5-7 days, refreshing the drug-medium every 2-3 days.
Viability Quantification and Analysis:
- After incubation, equilibrate the plate to room temperature.
- Add an equal volume of CellTiter-Glo 3D reagent to each well.
- Shake the plate for 5 minutes to induce lysis and then incubate for 25 minutes to stabilize the luminescent signal.
- Record luminescence using a plate reader.
- Normalize the luminescence of drug-treated wells to the DMSO control wells. Plot the percentage viability against the log10 of drug concentration and fit a dose-response curve to calculate the IC50 value.
Table 2: Key Research Reagent Solutions for PSC-Derived Organoid Work
| Reagent Category | Specific Examples | Function in Organoid Culture |
|---|---|---|
| Source Cells | Human ESCs, iPSCs | The starting cellular material with pluripotent capacity to differentiate into any germ layer. |
| Signaling Molecules | Activin A: Definitive Endoderm induction. Wnt3a & R-spondin: Posterior patterning & stem cell maintenance. Noggin: BMP inhibition for anterior foregut or intestinal specification. EGF: Epithelial growth and survival factor. FGF2, FGF4, FGF10: Patterning and growth of various organoids. | Recapitulate the developmental signaling microenvironment to direct differentiation and maintain tissue-specific stem cells. |
| Extracellular Matrix | Engelbreth-Holm-Swarm (EHS) Matrix (e.g., Matrigel, Cultrex BME) | Provides a 3D scaffold that supports complex tissue architecture and provides essential biochemical cues. |
| Basal Medium | Advanced DMEM/F12 | A nutrient-rich base medium optimized for low-serum and serum-free cell cultures. |
| Critical Supplements | B-27 & N-2 Supplements: Provide hormones, lipids, and proteins. N-Acetylcysteine: Antioxidant. Nicotinamide: Promotes epithelial differentiation. [1-2%] | A chemically defined supplement that supports the survival and growth of neural and other cell types. |
| Small Molecule Inhibitors | Y-27632 (ROCK inhibitor): Reduces apoptosis in dissociated cells. A83-01: Inhibits TGF-β signaling. SB202190: p38 MAPK inhibitor. | Enhances cell survival after passaging and helps maintain a proliferative state by inhibiting differentiation pathways. |
Workflow and Pathway Diagrams
Diagram: Predictive Clinical Drug Response Workflow
Diagram 1: From PSCs to Clinical Prediction. This workflow integrates PSC-derived organoid generation with AI modeling to predict patient-specific drug responses.
Diagram: Key Signaling Pathways in PSC to Gut Organoid Differentiation
Diagram 2: Signaling in Gut Organoid Development. Key developmental signaling pathways guide PSC differentiation into regional-specific gut organoids.
The integration of PSC-derived organoid technology with advanced computational models represents a paradigm shift in predictive oncology. The robust correlation between in vitro organoid drug sensitivity and clinical patient outcomes, as evidenced by hazard ratios and drug response patterns, provides a compelling validation of this approach. By adhering to the detailed protocols, reagent guidelines, and standardized workflows outlined in this document, researchers and drug developers can leverage this powerful technology to de-risk drug development pipelines, discover novel biomarkers, and ultimately, realize the promise of truly personalized cancer therapy.
The landscape of preclinical research is undergoing a profound transformation, driven by the convergence of ethical principles and regulatory modernization. The 3Rs principles (Replacement, Reduction, and Refinement), first articulated by Russell and Burch, have gained widespread recognition as foundational guidelines for humane animal research [87]. Simultaneously, the FDA Modernization Act 2.0, passed in late 2022, has authorized the use of non-animal alternatives for investigational new drug applications, marking a pivotal regulatory shift [88]. These frameworks are increasingly aligned in their support for New Approach Methodologies (NAMs) that leverage advanced technologies such as human pluripotent stem cells (hPSCs) and organoids [89] [20].
This paradigm shift is particularly evident in organoid generation from pluripotent stem cells, which offers unprecedented opportunities to model human development and disease with enhanced physiological relevance. Organoid technology not only provides human-relevant models that can improve the predictive power of preclinical drug development but also directly supports the ethical aims of the 3Rs by reducing reliance on animal experimentation [20]. The subsequent sections explore the regulatory and ethical foundations of this transition and provide detailed protocols for implementing these advanced models in research workflows.
Regulatory and Ethical Foundations
The 3Rs Principles: Evolving Interpretations
The 3Rs framework has evolved significantly since its original conception. Contemporary interpretations, shaped by advances in our understanding of animal sentience and scientific technological capabilities, offer clarified definitions:
Replacement: This principle now means "to conduct research that completely avoids the use of animals in scientific investigation, regulatory testing, and education" [87]. This expanded definition encompasses not only absolute replacement but also the development of proactive non-animal approaches that may open unprecedented research avenues, moving beyond simple one-to-one substitutes for animal tests.
Reduction: This refers to methods that minimize the number of animals required to obtain information of given precision while maintaining scientific validity [87]. Advanced organoid systems enable high-throughput screening approaches that can generate robust data while potentially reducing animal use in preliminary studies.
Refinement: This involves modifying procedures to minimize pain, suffering, and distress while enhancing animal welfare throughout their lifetime [87]. Although directly applicable to animal research, the refinement concept also extends to improving the physiological relevance of human cell-based models.
Current scientific consensus recognizes that the 3Rs principles apply to all animals, reflecting expanded understanding of sentience beyond traditional categories [87]. The 3Rs are now viewed not merely as a technical checklist but as a dynamic framework that promotes continuous improvement of both scientific outcomes and animal welfare [87].
FDA Modernization Act 2.0 and 3.0: Regulatory Implementation
The regulatory landscape has rapidly evolved to incorporate these ethical principles into practice. The FDA Modernization Act 2.0 provided the foundational authority for using non-animal alternatives in drug development [88]. In April 2025, the FDA announced a significant policy shift to actively phase out animal testing requirements for monoclonal antibodies and other drugs, replacing them with human-relevant methods including AI-based computational models, cell lines, and organoid toxicity testing [89].
This initiative represents a paradigm shift in drug evaluation that promises to accelerate therapeutic development while reducing animal experimentation [89]. The FDA's approach includes:
- Encouraging developers to leverage computer modeling and artificial intelligence to predict drug behavior and side effects
- Promoting use of lab-grown human organoids and organ-on-a-chip systems that mimic human organs
- Providing regulatory incentives for companies that submit strong safety data from non-animal tests
- Utilizing preexisting, real-world safety data from other countries with comparable regulatory standards [89]
The recently reintroduced FDA Modernization Act 3.0 aims to further strengthen these provisions, directing the FDA to fully implement measures to reduce unnecessary animal testing [88]. The agency aims to make animal studies the exception rather than the norm within the next three to five years [88].
Application Notes: Organoids in Biomedical Research
Advantages of Organoid Models in Drug Development
Organoid technologies are transforming pharmaceutical research by providing models that more accurately reflect human physiology, genetic variability, and disease mechanisms [20]. The comparative advantages of these systems are substantial:
Table 1: Comparison of Research Models in Drug Development
| Model System | Physiological Relevance | Human Specificity | Genetic Diversity | Throughput Potential | 3Rs Alignment |
|---|---|---|---|---|---|
| Traditional 2D Cultures | Low | Limited | Limited | High | Partial Replacement |
| Animal Models | Moderate (species-dependent) | No | Limited | Low | Reduction/Refinement |
| Organoid Systems | High | Yes | High (patient-derived) | Medium-High | Replacement/Reduction |
| Organ-on-Chip | Very High | Yes | Customizable | Medium | Replacement |
The convergence of stem cell and organoid technologies has catalyzed emergence of next-generation preclinical platforms, particularly valuable for precision medicine applications [20]. Patient-derived organoids (PDOs) retain patient-specific genetic, epigenetic, and phenotypic features, enabling individualized approaches to treatment selection and drug development [20].
Implementation Challenges and Solutions
Despite their significant advantages, organoid technologies present several implementation challenges that researchers must address:
Table 2: Challenges and Solutions in Organoid Implementation
| Challenge | Impact on Research | Emerging Solutions |
|---|---|---|
| Protocol Standardization | Inter-laboratory variability | Development of validated, detailed protocols; reference materials |
| Batch-to-Batch Variability | Reproducibility issues | Quality control measures; automation; standardized reagents |
| Limited Maturation | Reduced physiological relevance | Extended culture periods; improved differentiation protocols; vascularization |
| Scalability | Throughput limitations | Automated production systems; high-throughput screening formats |
| Regulatory Acceptance | Translation to clinical applications | Evidence generation; standardization; engagement with regulatory agencies |
Industry leaders emphasize that addressing these challenges requires coordinated efforts toward standardization and industrialization [90]. Recent conferences have highlighted the importance of increasing data sharing, harmonizing operational protocols, and strengthening dialogue with regulators to facilitate use of organoid data in drug development and approval processes [90].
Experimental Protocols
Generation of Human Cerebral Organoids from Pluripotent Stem Cells
This protocol details the generation of three-dimensional cerebral brain organoids from human induced pluripotent stem cells (hiPSCs), adapted from established methods [65]. These organoids self-organize into distinct brain regions, including a structured cerebral cortex with progenitor populations that give rise to cortical excitatory neurons, recapitulating key aspects of human cortical development.
Materials and Reagents
Table 3: Essential Reagents for Cerebral Organoid Generation
| Reagent/Category | Specific Examples | Function | Notes |
|---|---|---|---|
| Stem Cell Media | mTeSR Plus | Maintenance of pluripotency | Include ROCK inhibitor (Y-27632) for thawing and passaging |
| Induction Media | M1 Medium | Neural induction | Contains LDN193189, SB431542, IWR-1e, Trans-ISRIB, Chroman 1, Emricasan |
| Differentiation Media | F2 Medium, H3 Medium | Neural differentiation and maturation | F2 contains SB431542, CHIR99021; H3 is 1:1 DMEM/F12:Neurobasal |
| Extracellular Matrix | Matrigel | 3D structural support | Cold-handling essential; keep on ice |
| Dissociation Agent | TrypLE | Cell dissociation | Gentler than trypsin |
| Specialized Equipment | Aggrewell plates | Embryoid body formation | Enables uniform-sized EBs |
Step-by-Step Protocol
hiPSC Culture and Maintenance (Days -5 to 0)
- Coating: One day before thawing hiPSCs, coat 6-well plates with Matrigel (100 μL Matrigel in 6 mL cold DMEM/F12; 1 mL/well). Incubate at 37°C for at least 2 hours or overnight.
- Thawing: Rapidly thaw hiPSC vial in 37°C water bath. Transfer cells to 15 mL tube with 9 mL DMEM/F12. Centrifuge at 300 × g for 4 minutes.
- Seeding: Aspirate Matrigel from coated well and replace with 1 mL mTeSR Plus medium supplemented with ROCK inhibitor (Y-27632). Resuspend cell pellet in 1 mL mTeSR Plus with ROCK inhibitor and transfer to prepared well.
- Maintenance: Change medium daily with fresh mTeSR Plus (without ROCK inhibitor) until cells reach 60-80% confluence (typically 3-5 days). Passage using TrypLE when needed.
Embryoid Body (EB) Formation (Day 0)
- Preparation: Add 500 μL Anti-Adherence solution to one well of an Aggrewell plate. Prepare balanced centrifuge plate with DPBS.
- Dissociation: Dissociate hiPSCs with TrypLE (1 mL/well for 1-2 minutes at 37°C). Neutralize with DMEM/F12, collect cells, and centrifuge at 300 × g for 1 minute.
- Counting: Resuspend pellet in mTeSR Plus and count cells using Trypan blue exclusion.
- EB Formation: Aspirate Anti-Adherence solution, wash with 1 mL mTeSR Plus, and add 1 mL mTeSR Plus with ROCK inhibitor. Transfer 3 million cells to the Aggrewell well. Mix by pipetting and centrifuge at 100 × g for 3 minutes at room temperature to aggregate cells.
- Incubation: Place Aggrewell plate in incubator overnight.
Neural Induction (Days 1-5)
- Day 1: Prepare 6-well plate with 2 mL M1 medium per well. Using a cut p1000 tip, transfer EBs from Aggrewell to the 6-well plate. Place plate on shaker.
- Day 2: Tilt plate to let EBs settle. Aspirate half the media and add 2 mL fresh M1 medium.
- Days 3-5: Repeat Day 2 process with half media changes daily.
Cortical Organoid Culture and Maturation (Days 6-14+)
- Day 6: Aspirate most media and replace with 2 mL F2 medium.
- Day 7 (Embedding in Matrigel): Transfer EBs to EP tube, let settle, and wash with F2 medium. Remove supernatant to ~60 μL residual volume. Add 100 μL Matrigel, mix by pipetting, and spread mixture in center of low-attachment 6-well plate. Incubate 50 minutes to solidify. Gently add 3 mL F2 medium.
- Days 9-13: Perform half media changes with F2 medium.
- Day 14 (Matrigel Release): Use 10 mL pipette to mechanically dissociate Matrigel, releasing organoids. Transfer to new plate with H3 medium.
- Long-term Culture: Continue with half media changes twice weekly for up to several months, monitoring neural differentiation and maturation.
Figure 1: Cerebral Organoid Generation Workflow. The diagram illustrates the key stages in generating 3D cerebral organoids from human induced pluripotent stem cells, culminating in structured tissue containing essential neural cell types.
Characterization and Quality Control
- Immunohistochemistry: Confirm presence of key neural markers: PAX6+ radial glial cells (inner layer), TBR2/EOMES+ intermediate progenitors (middle layer), and CTIP2/BCL11B+ immature neurons (outer layer) [65].
- Functional Assessment: Evaluate electrophysiological activity using microelectrode array (MEA) technology when applicable [90].
- Morphological Analysis: Monitor self-organization into structured cerebral cortex regions over 8-12 weeks of maturation.
Alternative Protocol: 2D-to-3D Cerebral Organoid Generation
An alternative approach generates human cerebral organoids (hCOs) directly from 2D pluripotent stem cell colonies, bypassing EB aggregation [91]. This method offers advantages in efficiency and reproducibility:
- Neural Fate Induction: Subject 2D PSC cultures to neural induction conditions without cell dissociation.
- Direct 3D Transition: Transfer emerging neural structures to 3D culture conditions.
- Characterization: Evaluate cellular diversity and regional specification using single-cell RNA sequencing and immunohistochemistry.
This protocol demonstrates highly efficient neuroepithelial induction and may reduce variability associated with EB formation steps [91].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagent Solutions for Organoid Generation
| Reagent Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| Pluripotent Stem Cell Media | mTeSR Plus, StemFlex | Maintain pluripotency | Essential for high-quality hiPSC maintenance before differentiation |
| Neural Induction Supplements | LDN193189, SB431542, IWR-1e | SMAD inhibition; neural specification | Critical for efficient neural induction; concentration-dependent effects |
| Pro-Maturation Factors | CHIR99021, BDNF, GDNF | Enhance maturation; support neuronal survival | Particularly important for long-term cultures (>30 days) |
| Extracellular Matrices | Matrigel, Cultrex BME | 3D structural support | Lot-to-lot variability requires testing; cold-handling essential |
| Metabolic Supplements | B-27 without vitamin A, N-2 | Support neural metabolism | Vitamin A can promote non-neural differentiation in early stages |
| Cryopreservation Solutions | CryoStor CS10 | Cell/organoid banking | Maintain viability after freeze-thaw cycles |
The convergence of ethical frameworks like the 3Rs principles with regulatory advancements exemplified by the FDA Modernization Act 2.0 is accelerating the adoption of human-relevant research models. Organoid technology represents a transformative approach that simultaneously advances scientific capability and ethical practice [20]. The detailed protocols provided herein enable researchers to implement cerebral organoid models that recapitulate key aspects of human brain development while aligning with modern regulatory and ethical standards.
As the field progresses, ongoing efforts to standardize protocols, enhance maturation, and gain regulatory acceptance will further solidify the role of organoids in drug development and precision medicine. These advances promise to bridge the gap between preclinical models and human clinical responses, ultimately accelerating the development of safer, more effective therapeutics.
Conclusion
Organoid technology, rooted in the remarkable capabilities of pluripotent stem cells, represents a paradigm shift in biomedical research. While foundational principles are established, the field is rapidly advancing through interdisciplinary innovations in bioengineering, automation, and computational biology. Successfully addressing persistent challenges in standardization, vascularization, and functional maturation is the critical next step. The ongoing development of organoid biobanks and integrated atlases promises to enhance reproducibility and clinical predictive power. As these models become more sophisticated and accessible, they are poised to fundamentally accelerate drug discovery, advance personalized medicine, and reduce reliance on animal testing, ultimately bridging the long-standing gap between preclinical models and human patients.
References
- 1. Proteomic and functional comparison between human ... [https://elifesciences.org/reviewed-preprints/92025v1/pdf]
- 2. Totipotent vs Pluripotent Stem Cells (2025) [https://www.dvcstem.com/post/totipotent-vs-pluripotent]
- 3. Induced pluripotent stem cells (iPSCs) [https://www.nature.com/articles/s41392-024-01809-0]
- 4. Proteomic and functional comparison between human ... [https://pubmed.ncbi.nlm.nih.gov/39540879/]
- 5. Organoid factory: The recent role of the human induced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9869172/]
- 6. Human organoids in basic research and clinical applications [https://www.nature.com/articles/s41392-022-01024-9]
- 7. iPSC-derived and Patient-Derived Organoids: Applications ... [https://www.sciencedirect.com/science/article/pii/S2666027X24000501]
- 8. Pluripotent Stem Cells: Recent Advances and Emerging ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12025069/]
- 9. Clinical translation of human iPSC technologies: advances ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1627149/full]
- 10. of Generation - self neuromusculoskeletal tri-tissue... organized [https://pubmed.ncbi.nlm.nih.gov/39657678/]
- 11. of Generation - self ... | Axion Biosystems organized [https://www.axionbiosystems.com/resources/publication/generation-self-organized-neuromusculoskeletal-tri-tissue-organoids-human]
- 12. -Assembling Human Heart Generating Derived from... Self Organoids [https://app.jove.com/t/63097/generating-self-assembling-human-heart-organoids-derived-from]
- 13. Growing patterned human primitive heart organoids via pluripotent ... [https://communities.springernature.com/posts/growing-patterned-human-primitive-heart-organoids-via-pluripotent-stem-cell-self-organization]
- 14. Generation of Cerebral Organoids from Human Pluripotent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4160653/]
- 15. An integrated transcriptomic cell atlas of human neural ... [https://www.nature.com/articles/s41586-024-08172-8]
- 16. Intestinal Organoids: New frontiers in the study of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5250540/]
- 17. Intestinal Organoids [https://www.stemcell.com/discover-organoids/intestinal]
- 18. Organoids: The current status and biomedical applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC10192887/]
- 19. Mouse Intestinal Organoid Culture Protocols | STEMCELL Technologies [https://www.stemcell.com/intestinal-epithelial-organoid-culture-with-intesticult-organoid-growth-medium-mouse-lp.html]
- 20. Stem Cells and Organoids: A Paradigm Shift in Preclinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299584/]
- 21. Deriving Human Intestinal Organoids with Functional Tissue-Resident Macrophages All From Pluripotent Stem Cells - PubMed [https://pubmed.ncbi.nlm.nih.gov/39701210/]
- 22. Human pluripotent stem cell-derived intestinal organoids ... [https://www.sciencedirect.com/science/article/pii/S0171933525000147]
- 23. Quantitative profiling of human brain organoid cell diversity ... [https://www.sciencedirect.com/science/article/pii/S2211124725009398]
- 24. Quantitative profiling of human brain organoid cell diversity ... [https://pubmed.ncbi.nlm.nih.gov/40864552/]
- 25. Biomedical applications of organoids in genetic diseases - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11987506/]
- 26. An iPSC-based in vitro model recapitulates human thymic ... [https://www.nature.com/articles/s41467-025-62523-1]
- 27. The Use of Stem Cell-Derived Organoids in Disease Modeling [https://pmc.ncbi.nlm.nih.gov/articles/PMC8303386/]
- 28. Greater genetic diversity is needed in human pluripotent ... [https://www.nature.com/articles/s41467-022-34940-z]
- 29. Organoid Research [https://www.stemcell.com/technical-resources/area-of-interest/organoid-research.html]
- 30. A review of protocols for brain organoids and applications for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9803834/]
- 31. Bioengineering Approaches for the Advanced Organoid Research - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8682947/]
- 32. Generation of human brain region–specific organoids ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6241211/]
- 33. Morphodynamics of human early brain organoid ... - Nature [https://www.nature.com/articles/s41586-025-09151-3]
- 34. Brain organoids: a new paradigm for studying human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12620469/]
- 35. Liver organoids: Current advances and future applications for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11925438/]
- 36. Kidney Organoids: Current Advances and Applications [https://www.mdpi.com/2075-1729/15/11/1680]
- 37. Generation of human iPSC-derived pancreatic organoids ... [https://pubmed.ncbi.nlm.nih.gov/40756323/]
- 38. Organoid protocols [https://www.nature.com/collections/ghgbhacefc]
- 39. Organoids from pluripotent stem cells and human tissues [https://www.sciencedirect.com/science/article/pii/S153458072500005X]
- 40. Tumor organoid-immune co-culture models - Nature [https://www.nature.com/articles/s41420-025-02407-x]
- 41. Organoids: new frontiers in tumor immune ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1422031/full]
- 42. : Organoid and Applications Vascularization Strategies [https://pubmed.ncbi.nlm.nih.gov/40285576/]
- 43. Advancements in Stem Cell Research: Creating Vascularized ... [https://med.stanford.edu/cvi/mission/news_center/articles_announcements/2025/advancements-in-stem-cell-research-creating-vascularized-organoids.html]
- 44. Tumor organoid-immune co-culture models: exploring a ... [https://pubmed.ncbi.nlm.nih.gov/40268893/]
- 45. The Technological Advances and Prospects of Vascularized Brain... [https://www.sciopen.com/article/10.12290/xhyxzz.2025-0056]
- 46. Modeling neurodegenerative diseases with brain organoids [https://www.frontiersin.org/articles/10.3389/fcell.2025.1663286/full]
- 47. The application of organoids in treatment decision-making ... [https://molecular-cancer.biomedcentral.com/articles/10.1186/s12943-025-02429-0]
- 48. Organoid Culture [https://bio-protocol.org/exchange/minidetail?id=10319496&type=30]
- 49. From gut to liver: organoids as platforms for next-generation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11948905/]
- 50. Organoids and Spheroids Market Size, Share & Growth ... [https://www.metatechinsights.com/industry-insights/organoids-and-spheroids-market-3068]
- 51. Breakthroughs and challenges of organoid models for ... [https://www.nature.com/articles/s41420-025-02505-w]
- 52. Brain organoid protocols and limitations [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2024.1351734/full]
- 53. Balancing serendipity and reproducibility: Pluripotent stem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8190574/]
- 54. 2025 Trends: Organoids [https://www.genengnews.com/topics/genome-editing/2025-trends-organoids/]
- 55. Cell-Type Specific Signalling Networks in Heterocellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7060080/]
- 56. Iteration of Tumor Organoids in Drug Development [https://pubmed.ncbi.nlm.nih.gov/41155654/]
- 57. Advancements, Strategies, and Challenges in Organoid ... [https://www.sciencedirect.com/science/article/pii/S2095809925006083]
- 58. Establishment and Validation of a Model for Fetal Neural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10940837/]
- 59. Navigating Brain Organoid Maturation: From Benchmarking ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12383812/]
- 60. Three-dimensional midbrain organoids: a next-generation tool ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04660-4]
- 61. Development of a quantitative prediction algorithm for ... [https://www.nature.com/articles/s41467-021-24746-w]
- 62. Innovations in Organoid Engineering: Construction ... [http://www.xiahepublishing.com/2835-6357/FIM-2025-00023]
- 63. Generation of Individualized, Standardized, and Electrically ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12346388/]
- 64. In vitro modelling of alveolar repair at the air-liquid ... [https://www.nature.com/articles/s41598-020-62226-1]
- 65. Generation of Three Dimensional Cerebral Brain ... [https://www.protocols.io/view/generation-of-three-dimensional-cerebral-brain-org-dykk7uuw.html]
- 66. Biomedical applications of organoids derived from the ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1599384/full]
- 67. Scaling 3D Biology: Automation , Imaging, and the Future [https://www.news-medical.net/news/20250612/Scaling-3D-biology-Automation-imaging-and-the-future-of-high-throughput-research.aspx]
- 68. Generation of Human Induced Pluripotent Stem Cell ... [https://pubmed.ncbi.nlm.nih.gov/41182959/?utm_source=SimplePie&utm_medium=rss&utm_campaign=pubmed-2&utm_content=1VUFS2KU_hFnBUajs3hlb00hHBOGRVXZp7ZhDB0wD6N0LBD9Nk&fc=20250423082013&ff=20251103204950&v=2.18.0.post22+67771e2]
- 69. CellXpress. ai Cell Culture System | Molecular Devices Automated [https://www.moleculardevices.com/products/3d-biology/cellxpress-ai-automated-cell-culture-system]
- 70. Growing Interest in Organoids : AI & Automated Solutions... [https://emag.medicalexpo.com/growing-interest-in-organoids-ai-automated-solutions-by-molecular-devices/]
- 71. Roche | Mapping the future of organoids [https://www.roche.com/stories/mapping-the-future-of-organoids]
- 72. Jawbone-like organoids generated from human pluripotent ... [https://www.nature.com/articles/s41551-025-01419-3]
- 73. Market | Global Market Analysis Report - 2035 Organoids [https://www.futuremarketinsights.com/reports/organoids-market]
- 74. and Spheroids Market Trends Organoids -2034 2025 [https://www.gminsights.com/industry-analysis/organoids-and-spheroids-market]
- 75. of vascularized brain Generation to study neurovascular... organoids [https://elifesciences.org/articles/76707]
- 76. 3D vs 2D Cell Culture: Which Model Suits Your Lab in 2025? [https://www.pharmanow.live/knowledge-hub/research/3d-vs-2d-cell-culture-comparison]
- 77. Human organoids: model systems for human biology and ... [https://www.nature.com/articles/s41580-020-0259-3]
- 78. Organoids in Biomedical Research: Opportunities and ... [https://www.wpiinc.com/blog/post/organoids-in-biomedical-research-opportunities-and-challenges?srsltid=AfmBOooVd0PcBm7WYOMbunAoBn2mqVappW9aCrrKClJ1gsE_ajh1GPJT]
- 79. Organoid Research Expands at Cincinnati Children's [https://scienceblog.cincinnatichildrens.org/organoid-research-expands-at-cincinnati-childrens/]
- 80. Organoids VS Organ-on-a-chip: A Comparative Review ... [https://elveflow.com/microfluidic-reviews/organoids-vs-organ-on-a-chip/]
- 81. The Organoid Cell Atlas - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7801253/]
- 82. An integrated transcriptomic cell atlas of human... | Nature Genetics [https://www.nature.com/articles/s41588-025-02182-6?error=cookies_not_supported&code=fee36656-5741-49fc-9385-45c59bab6b8a]
- 83. PharmaFormer predicts clinical drug responses through ... [https://www.nature.com/articles/s41698-025-01082-6]
- 84. Pluripotent stem cell-derived organoids - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC5358106/]
- 85. Study on drug sensitivity and clinical application of tumor ... [https://pubmed.ncbi.nlm.nih.gov/41113053/]
- 86. Longitudinal prediction of drug response in high-grade ... [https://www.sciencedirect.com/science/article/pii/S2090123225005983]
- 87. Toward a common interpretation of the 3Rs principles in ... [https://www.nature.com/articles/s41684-024-01476-2]
- 88. FDA Announces Plan to Phase Out Animal Testing ... [https://www.hklaw.com/en/insights/publications/2025/04/fda-announces-plan-to-phase-out-animal-testing-requirement]
- 89. FDA Announces Plan to Phase Out Animal Testing ... [https://www.fda.gov/news-events/press-announcements/fda-announces-plan-phase-out-animal-testing-requirement-monoclonal-antibodies-and-other-drugs]
- 90. ACROBiosystems and Partners Conclude OrganoAsia 2025 [https://www.koreaherald.com/article/10590846]
- 91. Protocol for generating human cerebral organoids from two ... [https://www.sciencedirect.com/science/article/pii/S266616672500084X]