The Complete Guide to Trypsin-EDTA Cell Dissociation: From Basic Principles to Advanced Applications
This comprehensive article provides researchers, scientists, and drug development professionals with an in-depth understanding of the Trypsin-EDTA cell dissociation protocol. It covers the foundational science behind enzymatic dissociation, detailed step-by-step methodologies for various applications, advanced troubleshooting and optimization strategies, and a critical comparative analysis with alternative dissociation techniques. The content synthesizes current best practices and recent research findings to enable robust, reproducible cell culture workflows essential for biomedical research and therapeutic development.
The Complete Guide to Trypsin-EDTA Cell Dissociation: From Basic Principles to Advanced Applications
Abstract
This comprehensive article provides researchers, scientists, and drug development professionals with an in-depth understanding of the Trypsin-EDTA cell dissociation protocol. It covers the foundational science behind enzymatic dissociation, detailed step-by-step methodologies for various applications, advanced troubleshooting and optimization strategies, and a critical comparative analysis with alternative dissociation techniques. The content synthesizes current best practices and recent research findings to enable robust, reproducible cell culture workflows essential for biomedical research and therapeutic development.
Understanding Trypsin-EDTA: The Science Behind Cell Dissociation
Cell dissociation is a fundamental process in modern cell culture, essential for subculturing adherent cells, conducting cell-based assays, and facilitating bioproduction. This critical step involves breaking down tissue or detaching adherent cells from their culture surface to create single-cell suspensions while maintaining cellular integrity and viability. The process balances efficient cell release with preservation of cell health and surface proteins, requiring careful selection of dissociation methods tailored to specific cell types and research objectives. Within this domain, enzymatic dissociation—particularly using trypsin-EDTA solutions—remains the gold standard for many applications, though non-enzymatic and mechanical approaches offer viable alternatives for specialized needs. This application note provides a comprehensive overview of cell dissociation principles, detailed protocols, and advanced applications to support researchers in optimizing their cell culture workflows.
Core Principles of Cell Dissociation
Fundamental Mechanisms
Cell dissociation operates through three primary mechanisms, each targeting different cellular adhesion structures. Enzymatic methods employ proteases like trypsin to digest cell-surface proteins and extracellular matrix components that mediate attachment [1]. Non-enzymatic chemical approaches use chelating agents such as EDTA or EGTA to sequester divalent cations (Ca2+, Mg2+) that are essential for cadherin-mediated cell-cell adhesions and integrin-mediated cell-matrix interactions [2] [3]. Mechanical techniques physically disrupt attachments through scraping, pipetting, or tissue grinding, though these risk greater cellular damage [2].
Trypsin-EDTA: The Gold Standard
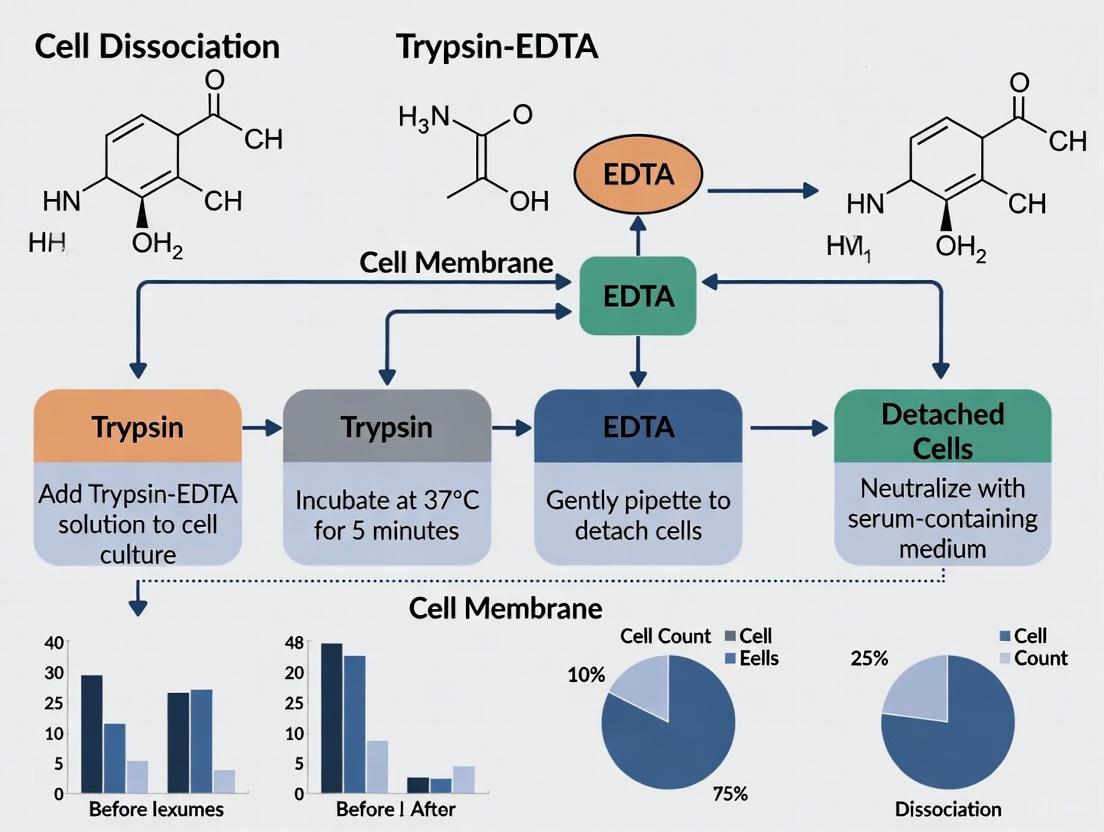
Trypsin, a serine protease, specifically cleaves peptide bonds at the C-terminal side of lysine and arginine residues, effectively digesting adhesion proteins [1]. EDTA (ethylenediaminetetraacetic acid) enhances this process by chelating calcium and magnesium ions, weakening calcium-dependent cell junctions and preventing trypsin autolysis [1] [3]. This combination synergistically improves dissociation efficiency across diverse cell types.
The activity of trypsin-EDTA is highly dependent on pH and temperature, with optimal performance at pH 7-7.6 and 37°C [1]. Solution color, often provided by pH indicators like phenol red, offers visual confirmation of appropriate pH conditions, with pink indicating optimal activity and orange suggesting reduced efficacy in acidic environments [1].
Comprehensive Dissociation Methods
Method Comparison and Selection
Selecting the appropriate dissociation method requires careful consideration of cell type, application requirements, and downstream processing needs. The table below summarizes the primary dissociation approaches and their characteristics:
Table 1: Comparison of Cell Dissociation Methods
| Method | Mechanism | Applications | Advantages | Limitations |
|---|---|---|---|---|
| Trypsin-EDTA [1] [4] | Proteolytic enzyme digestion + cation chelation | Strongly adherent cell lines; general subculturing | Highly effective for most adherent cells; well-established protocol | Potential damage to cell surface proteins; requires inhibition |
| TrypLE Express [4] [5] | Recombinant microbial protease | Strongly adherent cells; animal origin-free requirements | Direct trypsin substitute; reduced regulatory concerns; consistent performance | May require optimization for sensitive cells |
| Collagenase [4] [5] | Collagen-specific enzyme digestion | Primary tissues; fibroblast cultures; dense matrices | Effective for fibrous tissues; preserves cell surface markers | Longer incubation times; tissue-specific optimization needed |
| Dispase [4] [5] | Neutral protease digestion | Epidermal cell sheets; stem cell cultures | Gentle dissociation; maintains cell-cell contacts for cluster formation | Limited effectiveness for strongly adherent monolayers |
| Non-enzymatic Buffer [4] [5] | Cation chelation only | Lightly adherent cells; surface protein-sensitive applications | Preserves surface epitopes; no enzyme inhibition required | Ineffective for strongly adherent cells or tight junctions |
| Mechanical [2] | Physical disruption | Loosely associated tissues; enzyme-sensitive cells | Rapid; no chemical exposure | Low viability; inconsistent yield; not suitable for single-cell suspensions |
Impact of Dissociation Conditions on Cell Stress and Composition
Recent systematic studies have revealed significant biases introduced by different dissociation conditions. Research comparing warm (37°C) versus cold (ice) dissociation protocols demonstrated that warm dissociation induces substantial stress response genes including immediate-early genes (Fos, Jun, Junb) and heat shock proteins (Hspa1a, Hspa1b) [6]. This stress response varies considerably across cell types, with immune and endothelial cells showing particular sensitivity.
Furthermore, cell type representation in resulting suspensions differs markedly between protocols. Cold dissociation better preserves sensitive populations like podocytes, mesangial cells, and endothelial cells, while warm dissociation may more efficiently release certain epithelial populations such as the ascending loop of Henle and proximal tubule cells [6]. These findings highlight the critical importance of matching dissociation conditions to both target cell types and experimental objectives.
Table 2: Quantitative Comparison of Warm vs. Cold Dissociation Effects on Mouse Kidney Cell Populations [6]
| Cell Population | Cold Dissociation (% total) | Warm Dissociation (% total) | Fold Change | Stress Response Level |
|---|---|---|---|---|
| Podocytes | 2.78% | 0.03% | 92.7× decrease | High |
| Mesangial Cells | 1.42% | 0.21% | 6.8× decrease | High |
| Endothelial Cells | 4.56% | 2.11% | 2.2× decrease | High |
| aLOH | 2.52% | 4.99% | 2.0× increase | Low |
| Proximal Tubule | 63.34% | 71.36% | 1.1× increase | Low |
| Immune Cells | 4.89% | 3.12% | 1.6× decrease | Medium-High |
Detailed Experimental Protocols
Standard Trypsin-EDTA Cell Dissociation Protocol
The following protocol is adapted from established methodologies for dissociating adherent cell cultures [1] [4]:
Reagents and Equipment:
- Trypsin-EDTA solution (0.05%-0.25%, based on cell line)
- Balanced salt solution without Ca2+ and Mg2+ (e.g., DPBS)
- Complete growth medium (with serum)
- Centrifuge tubes
- Pre-warmed water bath (37°C)
- Inverted microscope
Procedure:
- Preparation: Pre-warm trypsin-EDTA solution, balanced salt solution, and complete growth medium to 37°C. Work aseptically in a biological safety cabinet.
- Media Removal: Aspirate and discard spent culture media from the flask.
- Rinsing: Gently rinse the cell layer with 2-3 mL of balanced salt solution per 25 cm² of growth surface. Ensure the solution flows across the entire cell sheet. Aspirate and discard the rinse solution.
- Trypsin Application: Add pre-warmed trypsin-EDTA solution (0.5 mL per 10 cm²) to the side of the flask opposite the cell layer. Gently rotate the flask to ensure complete coverage.
- Incubation: Incubate the flask at 37°C for 2-5 minutes. The optimal incubation time varies by cell line. Monitor detachment visually using an inverted microscope. Cells will appear rounded and refractile when detached.
- Neutralization: When >90% of cells are detached, add 2 volumes of complete growth medium containing serum to neutralize the trypsin. Gently pipette the solution across the cell layer to complete detachment.
- Cell Collection: Transfer the cell suspension to a centrifuge tube. Centrifuge at 100-300 × g for 5-10 minutes.
- Resuspension: Discard the supernatant and resuspend the cell pellet in fresh, pre-warmed complete growth medium.
- Counting and Assessment: Determine cell density and viability using trypan blue exclusion and a hemocytometer or automated cell counter. Cell viability should exceed 90% for healthy cultures.
Troubleshooting:
- Slow Detachment: Increase trypsin concentration within 0.05%-0.5% range or extend incubation time in 1-minute increments [3].
- Poor Viability: Reduce trypsin incubation time; use lower trypsin concentration; ensure prompt and complete neutralization [3].
- Cell Clumping: Add DNase (1-10 µg/mL) to digest released genomic DNA; filter through cell strainer [3].
Tissue Dissociation for Primary Cells
Dissociating primary tissues requires more specialized approaches. A combined enzymatic protocol for primary tissue disaggregation follows [4]:
- Tissue Preparation: Mince tissue into 3-4 mm pieces using sterile scalpels or scissors.
- Washing: Wash tissue fragments multiple times with HBSS (with Ca2+ and Mg2+ for collagenase; without for trypsin).
- Enzymatic Digestion:
- Option A (Trypsin): Incubate with 0.25% trypsin at 4°C for 6-18 hours for enzyme penetration, followed by 37°C for 20-30 minutes for activity [4].
- Option B (Collagenase): Incubate with collagenase (50-200 U/mL) at 37°C for 4-18 hours with agitation [4].
- Option C (Combined): Use dispase (0.6-2.4 U/mL) with collagenase (60-100 U/mL) for 1+ hours at 37°C [4].
- Cell Dispersion: Pass the digested tissue through sterile mesh (100-200 µm) to separate dispersed cells from undigested fragments.
- Washing: Pellet cells by centrifugation and resuspend in appropriate culture medium.
Method Selection Workflow
The following diagram illustrates the decision-making process for selecting appropriate dissociation methods:
Advanced Applications and Research Context
Single-Cell Technologies and Dissociation Artifacts
In single-cell RNA sequencing (scRNA-seq) workflows, dissociation-induced transcriptional changes represent a significant challenge. Systematic assessments reveal that warm dissociation (37°C) activates stress response pathways including immediate-early genes and heat shock proteins, potentially confounding biological interpretations [6]. Different cell populations exhibit varying sensitivity, with immune cells, endothelial cells, and podocytes showing particularly strong stress responses to warm dissociation conditions.
Single-nucleus RNA sequencing (snRNA-seq) has emerged as an alternative approach that bypasses many dissociation-related artifacts, as nuclei can be isolated from frozen tissue under conditions that minimize transcriptional responses [6]. However, snRNA-seq introduces its own biases, including underrepresentation of T cells, B cells, and NK lymphocytes [6].
Quantitative Analysis of Cluster Dissociation
Novel methods for quantifying cell cluster dissociation have been developed to evaluate therapeutic interventions. Light transmission aggregometry (LTA), traditionally used for platelet studies, has been adapted to characterize tumor cell cluster dissociation in response to fibrinolytic agents like alteplase and tenecteplase [7]. This approach provides quantitative metrics including:
- Lag time to initiation of dissociation
- Time to 25% dissociation
- Dissociation trend over time
This methodology enables precise evaluation of compounds targeting circulating tumor cell (CTC) clusters, which have significantly higher metastatic potential than single circulating tumor cells [7].
Market Trends and Bioprocessing Applications
The trypsin-EDTA solution market reflects the growing importance of cell dissociation in bioprocessing, with an estimated market size of USD 950 million in 2025 projected to grow at a CAGR of 7.5% to reach USD 1,750 million by 2033 [8]. Key drivers include:
- Escalating demand for biopharmaceuticals (insulins, vaccines)
- Advancements in regenerative medicine and cell-based therapies
- Expansion of biomanufacturing capabilities in emerging economies
- Increasing adoption of automation in cell culture
The cell culture application segment dominates the trypsin-EDTA market, underscoring its fundamental role across life science research and biopharmaceutical production [8].
Table 3: Key Research Reagent Solutions for Cell Dissociation
| Reagent | Composition | Primary Function | Applications |
|---|---|---|---|
| Trypsin-EDTA [1] [4] | Trypsin (0.05%-0.25%) + EDTA (0.02%-0.53 mM) | Proteolytic digestion of adhesion proteins + cation chelation | General cell culture; strongly adherent cell lines |
| TrypLE Express [4] | Recombinant fungal trypsin-like protease | Animal origin-free proteolytic digestion | Biotherapeutic production; regulated applications |
| Collagenase [4] | Collagen-specific enzymes (50-200 U/mL) | Digestion of collagenous extracellular matrix | Primary tissue dissociation; fibroblast cultures |
| Dispase [4] | Neutral bacterial protease (0.6-2.4 U/mL) | Gentle proteolytic digestion without disrupting cell-cell contacts | Stem cell cultures; epithelial sheet isolation |
| Cell Dissociation Buffer [4] [5] | EDTA-based formulation without enzymes | Cation chelation to disrupt calcium-dependent junctions | Surface protein-sensitive applications; flow cytometry |
| Soybean Trypsin Inhibitor [1] [3] | Proteinase inhibitor | Rapid trypsin inactivation | Serum-free cultures; sensitive cell types |
| DNase I [3] | Deoxyribonuclease I | Digestion of genomic DNA released from damaged cells | Reduction of cell clumping after dissociation |
Cell dissociation remains a critical step in modern cell culture, with implications ranging from basic research to biotherapeutic production. The trypsin-EDTA protocol continues to serve as the cornerstone methodology for most adherent cell cultures, though understanding its limitations and appropriate alternatives enables researchers to optimize conditions for specific applications. As single-cell technologies advance and bioprocessing demands grow, continued refinement of dissociation protocols will remain essential for generating reliable, reproducible data and products. By matching dissociation methods to cellular characteristics and experimental goals, researchers can maximize cell viability, preserve physiological states, and ensure the success of downstream applications.
The reproducibility and success of cell-based research and biomanufacturing hinge on the precise formulation and application of dissociation reagents and buffer systems. Within the context of trypsin-EDTA cell dissociation protocols, a thorough understanding of the key components—their concentrations, mechanisms, and interactions with supportive buffers—is fundamental. This document provides detailed application notes and protocols, framing them within the critical demands of modern drug development, including the production of biologics, vaccines, and cell-based therapies [8]. The shift toward personalized medicine and the growth of the biopharmaceutical sector, which is driving the Trypsin-EDTA solution market to a projected $1,750 million by 2033, further underscore the need for standardized, reliable, and well-understood dissociation techniques [9] [8].
Key Components: Composition and Concentration
The efficacy of cell dissociation is directly governed by the concentration and formulation of its active components. The selection of an appropriate trypsin-EDTA concentration is critical for balancing efficient cell detachment with the preservation of cell viability and surface integrity.
Trypsin-EDTA Formulations
Trypsin-EDTA solutions are available in standardized concentrations tailored to different cell types and applications. The table below summarizes the common formulations and their primary uses.
Table 1: Common Trypsin-EDTA Formulations and Applications
| Trypsin Concentration | EDTA Concentration | Primary Applications and Target Cell Types |
|---|---|---|
| 0.25% [8] | 1 mM (common) [10] | General purpose for strongly adherent mammalian cell lines; the most widely used concentration [4] [8]. |
| 0.05% [8] | 0.2 - 0.5 mM (common) | Lightly adherent or sensitive cell lines; applications requiring gentler proteolytic activity [4]. |
| TrypLE Express | N/A (non-enzymatic) | A recombinant alternative to trypsin; ideal for applications requiring animal origin-free reagents and minimal cell surface protein damage [4]. |
The optimal conditions (e.g., volume, incubation time) for any cell line should be determined empirically, with the goal of achieving ≥90% detachment while maintaining cell viability over 90% [4] [11].
Mechanism of Action
The dissociation process is a coordinated biochemical mechanism:
- Trypsin: A proteolytic enzyme that cleaves peptide bonds, specifically targeting lysine and arginine residues in proteins that mediate cell-to-substrate and cell-to-cell adhesion, such as fibronectin and laminin [10].
- EDTA (Ethylenediaminetetraacetic acid): A chelating agent that binds divalent cations like calcium (Ca²⁺) and magnesium (Mg²⁺). These ions are essential cofactors for cell adhesion molecules like cadherins and integrins. By removing these ions, EDTA disrupts intercellular connections and enhances the efficacy of trypsin [10].
The following diagram illustrates this synergistic mechanism of action:
Diagram: Synergistic mechanism of trypsin and EDTA in cell dissociation.
Experimental Protocols
A standardized and carefully optimized protocol is vital for successful cell passaging or harvesting. Deviations can lead to poor viability, clumping, or loss of key cellular functions.
Standard Trypsin-EDTA Dissociation Protocol for Adherent Mammalian Cells
This protocol is adapted from established cell culture basics and is suitable for most adherent mammalian cell lines [11].
Materials:
- Pre-warmed complete growth medium
- Pre-warmed balanced salt solution (e.g., PBS or DPBS) without calcium and magnesium [11]
- Pre-warmed Trypsin-EDTA solution (e.g., 0.25%)
- Culture vessel with adherent cells
- Centrifuge tubes
- Pipettes and a cell counter
Method:
- Remove Media: Aspirate and discard the spent cell culture media from the culture vessel [11].
- Wash Cell Layer: Rinse the cell layer with a balanced salt solution without calcium and magnesium (e.g., ~2 mL per 10 cm²) to remove any traces of serum, calcium, and magnesium that inhibit trypsin. Gently rock the vessel and discard the wash solution [11].
- Add Trypsin-EDTA: Add pre-warmed Trypsin-EDTA solution sufficient to cover the cell layer (e.g., ~0.5 mL per 10 cm²). Gently rock the vessel to ensure complete coverage [11].
- Incubate: Incubate the culture vessel at room temperature or 37°C for approximately 2-5 minutes. The actual time varies by cell line and should be determined empirically [11].
- Monitor Detachment: Observe cells under a microscope. If less than 90% of cells are detached, tap the vessel gently and continue incubation, checking every 30 seconds. Cells will appear rounded and refractile when detached [11].
- Neutralize: When ≥90% of cells are detached, add 2 volumes of pre-warmed complete growth medium (containing serum which inhibits trypsin) to neutralize the enzyme. Disperse the medium by pipetting over the cell layer surface to ensure complete resuspension [11].
- Centrifuge and Resuspend: Transfer the cell suspension to a centrifuge tube and pellet cells at approximately 200 × g for 5-10 minutes. Discard the supernatant and resuspend the cell pellet in a minimal volume of fresh, pre-warmed complete growth medium [11].
- Count and Seed: Determine cell count and viability using an automated cell counter or hemocytometer. Dilute the cell suspension to the recommended seeding density and pipet into new culture vessels [11].
The workflow for the standard cell dissociation process is summarized below:
Diagram: Standard workflow for trypsin-EDTA cell dissociation.
Protocol for Primary Tissue Dissociation using Trypsin
Dissociating primary tissue requires a more rigorous approach to obtain a high yield of viable single cells.
Materials:
- Minced primary tissue (3-4 mm pieces)
- Balanced salt solution without calcium and magnesium
- 0.25% trypsin solution
- Sterile scalpel or scissors
- Sterile stainless-steel or nylon mesh (100-200 µm)
Method:
- Mince Tissue: After dissection, mince the tissue into 3-4 mm pieces with a sterile scalpel or scissors [4].
- Wash Tissue Pieces: Wash the tissue pieces by resuspending them in a balanced salt solution without calcium and magnesium. Allow the pieces to settle and remove the supernatant. Repeat 2-3 times [4].
- Add Trypsin and Incubate: Place the container on ice, remove any remaining supernatant, and add 0.25% trypsin in a balanced salt solution (1 mL per 100 mg of tissue). Incubate at 4°C for 6-18 hours to allow enzyme penetration [4].
- Decant and Warm: Decant and discard the trypsin from the tissue pieces. Incubate the tissue pieces with the residual trypsin at 37°C for 20-30 minutes to activate the enzyme [4].
- Disperse and Filter: Add warm, complete media to the tissue pieces and gently disperse by pipetting. Filter the cell suspension through a sterile mesh (100-200 µm) to remove any remaining tissue clumps [4].
- Count and Seed: Count the cells and seed them into an appropriate culture vessel [4].
The Scientist's Toolkit: Essential Reagents and Materials
A successful dissociation experiment requires a suite of reliable reagents and equipment. The following table details the essential components of the researcher's toolkit.
Table 2: Essential Research Reagent Solutions for Cell Dissociation
| Item | Function and Key Characteristics |
|---|---|
| Trypsin-EDTA Solution | Proteolytic enzyme and chelating agent combination for detaching adherent cells from culture surfaces. Available in various concentrations (e.g., 0.05%, 0.25%) for different cell types [8] [10]. |
| TrypLE Express | A recombinant, animal-origin-free enzyme that functions as a direct substitute for trypsin. Reduces the risk of contamination and is gentler on cell surface proteins [4]. |
| Phosphate Buffered Saline (PBS) | An isotonic buffer solution used for washing cells and diluting reagents. It maintains pH and osmolarity, is non-toxic to cells, and is used to rinse away inhibitory ions before trypsinization [12] [13] [11]. |
| Dulbecco's PBS (DPBS) | A modified formulation of PBS that may include calcium and magnesium. For trypsinization, a formulation without calcium and magnesium is required to prevent inhibition of the enzyme [13]. |
| Complete Growth Medium | Cell culture medium containing serum (e.g., FBS) or other trypsin inhibitors. Used to neutralize trypsin activity immediately after cell detachment to prevent over-digestion and damage [11]. |
| Cell Dissociation Buffer | A non-enzymatic, often EDTA-based solution for gently dissociating lightly adherent cells or when intact cell surface proteins are critical for downstream applications [4]. |
| Soybean Trypsin Inhibitor | A specific inhibitor used to neutralize trypsin in serum-free culture conditions where serum cannot be used for inhibition [4]. |
Supporting Buffer Systems: PBS and DPBS
Buffer systems are the unsung heroes of cell culture, providing a stable physiological environment for cells during manipulation outside the incubator.
Composition and Function of PBS
Phosphate Buffered Saline (PBS) is a fundamental buffer that closely mimics the pH, osmolarity, and ion concentrations of the human body, making it non-toxic and ideal for cell contact [12] [13]. Its primary functions include washing cells, transporting tissues, and diluting reagents.
Table 3: Standard 1X PBS Formulation (pH 7.4)
| Component | Amount (per Liter) | Final Concentration |
|---|---|---|
| Sodium Chloride (NaCl) | 8 g | 137 mM |
| Potassium Chloride (KCl) | 0.2 g | 2.7 mM |
| Sodium Phosphate Dibasic (Na₂HPO₄) | 1.44 g | 10 mM |
| Potassium Phosphate Monobasic (KH₂PO₄) | 0.24 g | 1.8 mM |
PBS is simple to prepare and has a good shelf life, though it can precipitate in the presence of zinc ions or if frozen [12]. For specialized applications, formulations such as RNase-Free PBS (for RNA-sensitive workflows) and Low Endotoxin PBS (for cell therapy and immune assays) are available [13].
Dulbecco's Phosphate Buffered Saline (DPBS)
Dulbecco's PBS is a modification of the standard PBS formulation, containing slightly different concentrations of phosphate salts. Critically, DPBS is often available in two variants:
- With Calcium and Magnesium: Used in cell culture media and applications where these ions are needed for adhesion or enzymatic processes.
- Without Calcium and Magnesium: Specifically designed for procedures like cell washing prior to trypsinization, as the presence of these divalent cations would inhibit trypsin activity [13].
This highlights the necessity of selecting the correct buffer formulation for each specific step of the cell culture protocol.
Within the framework of a broader thesis on cell dissociation techniques, the optimization of physicochemical parameters is paramount for successful trypsinization. Trypsin-EDTA cell dissociation is a critical step in cell culture, directly impacting cell viability, yield, and the reliability of subsequent experimental data in drug development. This application note details the core protocols and the essential role of three critical parameters—pH, temperature, and osmotic balance—in ensuring efficient and reproducible cell dissociation. Maintaining these parameters within optimal ranges is fundamental to preserving cell integrity and biological activity during and after the enzymatic process [14] [15].
The Critical Parameters in Practice
The efficacy of Trypsin-EDTA is highly dependent on its physicochemical environment. Deviations from optimal conditions can lead to reduced enzymatic activity, cellular damage, and ultimately, experimental failure.
The following table summarizes the optimal ranges and key considerations for each critical parameter based on established cell culture methodologies.
Table 1: Optimal Ranges for Critical Parameters in Trypsin-EDTA Cell Dissociation
| Parameter | Optimal Range | Impact of Deviation | Key Considerations |
|---|---|---|---|
| pH | 7.4 - 7.6 [14] (Product range: 7.0 - 7.6 [15]) | Low pH (Acidic): Significantly reduces trypsin activity, leading to incomplete dissociation [14]. | Phenol red is often used as a visual pH indicator; a pink color indicates the correct pH range, while yellow/orange indicates acidity [14]. |
| Temperature | 37 °C [14] | Low Temp: Slows dissociation kinetics.High Temp/Prolonged Exposure: Can damage cell surface proteins and reduce viability [14]. | Incubation times are cell line-specific and must be determined empirically; typically 2-10 minutes at 37°C [14]. |
| Osmotic Balance | 270 - 310 mOsm/Kg H₂O [15] | Imbalance: Can cause osmotic stress, leading to cell shrinkage or swelling, and reduced post-dissociation viability. | Maintained using isotonic buffer solutions like Dulbecco's Phosphate Buffered Saline (DPBS) without Ca²⁺ or Mg²⁺ as the trypsin diluent [14] [15]. |
The Scientist's Toolkit: Essential Research Reagent Solutions
A successful dissociation protocol relies on a set of key reagents, each serving a specific function.
Table 2: Essential Reagents for Trypsin-EDTA Cell Dissociation
| Reagent / Solution | Function / Purpose |
|---|---|
| Trypsin-EDTA (0.25%) | Proteolytic enzyme (trypsin) cleaves cell-surface and cell-matrix adhesion proteins. EDTA chelates calcium and magnesium ions, weakening cell-cell junctions [14] [15]. |
| Balanced Salt Solution (without Ca²⁺ & Mg²⁺) | Used to wash the cell monolayer pre-trypsinization, removing residual divalent cations and serum that would inhibit trypsin activity [14]. |
| Complete Growth Medium (with Serum) | Contains serum proteins (e.g., in FBS) that act as trypsin inhibitors. Added after dissociation to rapidly halt tryptic activity and prevent cell damage [14]. |
| Soybean Trypsin Inhibitor | Specific, serum-free alternative for neutralizing trypsin, crucial for sensitive applications or when using serum-free media post-dissociation [15]. |
Experimental Protocols for Parameter Optimization and dissociation
This section provides a detailed methodology for standard cell dissociation and a specific protocol for monitoring detachment in microcarrier cultures, a key area of advanced research.
Standard Protocol for Adherent Cell Dissociation in Flasks
The following workflow outlines the core steps for dissociating cells from a culture flask, highlighting points where parameter control is critical.
Title: Standard Trypsin-EDTA Cell Dissociation Workflow
Detailed Methodology [14] [15]:
- Preparation: Pre-warm the Trypsin-EDTA solution (e.g., 0.25%), a balanced salt solution without calcium and magnesium (e.g., DPBS), and the complete growth medium to 37°C in a water bath. This prevents temperature shock during the procedure.
- Cell Inspection: Visually inspect the cells under a microscope to ensure they are healthy and at the appropriate confluency (typically 80-90%) for passaging. Check for any signs of contamination.
- Wash Step: Aspirate and discard the spent culture medium from the flask. Gently add a sufficient volume of the pre-warmed, calcium/magnesium-free buffer to the side of the flask opposite the cell layer. Rock the flask for 1-2 minutes to rinse the cells, then aspirate and discard the wash solution. This step is critical for removing residual serum, which inhibits trypsin.
- Trypsin Application: Add a minimal sufficient volume of the pre-warmed Trypsin-EDTA solution to the side of the flask to cover the cell monolayer completely after gentle rocking. The solution's pH should be in the optimal 7.4-7.6 range, often indicated by a pink color from phenol red.
- Incubation: Place the flask in a 37°C incubator for 2-3 minutes. The incubation time is cell line-dependent and must be optimized to prevent under- or over-trypsinization.
- Monitoring: Periodically check the cells under an inverted microscope. Detached cells will appear rounded and refractile. If fewer than 90% of cells are detached after 3-5 minutes, tap the flask firmly and return it to the incubator for another minute. Avoid prolonged exposure (>10-15 minutes) to active trypsin.
- Neutralization: Once cells are detached, immediately add a volume of pre-warmed complete growth medium that is at least double the volume of trypsin used. The serum in the medium acts as a trypsin inhibitor. For serum-free cultures, use a specific inhibitor like Soybean Trypsin Inhibitor.
- Cell Collection: Gently pipette the cell suspension across the surface of the flask to ensure all cells are collected and to break up any small clumps. Avoid vigorous pipetting, which can lyse cells.
- Centrifugation and Reseeding: Transfer the cell suspension to a centrifuge tube and spin at 100-300 x g for 5-10 minutes. Aspirate the supernatant, resuspend the cell pellet in fresh pre-warmed complete medium, and count the cells using a hemocytometer or automated cell counter with trypan blue exclusion. The cell suspension is now ready for reseeding or downstream analysis.
Advanced Protocol: Parameter Optimization in Microcarrier Cultures
For scalable processes in bioreactors, such as culturing cells on microcarriers (MCs), parameter control becomes even more critical. Recent research has focused on optimizing these parameters for efficient in situ cell detachment.
Key Optimized Parameters for Microcarrier Detachment [16]:
- Agitation Speed: Maintaining an appropriate agitation speed is crucial. A study on MA 104 cells on Cytodex 1 MCs found that an agitation speed of
1.25 x Njs(where Njs is the minimum speed to keep MCs suspended) was optimal. This ensures homogeneity and efficient enzyme-cell contact without generating damaging shear forces. - Trypsin Volume and Activity: The study demonstrated the feasibility of reducing the final trypsin volume to
50 mL per gramof microcarrier while maintaining a trypsin activity above380 USP/mL. This ensures complete cell dissociation while improving cost-effectiveness for large-scale production. - Washing Steps: Implementing
two washing stepswith a balanced salt solution post-medium removal was found to be crucial for efficient subsequent cell detachment and growth. - Real-time Monitoring: The use of capacitance sensors has been established as an effective Process Analytical Technology (PAT) tool. It allows for real-time, non-invasive monitoring of viable cell density during both growth and detachment phases. This method can predict cell detachment based on signals recorded within
3 to 7 minutesof trypsinization, enabling rapid process decisions.
The precise control of pH, temperature, and osmotic balance is non-negotiable in Trypsin-EDTA-based cell dissociation protocols. These parameters directly govern enzymatic kinetics and cellular homeostasis, influencing everything from basic research outcomes to the scalability of industrial bioprocesses. The standardized protocols and optimization strategies detailed herein provide a robust framework for researchers and drug development professionals to achieve high cell viability, yield, and experimental reproducibility. Integrating advanced monitoring tools like capacitance sensing further enhances process control, paving the way for more efficient and reliable scale-up in microcarrier-based culture systems, a vital component in modern biologics and vaccine manufacturing.
The choice between adherent mammalian and insect cell systems is a fundamental decision in bioprocess development, with significant implications for recombinant protein production and viral vector manufacturing. Each system offers distinct advantages and challenges, influencing critical factors such as cost, scalability, and product quality [17] [18]. Mammalian cells, particularly HEK293 and its derivatives, are the dominant platform for producing complex therapeutic proteins that require human-like post-translational modifications. In contrast, insect cells, such as Sf9 and Sf21, powered by the Baculovirus Expression Vector System (BEVS), provide a powerful and often more cost-effective alternative for producing multiprotein complexes and viral vectors [17]. This application note details the specific considerations, protocols, and quantitative comparisons for both systems, framed within the context of cell dissociation and subculturing practices essential for maintaining healthy cultures.
System Characteristics and Applications
Key Features and Industrial Context
Table 1: Comparative Analysis of Adherent Mammalian and Insect Cell Systems
| Feature | Adherent Mammalian Cells (e.g., HEK293) | Insect Cells (Sf9, Sf21) |
|---|---|---|
| Primary Use | Production of complex therapeutic proteins, viral vectors (LV, AAV) [18] | Recombinant multiprotein complexes, viral antigens, BEVS-based production [17] |
| Growth Environment | 37°C, humidified, 5% CO2 [11] | 27°C, non-humidified, no CO2 control [11] |
| Typical Culture Media | DMEM, RPMI-1640 (pH ~7.4) [11] | Grace's Insect Medium (more acidic) [11] |
| Adherence Nature | Strongly adherent | Variable; can be very tight under serum-free conditions [11] |
| Glycosylation Profile | Human-like, complex | Simpler, non-human; requires engineered cell lines (e.g., SfSWT-1) for human-like patterns [17] |
| Industrial Scalability | Shifting towards suspension for large-scale; adherent (e.g., iCELLis, roller bottles) used for commercial products like Zolgensma, Luxturna [18] | Excellent for suspension culture in large volumes; Sf9 cells are highly tolerant of shear stress [17] |
The manufacturing landscape illustrates that both systems have proven commercially viable. Adherent mammalian platforms have successfully supported the launch of gene therapies like Zolgensma and Luxturna, often utilizing scale-out technologies such as fixed-bed bioreactors or roller bottles [18]. Conversely, the insect cell-BEVS system is recognized for its high yield and cost-effectiveness, with Sf9 cells being particularly robust for virus amplification and protein synthesis in suspension cultures [17].
Visualizing the Cultivation Workflow
The following diagram outlines the general workflow for the culture and subculturing of both adherent mammalian and insect cells, highlighting key divergences in their handling.
Detailed Cell Dissociation and Subculture Protocols
Proper dissociation is critical for maintaining cell viability and functionality. The protocols below are tailored to the distinct biological needs of each cell type.
Protocol for Adherent Mammalian Cells
This protocol is optimized for strongly adherent mammalian cells like HEK293, using trypsin-EDTA for efficient detachment [11] [5].
Materials:
- Pre-warmed complete growth medium
- Pre-warmed balanced salt solution (e.g., PBS without calcium and magnesium)
- Pre-warmed Trypsin-EDTA solution (e.g., 0.25%)
- 37°C incubator with 5% CO₂
Procedure:
- Confirm Log Phase: Ensure cells are in log phase with viability >90% and sub-confluent [11].
- Remove Medium: Aspirate and discard the spent culture medium completely.
- Wash Cell Layer: Gently add a sufficient volume of pre-warmed PBS without Ca²⁺/Mg²⁺ to the flask (e.g., 2 mL per 10 cm² surface area). Rock the vessel gently and discard the wash. This step removes serum and divalent cations that inhibit trypsin [11] [5].
- Add Trypsin-EDTA: Add enough pre-warmed trypsin-EDTA to cover the cell layer (e.g., 0.5 mL per 10 cm²). Gently rock the vessel for complete coverage [11].
- Incubate: Place the culture vessel at 37°C for approximately 2 minutes. Incubation time varies by cell line. Observe cells under a microscope. If less than 90% are detached, prolong incubation in 30-second increments, tapping the vessel if needed [11].
- Neutralize: When most cells are detached and rounded, quickly add a volume of complete growth medium that is at least double the volume of trypsin-EDTA used. Serum in the medium inactivates the trypsin. Pipette the medium over the cell layer surface to ensure full dissociation [11] [5].
- Centrifuge and Resuspend: Transfer the cell suspension to a centrifuge tube and spin at 200 × g for 5–10 minutes. Resuspend the cell pellet in a minimal volume of fresh pre-warmed medium [11].
- Count and Seed: Perform a cell count and viability assessment (e.g., via Trypan Blue exclusion). Seed new culture vessels at the recommended density [11].
Protocol for Adherent Insect Cells
This protocol is designed for adherent insect cells like Sf9 and Sf21, which attach very tightly and require specific conditions [11].
Materials:
- Complete insect cell growth medium (e.g., Grace's Medium)
- Balanced salt solution or EDTA
- Appropriate dissociation reagent (e.g., TrypLE, Accutase, or trypsin-EDTA)
- Environment maintained at 27°C (non-humidified)
Procedure:
- Determine Confluency: Passage insect cells at confluency or slightly after. Passaging before confluency requires more force and can damage cells, while repeated passaging at high densities decreases viability [11].
- Remove Medium: Aspirate and discard the spent culture medium.
- Wash Cell Layer: Wash the cell monolayer with a balanced salt solution or EDTA to help initiate dissociation [11] [5].
- Add Dissociation Reagent: Apply the dissociation solution to the vessel. Insect cells are notoriously tightly attached under serum-free conditions and may require a stronger reagent or a unique approach [11].
- Incubate and Dislodge: Incubate the vessel at room temperature. Unlike mammalian cells, a 27°C incubator is not required for this step. Observe cells under a microscope. If cells remain adherent, a quick, sharp shake ("wrist-snapping motion") with the cap tightened may be necessary to dislodge them. Do not shake vigorously [11].
- Neutralize: Once cells are detached, add complete insect cell growth medium to neutralize the reaction. For some protocols, centrifugation is not required, and the cell suspension can be diluted directly for seeding new flasks [5].
- Count and Seed: Perform a cell count and seed new vessels at the recommended density. Maintain cultures in a 27°C environment without CO₂ control [11].
The Scientist's Toolkit: Essential Reagents
Table 2: Key Reagents for Cell Culture and Dissociation
| Reagent | Function | Application Notes |
|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme (trypsin) breaks down cell-adhesion proteins. EDTA chelates calcium and magnesium, further promoting dissociation [10]. | Standard for most adherent mammalian cells. Concentration (e.g., 0.05%, 0.25%) and incubation time must be optimized to avoid damage [11] [5]. |
| TrypLE | A recombinant fungal protease that functions like trypsin. | Animal origin-free alternative. Often used for robustly adherent cells and in insect cell culture [5]. |
| Accutase | A blend of proteolytic and collagenolytic enzymes. | Gentler on cell surface proteins. Ideal for sensitive cells like stem cells and primary cells [5]. |
| Collagenase | Enzyme that degrades native collagen. | Used for dissociating dense tissues or fibroblast cultures, often in combination with other enzymes [19] [5]. |
| Dispase | Neutral protease that dissociates cells without damaging cell membranes. | Suitable for maintaining cell-to-cell connections, such as harvesting epidermal sheets [5]. |
| EDTA Solution | Chelating agent that binds Ca²⁺ and Mg²⁺ ions, disrupting integrin-mediated adhesion. | Can be used alone for weakly adherent cells or in combination with enzymes to enhance efficiency [10] [5]. |
| Enzyme-Free Dissociation Buffer | Cell-stripping solution that chelates ions without enzymatic activity. | The gentlest method, preserving surface epitopes for immunostaining or flow cytometry [5]. |
Quantitative Data and Performance Metrics
Viral Vector Manufacturing Platforms
Table 3: Comparison of Adherent and Suspension Platforms for Viral Vector Manufacturing
| Metric | Adherent Platform | Suspension Platform |
|---|---|---|
| Industry Adoption (2021) | ~70% of viral vector products [18] | Growing adoption, viewed as "must-have" for some products [18] |
| Scalability Method | Scale-out (adding more surface area, e.g., stacks, rollers) [18] | Scale-up (increasing volume in a single bioreactor) [18] |
| Upfront Investment (CaPex) | Lower [18] | Higher |
| Process Development | Less complex bioengineering know-how required [18] | More expert bioengineering know-how needed [18] |
| Example Commercial Products | Luxturna (roller bottles), Zolgensma (iCELLis bioreactor) [18] | Glybera [18] |
Insect Cell Line Performance
Table 4: Operational Characteristics of Common Insect Cell Lines
| Cell Line | Origin | Key Features and Optimal Use |
|---|---|---|
| Sf9 | Ovary of Spodoptera frugiperda (subclone of Sf21) | High growth rate, tolerant of high densities and shear stress, uniform morphology. Ideal for virus amplification and high-yield protein production in suspension [17]. |
| Sf21 | Ovary of Spodoptera frugiperda | Highly susceptible to viral infection. Excellent for initial plaque assays and assessing virus cytopathic effects [17]. |
| HighFive | Ovary of Trichoplusia ni | Can offer higher expression levels for secreted proteins. May produce more proteases, risking target protein degradation [17]. |
Advanced Considerations and Future Directions
The field of cell culture is continuously evolving. Key areas of development include the engineering of insect cell lines, like SfSWT-1, to produce mammalian-like glycosylation patterns, thereby enhancing the therapeutic relevance of expressed proteins [17]. Furthermore, non-enzymatic dissociation methods—utilizing physical forces such as ultrasound or electrical fields—are being developed to improve cell viability and reduce artifacts in sensitive downstream applications like single-cell analysis [19]. For mammalian cell bioprocessing, the industry is grappling with the "scale-out vs. scale-up" dilemma, with a clear trend towards adapting suspension platforms to improve scalability and cost-effectiveness for high-dose therapeutics, while adherent processes remain viable for specific applications [18].
Within the context of trypsin-EDTA cell dissociation protocol research, rigorous safety and quality control are not merely supplementary steps but foundational requirements for generating reliable, reproducible, and translatable data. The process of cell dissociation, essential for subculturing and harvesting cells, can inadvertently introduce or concentrate contaminants that compromise experimental outcomes and patient safety in drug development. Chief among these concerns are bacterial endotoxins, potent pyrogenic molecules derived from the outer membrane of Gram-negative bacteria [20]. These lipopolysaccharides (LPS) are remarkably stable and can persist through standard sterilization processes, posing a significant risk to cell health, skewing experimental results in areas like immunology and oncology research, and presenting a profound safety hazard for any parenterally administered therapeutics [21] [20]. This application note provides detailed protocols and frameworks for researchers and drug development professionals to integrate robust endotoxin and contaminant testing into their cell culture workflows, with a specific focus on the trypsin-EDTA dissociation process.
Understanding Endotoxins and Their Impact on Research
What are Endotoxins?
Endotoxins are complex lipopolysaccharides (LPS) that form a protective barrier on the outer membrane of Gram-negative bacteria. Structurally, they consist of a toxic lipid A component, a core oligosaccharide, and an O-specific polysaccharide chain (O-antigen) [20]. The lipid A domain is responsible for most of the biological toxicity. A critical challenge in the lab is that endotoxins are highly heat-stable and can readily adsorb to the surfaces of common plasticware and glass, making them persistent contaminants that are not removed by standard autoclaving [20].
Consequences of Endotoxin Contamination
The presence of endotoxins in cell culture systems can lead to a cascade of undesirable effects, including:
- Alteration of Cell Phenotype and Function: Endotoxins are potent immune activators. In cell cultures, they can trigger cells, particularly immune cells or those with innate immune receptors, to undergo activation, proliferate, or secrete cytokines, thereby skewing experimental data [20].
- Reduced Transfection Efficiency: The presence of endotoxins can significantly impair the efficiency of transfection processes, a critical step in many genetic and drug discovery pipelines [20].
- Pyrogenic Response In Vivo: For any cell-derived product intended for therapeutic use in humans or animals, even trace amounts of endotoxin can induce fever, inflammation, shock, and sepsis upon administration [21] [20]. Regulatory agencies therefore set strict limits on allowable endotoxin levels in pharmaceuticals and biologics.
Essential Endotoxin Testing Methodologies
The Limulus Amebocyte Lysate (LAL) test is the industry standard for endotoxin detection. Its principle is based on the unique innate immune response of the Atlantic horseshoe crab (Limulus polyphemus), whose blood contains amoebocytes. When these cells encounter endotoxin, a cascade of enzymatic reactions is triggered, culminating in the formation of a clot [21] [20] [22]. The LAL test harnesses this reaction in a lysate prepared from these cells.
Comparison of Primary LAL Test Formats
Researchers can select from several LAL-based assay formats, each with distinct advantages and applications. The following table summarizes the key characteristics of the main quantitative methods.
Table 1: Comparison of Quantitative Bacterial Endotoxin Testing Methods
| Method | Principle of Detection | Sensitivity Range | Key Advantages | Key Considerations |
|---|---|---|---|---|
| Gel-Clot LAL [21] [20] | Visual observation of gel formation (clot) | Varies by reagent sensitivity (e.g., 0.03 EU/mL) | Economical, simple, qualitative/semi-quantitative | Subjectivity in reading; less precise |
| Turbidimetric LAL [21] | Measures increase in turbidity during clot formation | Down to 0.001 EU/mL [21] | Quantitative, can be automated | Requires instrumental analysis |
| Chromogenic LAL [21] [20] | Measures color change from a cleaved synthetic substrate | Down to 0.01 EU/mL [20] | Quantitative, high sensitivity, can be automated | Requires instrumental analysis |
| Fluorimetric LAL [20] | Measures fluorescence from a cleaved substrate | 0.01 - 10.0 EU/mL [20] | Quantitative, very high sensitivity, reduced interference | Requires specific fluorometer |
The LAL Coagulation Cascade
The biochemical principle of the LAL test involves a tightly regulated serine protease cascade. The following diagram illustrates the two primary pathways within the lysate that lead to clot formation.
Diagram Title: LAL Test Coagulation Cascade Pathways
Detailed Protocol: Endotoxin Testing for Trypsin-EDTA Reagents
This protocol is adapted for testing trypsin-EDTA solutions or other cell dissociation reagents for endotoxin contamination using a chromogenic LAL assay, providing a quantitative result.
Pre-Assay Preparation and Considerations
- Principle: A sample is mixed with LAL reagent and incubated. Endotoxins activate the enzymatic cascade, culminating in the cleavage of a colorimetric substrate. The released yellow p-nitroaniline is measured at 405 nm, with intensity proportional to the endotoxin concentration [21].
- Key Resources:
- LAL Reagent: Chromogenic formulation with a known sensitivity.
- Endotoxin Standard: Control Standard Endotoxin (CSE) derived from E. coli 0113, calibrated against a Reference Standard Endotoxin (RSE) [21] [20].
- Endotoxin-Free Water/Labware: Specifically certified for LAL testing. Standard "sterile" consumables may still contain endotoxins [20].
- Microplate Reader: Capable of reading absorbance at 405 nm.
- Sample Preparation: Trypsin-EDTA solutions may require dilution in endotoxin-free water to fall within the standard curve's dynamic range and to overcome any pH- or chemistry-mediated assay inhibition [20]. The required dilution factor must be validated via a spike recovery test.
Step-by-Step Workflow
Table 2: Step-by-Step Chromogenic LAL Assay Protocol
| Step | Procedure | Critical Parameters & Tips |
|---|---|---|
| 1. Preparation | [21] [20] | Use endotoxin-free tips and tubes. Change gloves frequently to prevent contamination. |
| 2. Standard Curve | [20] | A minimum of 3 standard points (e.g., 5.0, 0.5, 0.05 EU/mL) plus a blank is recommended. |
| 3. Sample Loading | [20] | Include a "spiked" sample control (your sample + a known amount of standard) to validate the dilution factor and check for inhibition/enhancement. Recovery should be 50-200% (target 75-150%) [20]. |
| 4. Reaction | [21] | Ensure precise and consistent pipetting timing, as the reaction is time-sensitive. |
| 5. Measurement & Analysis | [20] | The standard curve must have a correlation coefficient (r) of ≥ 0.98 [20]. Multiply the result from the curve by the sample's dilution factor to obtain the original concentration. |
Troubleshooting and Quality Controls
- Assay Validation: Each assay run must include a standard curve that meets linearity criteria (r ≥ 0.98) [20].
- Spike Recovery Test: This is essential to confirm that the sample matrix itself does not interfere with the LAL reaction. Failure to recover the spiked endotoxin within the acceptable range (typically 50-200%) invalidates the test for that sample, and the sample must be re-tested at a different dilution [20].
- Positive Product Control (PPC): For regulated environments, a PPC demonstrates that the test method is suitable for the product under test.
The Scientist's Toolkit: Essential Reagents for Contaminant Testing
Table 3: Key Research Reagent Solutions for Endotoxin Testing
| Reagent / Solution | Function & Application in Testing |
|---|---|
| Limulus Amebocyte Lysate (LAL) [21] [20] | The core enzyme reagent derived from horseshoe crab blood cells; detects endotoxin via a coagulation cascade. |
| Control Standard Endotoxin (CSE) [21] [20] | A standardized preparation of endotoxin used to calibrate the assay and create the standard curve for quantification. |
| Endotoxin-Free Water [20] | Used for reconstituting reagents, diluting samples and standards; prevents introduction of background endotoxin. |
| Chromogenic Substrate [21] | A synthetic peptide linked to p-nitroaniline; cleaved by the clotting enzyme to produce a measurable color change. |
| Trypsin-EDTA Solution [23] [24] | The subject of testing; a proteolytic enzyme (trypsin) and a chelating agent (EDTA) used to detach adherent cells for subculturing. |
| TrypLE Reagents [24] | Animal-origin-free, recombinant alternatives to trypsin; reduce the risk of introducing animal-sourced contaminants like viruses. |
Navigating Challenges and Future Outlook
A significant challenge in endotoxin testing, particularly with novel formulations like nanoparticles or complex biologics, is assay interference. Components can inhibit or enhance the LAL reaction, leading to underestimation or overestimation of endotoxin levels [20]. Rigorous validation with spike recovery tests is paramount. Furthermore, the LAL assay itself can be affected by (1,3)-β-D-glucans from fungi, which activate an alternative pathway via Factor G [21] [22]. Using LAL reagents formulated with specific buffers to block the glucan pathway can mitigate this.
The field is evolving with the increased adoption of recombinant assay methods, which use synthetic factors instead of native horseshoe crab lysate [20]. These methods address concerns about sustainable harvesting of horseshoe crabs and offer a more standardized, animal-free reagent. However, they may require additional validation for use in regulated workflows. For laboratories where results from different LAL assays differ by more than 25%, verification with the in vivo rabbit pyrogen test is recommended [21]. The integration of automation and high-throughput systems is also streamlining BET in industrial bioprocessing, ensuring that millions of cells processed for therapies are safe and contaminant-free [8].
Mastering the Protocol: Step-by-Step Guide and Specialized Applications
Standardized Protocol for Routine Subculturing of Adherent Cells
Routine subculturing, or passaging, is a fundamental process in maintaining the health and longevity of adherent mammalian cell lines. It involves the detachment of cells from a culture surface and their subsequent transfer to new vessels to provide space and nutrients for continued growth [25]. The dissociation of adherent cells is most commonly achieved using a trypsin-EDTA solution, a proteolytic enzyme mixture that cleaves the proteins responsible for cell adhesion [26]. This application note details a standardized protocol for the trypsin-EDTA based subculturing of adherent cells, framed within broader research on optimizing cell dissociation techniques. The consistent application of this protocol is crucial for researchers, scientists, and drug development professionals to ensure experimental reproducibility, maintain cell line integrity, and generate reliable data for pharmaceutical and clinical applications [27] [28].
Principles of Cell Dissociation with Trypsin-EDTA
Trypsin-EDTA solution functions through a synergistic biochemical mechanism to efficiently detach adherent cells. Trypsin, a serine protease, cleaves peptide bonds on the carboxyl side of lysine and arginine residues in proteins [26]. In the context of cell adhesion, it digests key extracellular matrix (ECM) proteins and cell surface molecules, such as fibronectin and integrins, which anchor the cell to the substrate [26] [28].
EDTA (Ethylenediaminetetraacetic acid) complements this action by chelating, or binding, divalent cations such as calcium and magnesium (Ca²⁺ and Mg²⁺) [26]. These ions are essential cofactors for many cell adhesion molecules, including cadherins that mediate cell-to-cell junctions [26]. By sequestering these cations, EDTA disrupts these interactions, leading to further dissociation of the cell monolayer [28].
The combination of trypsin and EDTA ensures efficient and complete detachment of cells into a single-cell suspension, which is vital for accurate cell counting and uniform reseeding [26]. However, it is critical to neutralize trypsin's activity promptly after detachment, as prolonged exposure can damage membrane proteins, alter the cell cytoplasm, and reduce viability [29] [26].
Materials and Reagents
Research Reagent Solutions
The following table details the essential materials required for the subculturing procedure.
Table 1: Essential Reagents and Materials for Subculturing
| Item | Function & Specification |
|---|---|
| Trypsin-EDTA Solution | Proteolytic enzyme solution (typically 0.05-0.25%) with a chelating agent to dissociate adherent cells by cleaving adhesion proteins and sequestering calcium [26]. |
| Complete Growth Medium | Culture medium (e.g., DMEM, RPMI) supplemented with serum (e.g., 10% FBS), glutamine, and antibiotics. Provides nutrients and is used to neutralize trypsin activity [27]. |
| Balanced Salt Solution (without Ca²⁺/Mg²⁺) | e.g., Dulbecco's Phosphate-Buffered Saline (DPBS). Used to wash cells prior to dissociation, removing residual serum and ions that inhibit trypsin [11]. |
| Adherent Cell Line | Cells of interest, cultured in an appropriate vessel and monitored until they reach 70-80% confluency [27]. |
| Cell Culture Vessels | Sterile flasks, dishes, or plates. Vented caps allow for proper gas exchange [11] [27]. |
Equipment
- Laminar flow hood (Class II recommended) [27]
- Humidified CO₂ incubator (set to 37°C, 5% CO₂) [11]
- Inverted phase-contrast microscope [28]
- Centrifuge [11]
- Hemocytometer or automated cell counter [11] [25]
- Water bath or bead bath (set to 37°C) [27]
- Serological pipettes, micro-pipettes, and sterile tips [27]
Methodology: Standardized Subculturing Protocol
Pre-subculture Assessment and Preparation
- Microscopic Examination: Prior to passaging, examine cells daily under an inverted microscope. Check for cell health, morphology, and confirm the absence of bacterial or fungal contamination [28]. Cells should be subcultured when they are in the log phase of growth and have reached 70-80% confluency [11] [25] [27].
- Warm Reagents: Pre-warm complete growth medium, trypsin-EDTA solution, and balanced salt solution to 37°C in a water bath [27].
- Aseptic Technique: Perform all subsequent steps in a laminar flow hood using sterile technique. Spray all surfaces and equipment with 70% ethanol before placing them in the hood [27].
Cell Dissociation and Passaging Workflow
The following diagram illustrates the key stages of the subculturing protocol.
Diagram 1: Adherent Cell Subculture Workflow.
Step-by-Step Detailed Protocol:
- Remove Spent Medium: Aspirate and discard the spent cell culture media from the culture vessel [11].
- Wash Cell Layer: Add a balanced salt solution without calcium and magnesium (approximately 2 mL per 10 cm² surface area) to wash the cells. Gently rock the vessel and then aspirate the solution. This step removes any traces of serum that would inhibit trypsin activity [11].
- Add Trypsin-EDTA: Add pre-warmed trypsin-EDTA solution to the side of the vessel (approximately 0.5 mL per 10 cm²). Gently rock the container to ensure complete coverage of the cell layer [11].
- Incubate for Detachment: Incubate the culture vessel at 37°C for approximately 2-5 minutes. The actual incubation time varies with the cell line [11].
- Monitor Detachment: Observe the cells under the microscope. Cells will round up and detach. If less than 90% are detached after the initial incubation, tap the vessel gently or extend the incubation time in 30-second increments [11].
- Neutralize Trypsin: When ≥90% of cells have detached, promptly add a volume of pre-warmed complete growth medium equivalent to at least twice the volume of trypsin used. Disperse the medium by pipetting over the cell layer surface several times to ensure a single-cell suspension and neutralize the enzyme [11] [26].
- Transfer and Centrifuge: Transfer the cell suspension to a conical tube and centrifuge at 200 × g for 5-10 minutes to form a cell pellet [11].
- Resuspend and Count: Resuspend the cell pellet in a minimal volume of fresh, pre-warmed complete growth medium. Remove a sample for counting and viability assessment using a hemocytometer and Trypan Blue exclusion or an automated cell counter [11] [25]. Cell viability should be greater than 90% at the time of subculturing [11].
Post-subculture Seeding
- Dilute Cell Suspension: Based on the cell count, dilute the cell suspension with complete growth medium to achieve the recommended seeding density for the specific cell line.
- Seed New Vessels: Pipette the appropriate volume of cell suspension into new, labeled culture vessels.
- Incubate: Loosen the caps on non-vented flasks and place all vessels in the incubator (37°C, 5% CO₂) [11].
- Documentation: Maintain a detailed cell culture log, listing the passage number, split ratio, seeding density, morphological observations, and date [25].
Quality Control and Troubleshooting
Quantitative Parameters for Subculturing
Adherent cell growth follows a characteristic pattern. Subculturing should be performed during the log phase to maintain optimal health.
Table 2: Key Growth Phase Parameters for Subculturing
| Growth Phase | Description | Cell Confluency | Action |
|---|---|---|---|
| Lag Phase | Period of adaptation after seeding; slow growth [25]. | < 40% | Monitor; not ideal for passaging. |
| Log Phase | Exponential cell proliferation; optimal health [25]. | 70-80% | Ideal time for subculturing. |
| Stationary Phase | Growth plateaus due to confluency or nutrient depletion [25]. | 100% (Confluent) | Avoid passaging; cells take longer to recover. |
| Death Phase | Cell death due to nutrient exhaustion and toxic metabolite accumulation [28]. | Variable, deteriorating | Do not passage; discard culture. |
Table 3: Common Subculturing Problems and Solutions
| Problem | Possible Cause | Suggested Solution |
|---|---|---|
| Slow or Incomplete Detachment | Insufficient trypsin activity; high cell density; outdated reagents. | Use fresh, pre-warmed trypsin-EDTA. Ensure wash step is thorough. Optimize incubation time for cell type [26]. |
| Rapid pH Shift in Medium | Incorrect CO₂ tension in incubator; high cell concentration. | Check and calibrate CO₂ levels (5-10% based on bicarbonate concentration). Subculture cells before over-confluency [25]. |
| Poor Cell Viability Post-Seeding | Over-exposure to trypsin; inaccurate centrifugation speed/time. | Neutralize trypsin immediately after detachment. Adhere to recommended centrifugation protocols (e.g., 200 × g for 5-10 min) [11] [29]. |
| Morphological Changes | Contamination; repeated passaging at high density. | Check for contamination. Avoid over-confluency and track passage numbers to prevent genotypic drift [25] [27]. |
Applications in Drug Development and Research
The standardized subculturing of adherent cells is a cornerstone technique enabling various downstream applications critical in pharmaceutical and biomedical research.
- High-Throughput Drug Screening: Consistent and healthy cell cultures are essential for generating reliable data in assays that test compound efficacy and toxicity [30]. Automated trypsinization, using robotic liquid handlers, increases throughput, consistency, and reduces human error in these workflows [31].
- Cell-Based Assays: Subculturing is used to prepare cells for proliferation assays, transfection studies, and flow cytometry, ensuring that experiments begin with a uniform single-cell suspension [31] [26].
- Biomanufacturing and Cell Therapy: In upstream bioprocessing, robust subculturing protocols are vital for scaling up cell cultures to produce therapeutic proteins, vaccines, and advanced therapy medicinal products (ATMPs) [32]. Process intensification techniques like perfusion culture rely on precise cell biology management [32].
This application note provides a comprehensive and standardized protocol for the routine subculturing of adherent cells using trypsin-EDTA. Adherence to this protocol—emphasizing aseptic technique, precise timing, proper reagent preparation, and consistent quality control—is fundamental to maintaining healthy, reproducible cell cultures. As the field advances with trends like automation [31], advanced real-time monitoring [32], and the development of recombinant trypsin alternatives [26], the underlying principles of careful and consistent cell culture practice remain paramount for successful research and drug development outcomes.
Within the broader context of methodological research on trypsin-EDTA cell dissociation, the preparatory steps of reagent warming, equipment sterilization, and aseptic technique are critical determinants of experimental success. These pre-processing protocols directly impact cellular viability, the reproducibility of dissociation kinetics, and the reliability of downstream applications such as single-cell sequencing, flow cytometry, and adhesion studies [33] [34]. Trypsin, a proteolytic enzyme, functions by cleaving adhesion proteins at the C-terminal side of lysine and arginine residues, but its activity is intrinsically linked to temperature and the absence of inhibitors like serum [24]. Proper reagent preparation, including temperature equilibration, ensures consistent enzymatic activity, while strict asepsis preserves cellular integrity by preventing microbial contamination that could compromise experimental outcomes and lead to erroneous biological conclusions [4]. This application note details standardized protocols to optimize these foundational steps, thereby minimizing the unintended cellular stress and proteome alterations associated with trypsinization [35].
Quantitative Data on Trypsin-Induced Cellular Effects
A comprehensive understanding of trypsin's effects underscores the necessity of precise preparatory control. The following tables summarize key quantitative findings on its impact on cellular physiology.
Table 1: Documented Effects of Trypsin-EDTA on Cell Physiology
| Cellular Component/Process | Observed Effect | Quantitative Change/Correlation | Experimental Method | Citation |
|---|---|---|---|---|
| Cytoplasmic Content | Alteration of small solutes (electrolytes, metabolites) | Cytoplasmic modification begins within seconds | Terahertz Sensing, Confocal Microscopy | [29] |
| Cell Volume & Mechanics | Change in cell volume due to mechanical re-equilibrium | Non-linear correlation with cell height | Terahertz Sensing | [29] |
| Cell Surface Proteome | Cleavage of cell surface proteins | 36 proteins differentially expressed | 2D-DIGE, MALDI-TOF MS | [35] |
| Apoptosis Regulation | Up-regulation of pro-apoptotic proteins | p53 and p21 up-regulated; bcl-2 down-regulated | Immunoblotting | [35] |
| Functional Adhesion | Altered re-adhesion to various surfaces | Adhesivity significantly altered post-detachment | Label-free Optical Sensor | [34] |
Table 2: Comparative Efficacy of Tissue Dissociation Methods
| Dissociation Method | Dissociation Agent(s) | Reported Efficiency | Reported Viability | Typical Application | |
|---|---|---|---|---|---|
| Chemical Only | 1% Collagenase + Pronase/Hyaluronidase | 37-42% | >90% | Frozen bovine liver biopsy cores | [33] |
| Chemical-Mechanical | 1% Collagenase + Pronase + Controlled Force | 93% ± 8% | Not specified | Frozen bovine liver biopsy cores | [33] |
| Enzymatic (Standard) | 0.25% Trypsin-EDTA | High for monolayers | >90% (if optimized) | Strongly adherent cell lines | [4] [24] |
| Non-Enzymatic | Cell Dissociation Buffer (EDTA-based) | Lower for strong adhesion | Retains surface protein integrity | Weakly adherent cells, flow cytometry | [4] [34] |
Detailed Experimental Protocols
Standardized Trypsin-EDTA Cell Dissociation Protocol
The following step-by-step protocol is optimized for adherent mammalian cell lines to ensure high viability and reproducibility.
Title: Standard Trypsin-EDTA Dissociation of Adherent Cell Monolayers
1. Reagent Preparation and Warming:
- Pre-warm Trypsin-EDTA solution (e.g., 0.25% for standard cell lines), a balanced salt solution (e.g., DPBS without Calcium and Magnesium), and complete growth medium to 37°C in a water bath [4] [24].
- Critical Note: Minimize the dwell time of trypsin at 37°C before use to prevent activity loss. Accurately timed warming ensures consistent enzymatic activity, which is crucial as studies show cytoplasmic alterations can begin within seconds of trypsin exposure [29] [4].
2. Equipment Sterilization and Aseptic Setup:
- Perform all subsequent steps within a certified Class II biosafety cabinet.
- Wipe down all reagent containers, pipettes, and surfaces with 70% ethanol before placing them in the cabinet.
- Use sterile pipettes and consumables. The aseptic technique is paramount to prevent contamination, which can skew downstream assays like single-cell sequencing [33].
3. Cell Monolayer Washing:
- Aspirate and discard the spent cell culture media completely.
- Wash the cell monolayer with 3-5 mL of pre-warmed DPBS (without Ca2+ and Mg2+) per 75 cm² flask to remove any residual serum, which contains trypsin inhibitors [24].
- Gently rock the flask for 1-2 minutes to bathe the entire cell sheet, then completely aspirate the wash solution [4]. This step is critical for achieving efficient trypsin activity.
4. Trypsin-EDTA Application and Incubation:
- Add the pre-warmed Trypsin-EDTA solution at a volume of 2-3 mL per 25 cm² of surface area, ensuring it covers the entire cell monolayer [4].
- Incubate the flask at 37°C for 5-15 minutes. The optimal time is cell line-dependent and must be determined empirically [4].
- Gently rock the flask periodically and monitor detachment under an inverted microscope. Critical Note: Avoid prolonged incubation, as it leads to increased proteolytic damage, cleavage of surface markers, and reduced cell viability, as evidenced by proteomic studies [35].
5. Enzymatic Inhibition and Cell Harvesting:
- Once cells are fully detached (they will appear rounded and float), immediately add a volume of pre-warmed complete growth medium that is at least double the volume of trypsin used. The serum in the medium inactivates the trypsin [4] [24].
- For animal origin-free systems, use a specialized trypsin neutralizer solution or simply dilute the cell suspension significantly with buffer [24].
- Gently pipette the cell suspension to break up any clumps and transfer it to a 15 mL conical tube.
6. Cell Pellet Formation and Resuspension:
- Centrifuge the tube at approximately 100 × g for 5-10 minutes to form a cell pellet [4].
- Carefully decant the supernatant and resuspend the cell pellet in 2-5 mL of fresh, pre-warmed complete growth medium.
7. Viability and Density Assessment:
- Determine viable cell density and percent viability using an automated cell counter (e.g., Countess) or manual hemocytometer with Trypan Blue exclusion. Viability should routinely be >90% [4].
- The cells are now ready for downstream applications, such as sub-culturing, flow analysis, or re-plating for adhesion experiments [34].
Protocol for Assessing Post-Dissociation Cellular Adhesion
The choice of dissociation method directly influences subsequent cell adhesion, which can be quantified using the following protocol, adapted from label-free optical sensor studies [34].
Title: Quantifying Post-Dissociation Cell Adhesion
- Surface Preparation: Prepare a multi-well plate with at least two different surfaces of interest (e.g., non-coated, fibronectin-coated, RGD-coated) [34].
- Cell Processing: Dissociate a single population of cells (e.g., HeLa cells) and split into aliquots. Treat each aliquot with a different dissociation reagent (e.g., Trypsin-EDTA, EDTA alone, Cell Dissociation Buffer) for a standardized time [34].
- Re-plating: Immediately after neutralization and counting, seed a consistent number of cells from each treatment group onto the pre-prepared surfaces.
- Adhesion Measurement: Incubate for a short, defined period (e.g., 20-60 minutes) to allow for initial adhesion. Then, use a label-free optical sensor or other suitable method to quantify the number of adhered cells and the strength of adhesion on each surface for each dissociation group [34].
- Data Analysis: Compare adhesion metrics across dissociation methods and surfaces. The conclusion on which surface is "more adhesive" may vary significantly depending on the dissociation method used [34].
Mechanistic Analysis of Trypsin Action and Workflow
The following diagrams illustrate the experimental workflow for standardized dissociation and the subsequent cellular impacts of trypsinization.
Diagram 1: Cell dissociation workflow.
Diagram 2: Trypsin-induced cellular effects.
The Scientist's Toolkit: Research Reagent Solutions
Selecting the appropriate dissociation reagent is crucial for specific applications and cell types. The table below catalogs key reagents and their functions.
Table 3: Essential Reagents for Cell Dissociation Research
| Reagent / Solution | Key Function / Composition | Primary Application & Rationale |
|---|---|---|
| Trypsin-EDTA (0.25%) | Proteolytic enzyme (cleaves lysine/arginine) + chelating agent (binds Ca2+/Mg2+). | General purpose for strongly adherent cell lines. EDTA enhances trypsin efficacy by weakening cell-cell adhesions [4] [24]. |
| TrypLE Express | Recombinant, fungal-derived trypsin-like enzyme. | Gentle, animal origin-free (AOF) substitute for porcine trypsin; stable at room temperature and inactivated by dilution [4] [24]. |
| Cell Dissociation Buffer | Non-enzymatic, Hanks'- or PBS-based buffer with chelating agents and salts. | Gently detaches weakly adherent cells while preserving cell surface protein integrity for flow cytometry or ligand-binding studies [24] [34]. |
| Collagenase (Type I, II, IV) | Enzyme digesting native collagen fibrils. | Disaggregation of connective tissues (e.g., liver, fat, epithelium). Type selection is tissue-dependent [4] [33] [24]. |
| Dispase II | Neutral protease (metalloenzyme) from Bacillus polymyxa. | Harvests cells as intact sheets; ideal for detaching epithelial cells and primary cultures with minimal membrane damage [4] [24]. |
| Versene Solution (EDTA) | Low-concentration EDTA in a balanced salt solution. | Mild, non-enzymatic dissociation of epithelial cells or as a wash prior to trypsinization to enhance efficiency [24]. |
| Soybean Trypsin Inhibitor | Protein that binds and irreversibly inactivates trypsin. | Used post-dissociation to halt proteolytic activity immediately, especially in serum-free conditions [4] [24]. |
In cell biology and immunology research, the term "neutralization" applies to two distinct concepts: the inhibition of biological activity by antibodies in serum and the quenching of enzymatic activity by specific inhibitors during laboratory protocols. The choice between serum-based neutralization and inhibitor-based neutralization is highly context-dependent, dictated by the experimental media and specific research objectives. Serum neutralization is predominantly used in virology and immunology to assess immune responses against pathogens [36] [37], while specific inhibitors are essential in molecular and cell biology for controlling proteolytic activity during cell culture maintenance [24] [38]. This application note delineates these two neutralization methodologies, providing structured quantitative comparisons and detailed protocols to guide researchers in selecting the appropriate system for their experimental needs.
Serum-Mediated Neutralization Assays
Serum neutralization assays measure the capacity of antibodies in serum to inhibit biological processes such as viral infection or receptor-ligand interactions. These assays are crucial for evaluating immune responses in vaccine development, infectious disease studies, and therapeutic antibody characterization.
Key Assay Formats and Quantitative Outcomes
Table 1: Serum Neutralization Assay Formats and Key Parameters
| Assay Format | Detection Method | Target System | Key Quantitative Findings | Reference |
|---|---|---|---|---|
| Focus Reduction Neutralization Test (FRNT) | Immunostaining / Peroxidase substrate | SARS-CoV-2 (Vero E6 cells) | Measures 50% neutralization titer (FRNT50); High-throughput 96-well format | [36] |
| ACE2 Competition Cell-Based ELISA | Colorimetric ELISA | SARS-CoV-2 RBD / ACE2 interaction | Reports serum neutralization efficiency via ACE2 competition | [39] |
| CoreTIA (Transduction Inhibition Assay) | Luminescence (Luciferase) | AAV Neutralizing Antibodies (HEK293T cells) | Defines ND50 (50% Neutralizing Dose); Bayesian statistical analysis | [37] |
| Longitudinal nAB Durability Study | Immunoassay | SARS-CoV-2 variants | 2.5-4.6-fold reduction in Geometric Mean Titer (GMT) over 6 months in older adults | [40] |
Detailed Protocol: Focus Reduction Neutralization Test (FRNT) for SARS-CoV-2
Day 1: Cell Seeding
- Grow Vero C1008 (clone E6) cells to confluence in T175 flasks.
- Remove media and wash the cell monolayer with 5 mL of DPBS.
- Add 5 mL of 0.25% Trypsin-EDTA and incubate at 37°C for 10-15 minutes.
- Add 5 mL of complete DMEM (cDMEM) to neutralize trypsin and mix thoroughly.
- Count cells using trypan blue and resuspend at a density of 2.75 × 105 cells/mL in cDMEM.
- Seed a 96-well tissue culture-treated plate with 150 µL of cell suspension per well (4.125 × 104 cells/well).
- Incubate at 37°C with 5% CO2 for approximately 24 hours to achieve 100% confluency [36].
Day 2: Serum-Virus Incubation and Infection
- Prepare serial dilutions of heat-inactivated serum or plasma samples in a separate 96-well plate.
- Incubate a fixed titer of infectious SARS-CoV-2 virus with the serum dilutions for 1 hour at 37°C.
- Remove media from the Vero E6 cell plate and inoculate each well with the serum-virus mixture.
- Centrifuge the plate at 800 × g for 30 minutes at 4°C to enhance viral adsorption (spinoculation).
- After a specified incubation period (e.g., 1 hour), remove the inoculum and overlay cells with a semi-solid medium (e.g., methylcellulose) to prevent viral spread.
- Incubate the plate at 37°C with 5% CO2 for 24-48 hours to allow focus formation [36].
Day 3: Immunostaining and Visualization
- Remove the overlay and fix cells with 4% Paraformaldehyde (PFA) for 30 minutes.
- Permeabilize cells with 0.1% saponin in PBS containing 1% BSA.
- Stain for SARS-CoV-2 foci using a primary antibody against the spike protein (e.g., CR3022), followed by a biotinylated secondary antibody and an ExtrAvidin-HRP conjugate.
- Develop foci using a precipitating HRP substrate like KPL TrueBlue.
- Quantify foci using an automated ELI-SPOT reader or manual microscopy. The neutralization titer (FRNT50) is defined as the serum dilution that reduces the number of foci by 50% compared to the virus-only control wells [36].
Figure 1: FRNT Experimental Workflow. This diagram outlines the key steps for performing a Focus Reduction Neutralization Test, highlighting the biosafety requirement for working with live SARS-CoV-2.
Specific Inhibitor-Based Neutralization
In cell culture, "neutralization" most commonly refers to the process of inhibiting proteolytic enzymes used for cell dissociation, such as trypsin, to prevent damage to cell surface proteins and ensure high cell viability post-detachment.
Cell Dissociation Enzymes and Their Neutralization Methods
Table 2: Cell Dissociation Reagents and Corresponding Neutralizers
| Dissociation Reagent | Mechanism of Action | Recommended Neutralization Method | Key Considerations | Reference |
|---|---|---|---|---|
| Trypsin | Cleaves peptide bonds after Lysine and Arginine | Serum-containing media or Specific Trypsin Inhibitors | Can degrade surface proteins; requires rapid inhibition | [24] |
| Trypsin-EDTA | Trypsin proteolysis + EDTA chelation of Ca²⁺/Mg²⁺ | Serum-containing media or Specific Trypsin Inhibitors | EDTA enhances cell detachment; same neutralization as trypsin | [24] |
| TrypLE | Recombinant trypsin-like enzyme | Inhibition by Dilution | Animal origin-free; gentle on cells; no inhibitor needed | [24] |
| Accutase | Mixture of proteolytic and collagenolytic enzymes | Dilution + Centrifugation | Considered mild but can cleave specific surface proteins (e.g., FasL) | [38] |
| Nattokinase | Serine protease from B. subtilis | Dilution + Centrifugation | Food-grade, non-animal derived; emerging alternative | [41] |
| Non-Enzymatic Buffers | Chelation (EDTA) without enzymes | Dilution + Centrifugation | Preserves surface protein integrity; ideal for flow cytometry | [24] [38] |
Impact of Dissociation Methods on Cell Surface Proteins
The choice of dissociation reagent directly impacts the integrity of cell surface proteins, which is a critical factor for downstream applications like immunophenotyping or functional assays.
Table 3: Effects of Cell Dissociation Methods on Surface Markers
| Dissociation Method | Effect on Surface FasL/Fas | Effect on Surface F4/80 | Cell Viability | Recovery Time | Reference |
|---|---|---|---|---|---|
| Cell Scraping | Minimal decrease (Gold Standard) | Preserved | Variable (mechanical stress) | Immediate | [38] |
| EDTA-based Buffer | Slight decrease after 30 min | Preserved | High | Immediate | [38] |
| Accutase | Significant decrease (Reversible) | Preserved | High (even after 90 min) | ~20 hours | [38] |
| Trypsin | Expected significant decrease | Expected significant decrease | High (with proper neutralization) | ~24 hours | [24] [38] |
Detailed Protocol: Trypsin-EDTA Cell Dissociation and Neutralization
Cell Dissociation
- Remove culture media from adherent cells and wash the monolayer with a balanced salt solution (e.g., PBS or HBSS) without calcium and magnesium to remove residual serum and divalent cations [24].
- Add sufficient pre-warmed 0.25% Trypsin-EDTA solution to cover the cell layer (e.g., 2-3 mL for a T75 flask).
- Incubate at 37°C for 2-5 minutes. Observe cells under a microscope until they round up and begin to detach. Gently tap the vessel to aid detachment [42].
- Once the majority of cells are detached, promptly add a volume of complete growth medium that is at least equal to the volume of trypsin-EDTA used. The serum in the medium (typically FBS) will neutralize the trypsin activity [24] [42].
- Pipette the cell suspension gently to break up clumps and transfer to a sterile conical tube.
Alternative Neutralization with Defined Inhibitors For serum-free workflows or when preserving surface proteins is critical, use a defined trypsin inhibitor:
- After trypsinization, transfer the cell suspension to a tube containing a pre-diluted, specific trypsin inhibitor (e.g., Soybean Trypsin Inhibitor).
- Mix gently and incubate for 2-5 minutes.
- Centrifuge the cells at 300 × g for 5 minutes to pellet them.
- Carefully aspirate the supernatant containing the neutralized trypsin and resuspend the cell pellet in fresh culture medium or buffer for downstream applications [24].
Cell Counting and Seeding
- Take an aliquot of the resuspended cells and mix with Trypan Blue solution (typically 1:1).
- Count viable (unstained) and non-viable (blue) cells using a hemocytometer or automated cell counter.
- Calculate total and viable cell concentration.
- Seed cells at the desired density for subsequent experiments or continued culture [36] [37].
Figure 2: Cell Dissociation and Neutralization Workflow. This diagram illustrates the critical steps for detaching adherent cells using trypsin-EDTA and the essential neutralization step to ensure cell viability.
The Scientist's Toolkit: Key Reagents
Table 4: Essential Reagents for Neutralization Protocols
| Reagent/Category | Specific Examples | Function/Application | Reference |
|---|---|---|---|
| Cell Dissociation Enzymes | 0.25% Trypsin-EDTA, TrypLE Express, Accutase | Detach adherent cells for subculturing or analysis | [24] [38] |
| Neutralizers/Inhibitors | Fetal Bovine Serum (FBS), Soybean Trypsin Inhibitor | Halt proteolytic activity of dissociation enzymes to prevent cell damage | [24] |
| Serum Neutralization Assay Components | Vero E6 cells, SARS-CoV-2 virus, Anti-Spike Antibody | Key reagents for virus-focused serum neutralization assays (e.g., FRNT) | [36] |
| Specialized Inhibitors | NC-002 (Ac-LLR-ek) Epoxyketone | Cell-permeable, specific inhibitor of proteasome trypsin-like sites for research | [43] |
| Novel & Alternative Enzymes | Nattokinase, Dispase, Collagenase | Food-grade or gentler alternatives to traditional dissociation enzymes | [41] [24] |
Serum-mediated and inhibitor-based neutralization methods address fundamentally different challenges in biomedical research. Serum neutralization assays are indispensable tools for immunology and virology, providing critical data on immune protection and antibody efficacy. In contrast, the neutralization of proteolytic enzymes is a routine but vital step in cell culture that directly impacts cell health, surface marker integrity, and experimental reproducibility. The protocols and data summarized herein provide a framework for selecting the appropriate neutralization strategy based on the specific experimental media and research objectives, underscoring the importance of context in methodological applications.
Cell Counting and Viability Assessment Post-Dissociation
Tissue dissociation is a critical preliminary step in biological research, enabling the isolation of individual cells from solid tissues for downstream applications such as cell culture, flow cytometry, and single-cell RNA sequencing. The effectiveness of this process directly impacts experimental outcomes, making accurate cell counting and viability assessment paramount. Within the broader context of trypsin EDTA cell dissociation protocol research, maintaining cellular integrity while achieving high dissociation efficiency presents a significant challenge. This application note provides detailed methodologies and current data to guide researchers in accurately evaluating single-cell suspensions following enzymatic dissociation, with particular emphasis on trypsin-based approaches.
Quantitative Analysis of Dissociation Outcomes
Comparative Performance of Dissociation Methods
Table 1: Comparison of Tissue Dissociation Methods and Outcomes
| Method | Cell Viability (%) | Processing Time | Tissue Utilization | Key Advantages |
|---|---|---|---|---|
| Hypersonic Levitation & Spinning (HLS) | 92.3% [44] | 15 minutes [44] | 90% [44] | Preserves rare cell populations, non-contact method [44] |
| Traditional Enzymatic Methods | ~70% (typical benchmark) [45] | 60 minutes [44] | 70% [44] | Well-established protocols, widely accessible [4] |
| Trypsin-EDTA (T/E) | Superior preservation (in organoid study) [46] | Varies by protocol | Lower cell count/mg tissue [46] | High viability for sensitive applications [46] |
| Collagenase/Hyaluronidase | Good viability with optimization [47] | Varies by protocol | Higher total cell counts [46] | Superior for stem cell population preservation [46] |
Tissue-Specific Optimization Parameters
Table 2: Optimal Enzymatic Dissociation Parameters for Different Tissues
| Tissue Type | Recommended Enzyme(s) | Optimal Incubation Time | Critical Factors for Viability |
|---|---|---|---|
| Adrenal Medullary Tumors | Multi Tissue Dissociation Kit (MTDK), Collagenase IV [47] [48] | 20 minutes [47] [48] | Incubation time is most critical factor [47] |
| Adrenocortical Tumors | Collagenase IV, MTDK [47] [48] | 20-35 minutes [47] [48] | Post-dissociation debris removal [47] |
| Pituitary Neuroendocrine Tumors (PitNETs) | MTDK, Collagenase IV [47] [48] | 7-15 minutes [47] [48] | Small tissue samples (7-10 mg) [47] |
| Thyroid Carcinomas | MTDK [47] [48] | 20-30 minutes [47] [48] | Protocol effectiveness varies significantly [47] |
| Colorectal Cancer (Organoid Generation) | Collagenase, Hyaluronidase [46] | Protocol-dependent | Higher proportions of LGR5+/CD133+ stem cells [46] |
Experimental Protocols
Standardized Trypsin-EDTA Dissociation Protocol
Reagents Required:
- Trypsin-EDTA solution (0.25%)
- Balanced salt solution without calcium and magnesium (e.g., DPBS)
- Complete growth medium with serum
- Soybean trypsin inhibitor (for serum-free applications)
Procedure: [4]
- Preparation: Pre-warm Trypsin-EDTA solution and complete growth medium to 37°C. Minimize dwell time.
- Wash Step: Aspirate spent medium from culture vessel and discard. Wash cell monolayer with 5 mL of Dulbecco's Phosphate Buffered Saline (DPBS) without calcium and magnesium. Aspirate and discard.
- Enzyme Application: Add an appropriate volume of Trypsin-EDTA solution to flask (e.g., 5 mL for a 75 cm² flask). Ensure complete coverage of cell monolayer.
- Incubation: Incubate at 37°C until cells have detached. Monitor progress carefully using an inverted microscope to avoid over-digestion. Gently tap flask to dislodge cells if necessary. Typical dissociation occurs within 5-15 minutes.
- Neutralization: Add 5-10 mL of pre-warmed complete medium to flask. Tilt flask in all directions to thoroughly rinse flask and transfer cell suspension to a 15 mL conical tube.
- Centrifugation: Centrifuge at 100 × g for 5-10 minutes.
- Resuspension: Discard supernatant and resuspend cell pellet with 2-5 mL of pre-warmed complete medium.
- Assessment: Determine viable cell density and percent viability using an automated cell counter or manual methods.
Technical Notes: [4]
- For primary tissue dissociation, mince tissue into 3-4 mm pieces before trypsin application.
- For cold trypsin incubation, incubate at 4°C for 6-18 hours followed by 37°C for 20-30 minutes.
- Use soybean trypsin inhibitor when using serum-free media to halt trypsin activity.
Cell Viability Assessment Methods
Table 3: Cell Viability Assessment Methods Post-Dissociation
| Method | Principle | Procedure | Advantages/Limitations |
|---|---|---|---|
| Trypan Blue Exclusion | Membrane-impermeable dye stains dead cells blue [45] | Mix cell suspension with Trypan Blue; count under microscope [45] | Simple, cost-effective; stains debris compromising quantification [45] |
| Propidium Iodide (PI) | Membrane-impermeable DNA dye emits red fluorescence [45] | Incubate cells with PI; analyze via flow cytometry [45] | Quantitative; requires flow cytometry equipment [45] |
| AO/PI Staining | Acridine Orange (AO) stains all cells; PI stains dead cells [45] | Simultaneous staining with AO and PI; fluorescence analysis [45] | Distinguishes live/dead populations accurately [45] |
| SYTO9/PI Combination | SYTO9 stains all cells green; PI stains dead cells red [45] | Dual staining; analyze via fluorescence microscopy or flow cytometry [45] | Clear differentiation; compatible with multiple platforms [45] |
Post-Dissociation Quality Control Workflow
Figure 1: Comprehensive workflow for post-dissociation quality control and viability assessment.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Essential Reagents for Cell Dissociation and Viability Assessment
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Proteolytic Enzymes | Trypsin-EDTA, TrypLE Express [4] | Digests cell-adhesion proteins; primary workhorse for dissociation [4] |
| Collagenases | Collagenase I, Collagenase IV [47] [48] | Degrades collagen in extracellular matrix; essential for tough tissues [47] |
| Specialized Enzymes | Hyaluronidase, Dispase [46] [47] | Targets specific ECM components; preserves sensitive cell types [46] |
| Commercial Kits | Multi Tissue Dissociation Kit (MTDK) [47] [48] | Optimized enzyme cocktails for specific tissue types [47] |
| Viability Stains | Trypan Blue, Propidium Iodide, 7-AAD [47] [45] | Distinguishes live/dead cells; critical for quality assessment [45] |
| Nucleic Acid Stains | Acridine Orange, SYTO9 [45] | Labels DNA/RNA; identifies viable cells and cell cycle stages [45] |
| Inhibitors & Buffers | Soybean Trypsin Inhibitor [4] | Stops enzymatic activity; preserves cell surface markers [4] |
Advanced Techniques and Emerging Technologies
Automated Tissue Dissociation Systems
Recent advancements in dissociation technology include automated systems that significantly improve reproducibility and cell viability. The PythoN i tissue dissociation machine demonstrates the capability to dissociate most tissues within 15 minutes while maintaining 90% cell viability, substantially improving upon the typical 70% viability benchmark achieved with manual methods [45]. Similarly, Hypersonic Levitation and Spinning (HLS) technology represents a revolutionary non-contact approach that enables "press-and-rotate" operations within a confined flow field, generating microscale "liquid jets" that exert precise hydrodynamic forces [44]. This method achieves 92.3% cell viability while preserving rare cell populations often lost in traditional dissociation protocols [44].
Troubleshooting Common Issues
Cell Clumping: Insufficient dissociation leads to cell clumps that create significant challenges for single-cell applications. Visually inspect suspension using brightfield or confocal microscopy. To minimize clumping, filter through appropriate mesh sizes and ensure accurate cell counts to prevent overloading during downstream processing [45].
Stress-Related Artifacts: Tissue dissociation induces stress responses that can confound experimental results by altering transcriptional profiles. Cells may express heat shock proteins or display artificial activation states not present in vivo [45]. Minimize time between sample extraction and processing, and consider identifying stress marker genes for screening in downstream applications [45].
Accurate cell counting and viability assessment following dissociation are fundamental to obtaining reliable research data, particularly within trypsin EDTA-based research workflows. The protocols and data presented herein provide researchers with current methodologies to optimize these critical steps. As dissociation technologies continue to evolve, particularly through automation and non-contact methods, researchers must adapt their quality control practices accordingly. By implementing rigorous post-dissociation assessment and understanding the quantitative outcomes achievable with different methods, scientists can significantly enhance the reproducibility and biological relevance of their single-cell research.
Solving Common Challenges: Optimization Strategies for Difficult Cells and Tissues
Within the broader research on trypsin EDTA cell dissociation protocols, a significant challenge persists: the trade-off between achieving complete cell detachment and maintaining high cell viability and functionality. Incomplete detachment can compromise experimental yields, while poor viability can skew research results and diminish the effectiveness of downstream applications in drug development [49]. This guide addresses the core mechanisms behind these issues—namely, enzyme-induced damage to cell surface proteins and cytoplasmic components—and provides validated, quantitative protocols to overcome them [50] [51] [29].
Core Problem Analysis
The undesirable effects of trypsin-EDTA are not merely functional but structural, impacting key cellular components essential for post-detachment integrity.
- Cell Surface Protein Damage: Trypsin cleaves peptides on the C-terminal side of lysine and arginine residues [52]. This non-specific proteolysis can destroy critical cell surface molecules. Research demonstrates that trypsin-EDTA treatment cleaves N-cadherin, a protein essential for cell-cell adhesion, thereby significantly delaying subsequent aggregate formation in suspension culture [51]. Furthermore, a comparative study on mesenchymal stem cells (MSCs) showed that trypsin treatment within 30 minutes significantly reduced the expression of crucial surface markers like CD44, CD73, CD105, CD140a, and CD201, which are vital for cell identification and function [52].
- Cytoplasmic Alterations: The impact of trypsin extends beyond the membrane. Real-time terahertz sensing studies on epithelial cells have revealed that trypsin proteolysis induces cytoplasmic modification from the very first seconds of exposure. This alteration is associated with a transfer of small solutes, such as electrolytes and metabolites, and is correlated with changes in cell volume, indicating profound and immediate cellular stress [29].
The following diagram summarizes the pathway of trypsin-induced damage and the resulting cellular outcomes.
Quantitative Data and Troubleshooting
The following table synthesizes key quantitative findings from the literature, linking specific trypsinization conditions to their observed outcomes and providing evidence-based solutions.
Table 1: Troubleshooting Guide for Trypsin-EDTA Cell Dissociation
| Observed Problem | Evidence-Based Cause | Reported Quantitative Effect | Recommended Solution |
|---|---|---|---|
| Weak Adhesion & Delayed Aggregate Formation | Trypsin-EDTA cleaves N-cadherin and integrins [50] [51] | N-cadherin loss delayed aggregate formation; 0.025% trypsin preserved α5β1 integrins and enabled ~100% cell retention under shear stress [50] [51] | Use low trypsin concentration (0.025%) and minimize incubation time [50] |
| Loss of Cell Surface Antigens | Non-specific proteolysis of surface markers (CD44, CD73, CD105, etc.) [52] | Trypsin significantly reduced multiple MSC surface antigen-positive cell populations within 30 minutes [52] | Switch to a recombinant trypsin substitute (TrypLE), which showed no significant loss of surface antigens tested in 30 min [52] |
| Poor Cell Viability & Cytoplasmic Damage | Trypsin alters cytoplasm composition, causing solute transfer and cell volume change [29] | Cytoplasmic modification detected within seconds of trypsin exposure [29] | Optimize neutralization; use Trypsin Neutralizing Solution (TNS) over DPBS alone to instantly halt trypsin activity [50] |
Optimized Experimental Protocols
Protocol for Gentle Cell Detachment for Strong Post-Seeding Adhesion
This protocol is adapted from a study focusing on endothelial cell adhesion under shear stress, ideal for experiments requiring robust subsequent cell attachment, such as in vascular graft seeding or tissue engineering [50].
Workflow Overview:
Detailed Methodology:
- Preparation: Pre-warm Dulbecco's Phosphate Buffered Saline (DPBS) without calcium and magnesium, 0.025% Trypsin-EDTA solution, and Trypsin Neutralizing Solution (TNS) to 37°C.
- Rinsing: Aspirate the culture medium from the adherent cells and gently rinse the cell layer with DPBS to remove any residual serum that would inhibit trypsin.
- Trypsinization: Add enough pre-warmed 0.025% Trypsin-EDTA solution to cover the cell monolayer.
- Incubation: Incubate the cells at 37°C for approximately 5 minutes. Visually monitor the cells under a microscope. The cells should detach and become rounded. Avoid prolonged incubation.
- Neutralization: Promptly add a volume of pre-warmed TNS that is equal to the volume of trypsin-EDTA used. Gently pipette the solution over the cell layer to ensure complete neutralization and to aid in dislodging all cells.
- Cell Collection: Transfer the cell suspension to a centrifuge tube. Centrifuge at a recommended speed (e.g., 300-400 x g for 5 minutes) to pellet the cells.
- Resuspension: Aspirate the supernatant and gently resuspend the cell pellet in an appropriate volume of fresh, pre-warmed complete culture medium or buffer for downstream applications.
Protocol for Preserving Surface Marker Integrity
Based on a comparative study of cell-detaching methods, this protocol is critical for flow cytometry analysis or when maintaining the identity of MSCs and other sensitive cell types is paramount [52].
Detailed Methodology:
- Reagent Selection: Use a recombinant fungal-derived trypsin substitute, such as TrypLE, which is designed to have trypsin-like activity but with reduced non-specific proteolysis.
- Detachment: Aspirate culture medium, rinse with DPBS, and add pre-warmed TrypLE to cover the cell monolayer.
- Timed Incubation: Incubate at 37°C for no longer than 30 minutes. The study showed that TrypLE effectively dissociated cells within 5 minutes without altering the expression of key surface antigens (CD44, CD73, CD105, etc.) within this 30-minute window [52].
- Neutralization and Collection: The trypsin-like activity of TrypLE can be neutralized with serum-containing medium or specific inhibitors. Collect the cell suspension by gentle pipetting and proceed with centrifugation and resuspension as required.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Optimized Cell Dissociation
| Reagent / Solution | Function / Rationale | Key Feature |
|---|---|---|
| Recombinant Trypsin-EDTA (0.025%) | Low-concentration enzymatic dissociation. | Minimizes cleavage of adhesion proteins & surface markers [49] [50]. |
| TrypLE Select | Animal-origin-free, recombinant trypsin substitute. | Preserves surface antigen expression; ideal for MSC work & clinical applications [52]. |
| Trypsin Neutralizing Solution (TNS) | Instantly inactivates trypsin post-detachment. | More effective than dilution alone; prevents ongoing proteolytic damage [50]. |
| DPBS (without Ca2+/Mg2+) | Rinsing solution to remove serum & divalent cations. | Chelates calcium/magnesium with EDTA, weakening cell adhesion [49] [50]. |
Achieving successful cell dissociation requires a deliberate balance between efficiency and cellular integrity. The protocols and data presented herein, centered on the principles of using milder enzymatic conditions and advanced reagents like recombinant trypsin or TrypLE, provide a robust framework for researchers to overcome the persistent challenges of incomplete detachment and poor viability. By integrating these optimized methods, scientists can enhance the reliability of their data and the efficacy of downstream applications in drug development and regenerative medicine.
Optimizing Incubation Time and Enzyme Concentration for Specific Cell Types
Within cell culture laboratories, the dissociation of adherent cells is a foundational step for subculturing, downstream analysis, and cell-based production. The trypsin-EDTA cell dissociation protocol is a cornerstone technique for this purpose. Its effectiveness, however, is not universal; suboptimal application can lead to poor cell viability, reduced yield, and altered cell phenotypes, directly impacting experimental reproducibility and manufacturing efficiency in drug development [38]. This application note provides a structured framework for researchers and scientists to optimize the two most critical variable parameters in this protocol: incubation time and enzyme concentration, tailored to specific cell types.
The core mechanism of trypsin-EDTA action is well-established. Trypsin, a proteolytic enzyme, cleaves peptide bonds on cell surface proteins that facilitate attachment to the culture substrate and neighboring cells. EDTA, a chelating agent, complements this action by binding calcium and magnesium ions, which are essential for the integrity of cell-to-cell junctions [53]. The standard formulation used across many laboratories is a 0.25% trypsin solution with 1 mM EDTA, often prepared in a balanced salt solution like Hank's Balanced Salt Solution (HBSS) to maintain osmotic balance and pH during the dissociation process [53]. While this provides a reliable starting point, a one-size-fits-all approach overlooks the intrinsic biological diversity of different cell lines and primary tissues.
The imperative for optimization stems from the significant trade-offs involved. Excessive incubation time or overly high enzyme concentration risks damaging cell surface proteins, reducing viability, and triggering stress responses [38]. Conversely, insufficient dissociation leads to low cell yield and the potential selection for weakly adherent cell subpopulations, compromising experimental integrity. Furthermore, as research moves towards more complex systems like microcarrier-based bioreactors for vaccine production or cell therapy, precise control over dissociation parameters becomes crucial for scalability and cost-effectiveness [16]. This document outlines a systematic, data-driven approach to achieving this precision.
Key Optimization Parameters and Reagent Toolkit
Successful cell dissociation requires a balance between efficiency and cell health. The following parameters must be carefully considered and controlled during protocol development.
Critical Optimization Variables
- Enzyme Concentration: Typically ranges from 0.05% to 0.25% trypsin [8]. Higher concentrations accelerate dissociation but increase the risk of damaging critical cell surface receptors and integrins.
- Incubation Time: The duration of enzyme exposure is cell-type-dependent and must be determined empirically. Prolonged exposure is a primary cause of reduced cell viability and proteolytic damage [38].
- Temperature: Most protocols are performed at room temperature or 37°C. Higher temperatures increase enzymatic activity, reducing required incubation time but demanding more precise monitoring to prevent over-digestion [11].
- Agitation: In microcarrier cultures, a defined agitation speed is critical. An optimal speed (e.g., 1.25 x Njs, the minimum speed to keep microcarriers suspended) ensures uniform enzyme contact without subjecting cells to damaging shear forces [16].
- Cell Confluence: Passaging cells during the log phase of growth, typically at 70-90% confluence, ensures optimal recovery and consistency. Over-confluent cultures can be more resistant to dissociation.
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and their roles in the cell dissociation workflow.
Table 1: Essential Reagents for Cell Dissociation Protocols
| Reagent | Function & Rationale |
|---|---|
| Trypsin-EDTA Solution | Proteolytic enzyme (trypsin) cleaves adhesion proteins; chelating agent (EDTA) binds Ca²⁺/Mg²⁺ ions, disrupting cell junctions. Standard concentrations are 0.05% and 0.25% [53] [8]. |
| Balanced Salt Solution (e.g., HBSS, PBS) | Used for rinsing cells pre-digestion to remove residual media containing serum (which inhibits trypsin) and divalent cations [53] [11]. Provides a stable ionic and osmotic environment. |
| Trypsin Neutralization Solution | Typically serum-containing medium (e.g., FBS) or specialized serum-free solutions. Critical for halting trypsin activity immediately after detachment to prevent cell damage [53] [11]. |
| Alternative Enzymes (e.g., Accutase, Liberase) | Used for sensitive cells or specific tissues. Accutase is a blend of proteases and collagenases considered milder, but can still cleave specific proteins like FasL [38]. Liberase is used for complex tissues like adipose for MSC isolation [54]. |
| Viability Stain (e.g., Trypan Blue) | Allows for the quantification of cell viability and total cell yield post-dissociation, which are key metrics for protocol optimization [11]. |
Established Protocols and Methodologies
A robust dissociation protocol begins with a standardized baseline method, which can then be systematically altered for optimization.
Standard Trypsin-EDTA Protocol for Adherent Cells
This protocol is adapted from established mammalian cell culture methods and serves as a reference point [11].
- Preparation: Pre-warm trypsin-EDTA solution, complete growth medium, and balanced salt solution (without Ca²⁺/Mg²⁺) to 37°C or room temperature. Ensure all equipment is sterile.
- Aspiration: Remove and discard the spent cell culture media from the culture vessel.
- Rinse: Gently wash the cell monolayer with a balanced salt solution (e.g., 2 mL per 10 cm² surface area) to remove all traces of serum and divalent cations. Thorough removal is essential for efficient trypsin activity.
- Trypsinization: Add pre-warmed trypsin-EDTA solution to the side of the vessel (e.g., 0.5 mL per 10 cm²). Gently rock the vessel to ensure complete coverage of the cell layer.
- Incubation: Incubate the vessel at the recommended temperature (e.g., room temperature for ~2 minutes). The actual time is cell line-dependent and must be determined visually.
- Monitoring: Observe cells under a microscope every 30-60 seconds. Cells will retract and round up. Gently tap the vessel to aid detachment. The process is complete when ≥90% of cells are detached.
- Neutralization: When cells are detached, add a volume of complete growth medium that is at least double the volume of trypsin used. Pipette the medium over the surface to ensure complete neutralization of the enzyme.
- Centrifugation: Transfer the cell suspension to a centrifuge tube and pellet cells at approximately 200 x g for 5-10 minutes.
- Resuspension and Seeding: Resuspend the cell pellet in fresh pre-warmed growth medium. Perform a cell count and viability assessment, then seed the cells into new culture vessels at the recommended density.
The workflow for this standard protocol is summarized in the diagram below.
Diagram 1: Standard trypsin-EDTA workflow
Quantitative Optimization Data from Recent Studies
Recent studies provide quantitative data on optimizing these parameters for specific applications, moving beyond standard cell lines.
Table 2: Experimentally-Determined Optimal Parameters from Recent Studies
| Cell Type / System | Objective | Key Optimized Parameters & Results | Citation |
|---|---|---|---|
| MA 104 on Cytodex 1 MCs | Scale-down in situ detachment for bioreactor seed train. | Trypsin Volume: Reduced to 50 mL/g of MCs.Agitation: 1.25 x Njs (just-suspended speed).Time: Up to 60 min monitored via capacitance.Outcome: Maintained activity >380 USP/mL, complete dissociation. | [16] |
| Bovine Adipose Tissue (MSCs) | Maximize cell isolation yield for cultured meat. | Enzyme: Liberase 0.1% for 3 hours.Comparison: Significantly higher yield vs. Collagenase I 0.1% (3h).Outcome: Yield of 30.48-67.1 x 10⁶ cells/g tissue. | [54] |
| Murine Macrophages (RAW264.7) | Preserve surface protein integrity (FasL/FasR). | Method Tested: Accutase (10-30 min).Result: Significant decrease in FasL/FasR vs. EDTA-based methods.Recovery: 20h required for surface marker recovery. | [38] |
Optimization Strategies and Experimental Design
To tailor the dissociation protocol for a specific cell type, a systematic experimental approach is required.
Designing an Optimization Experiment
A robust optimization study should test a matrix of conditions. The following workflow outlines a generalized strategy for determining the optimal incubation time and enzyme concentration for a new or sensitive cell type.
Diagram 2: Optimization experiment design
Advanced Monitoring and Alternative Methods
- Real-time Process Monitoring: In microcarrier cultures, capacitance sensors have been successfully used as a Process Analytical Technology (PAT) tool to monitor cell detachment in real-time without offline sampling. The drop in capacitance signal correlates directly with cell detachment, allowing for precise process control and endpoint determination [16].
- Considering Alternative Enzymes: While trypsin-EDTA is ubiquitous, it is not always the best choice. For cells with critical surface markers or those derived from complex tissues, alternatives should be considered.
- Accutase: Often marketed as a milder enzyme, it is effective for detaching sensitive cells like stem cells and neurons with high viability. However, it can cleave specific surface proteins like FasL and Fas receptor, requiring a ~20-hour recovery period for re-expression [38].
- Enzyme Blends: For primary tissue dissociation (e.g., adipose tissue for MSC isolation), blends like Liberase or Collagenase/Trypsin mixtures can yield superior results by targeting multiple components of the extracellular matrix [54].
- Non-Enzymatic Methods: Versene (EDTA-based solution) or physical scraping are the least disruptive to surface proteins but are often only suitable for weakly adherent cells or when protein integrity is the sole priority [38].
Optimizing trypsin-EDTA incubation time and enzyme concentration is not a mere procedural detail but a critical factor in ensuring cellular fidelity and experimental success. The data and protocols presented herein demonstrate that a standardized, one-size-fits-all approach is insufficient for rigorous scientific research and bioprocess development. The optimal dissociation protocol is a balance achieved through systematic testing of key variables—primarily enzyme concentration, incubation time, and physical parameters—against the critical metrics of cell yield, viability, and functional integrity.
As the field advances, several key trends are emerging. There is a growing emphasis on high-purity, chemically defined formulations to meet regulatory requirements for therapeutic manufacturing [8]. The integration of advanced monitoring techniques, such as capacitance sensors in bioreactors, provides a path toward real-time, data-driven process control, moving beyond static timed incubations [16]. Furthermore, the exploration of specialized enzyme blends for complex tissues and primary cells highlights a move towards precision dissociation [54]. By adopting the systematic, quantitative optimization framework outlined in this application note, researchers and drug development professionals can enhance the reproducibility, efficiency, and quality of their cell-based work, directly contributing to the advancement of robust and reliable biological science.
Cell dissociation using trypsin-EDTA is a cornerstone technique in cell culture, enabling the detachment of adherent cells for subculturing or experimental analysis. However, this proteolytic process inherently compromises cell surface proteins, potentially altering cellular phenotypes and functions. Understanding the recovery timeline of these proteins post-digestion is critical for designing robust experiments, particularly in drug development, flow cytometry, and cell-based therapies. This application note synthesizes recent findings on trypsin-EDTA-induced surface protein damage, provides protocols to quantify recovery, and outlines strategies to minimize experimental artifacts.
Quantitative Impact of Trypsin-EDTA on Surface Proteins
Studies demonstrate that trypsin-EDTA cleaves specific surface proteins, necessitating a recovery period for reexpression. Key quantitative findings are summarized below:
Table 1: Surface Protein Damage and Recovery Post-Trypsin-EDTA Treatment
| Protein Affected | Cell Type | Reduction in Expression Post-Detachment | Time to Full Recovery | Assessment Method |
|---|---|---|---|---|
| Fas Ligand (FasL) | Murine macrophages (RAW264.7) | Significant decrease vs. EDTA-based methods [38] | 20 hours [38] | Flow cytometry (MFI) |
| Fas Receptor (Fas) | Murine macrophages (RAW264.7) | Significant decrease vs. EDTA-based methods [38] | 20 hours [38] | Flow cytometry (MFI) |
| F4/80 | Murine macrophages (RAW264.7) | No significant change [38] | Not applicable | Flow cytometry (MFI) |
| LGR5+/CD133+ stem cells | Colorectal cancer organoids | Lower yield vs. collagenase/hyaluronidase [55] | Protocol-dependent | Flow cytometry [55] |
- Mechanism of Damage: Trypsin cleaves peptide bonds after lysine or arginine residues, degrading extracellular domains of proteins like FasL. EDTA chelates calcium, further disrupting cell-adhesion interactions [38] [4].
- Recovery Dynamics: Surface protein reexpression requires de novo protein synthesis and membrane trafficking, with full restoration of FasL/Fas taking ~20 hours post-trypsin-EDTA treatment [38].
Experimental Protocols for Assessing Protein Recovery
Protocol: Flow Cytometry-Based Recovery Assessment
Objective: Quantify surface protein reexpression over time post-detachment. Materials:
- Trypsin-EDTA solution (e.g., 0.05%–0.25%) [56]
- Cell Dissociation Buffer (enzyme-free; for controls) [4]
- Complete growth medium
- Antibodies for target proteins (e.g., anti-FasL)
- Flow cytometer
Steps:
- Detach Cells:
Seed for Recovery:
Stain and Analyze:
- Detach control and test cells using gentle, enzyme-free methods (e.g., EDTA-based buffer) to avoid retrypsinization [38].
- Stain with fluorophore-conjugated antibodies and analyze via flow cytometry. Compare mean fluorescence intensity (MFI) to untreated controls.
Protocol: Western Blot Validation of Protein Cleavage
Objective: Confirm proteolytic cleavage of surface proteins (e.g., FasL). Steps:
- Treat cells with trypsin-EDTA or EDTA-only buffer (control) for 10–30 minutes [38].
- Collect supernatants and cell lysates.
- Detect fragments using antibodies targeting extracellular domains (e.g., ~20 kDa FasL fragments in supernatants post-trypsin) [38].
Workflow Visualization
Diagram 1: Experimental Workflow for Protein Recovery Studies
Title: Protein Recovery Workflow
Diagram 2: Surface Protein Recovery Timeline
Title: Recovery Timeline Post-Trypsin
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Reagents for Cell Dissociation and Recovery Studies
| Reagent/Material | Function | Example Use-Case |
|---|---|---|
| Trypsin-EDTA Solution | Proteolytic enzyme + chelator for cell detachment | General subculturing; not recommended for surface protein studies post-digestion [56] [4] |
| Recombinant Trypsin (e.g., TrypsinNex) | Animal origin-free trypsin with consistent activity | Biopharmaceutical manufacturing; reduces batch variability [57] |
| Enzyme-Free Dissociation Buffer | Gentle detachment via Ca2+/Mg2+ chelation | Preserving surface proteins for immediate flow cytometry [38] [4] |
| TrypLE Express | Recombinant fungal-derived protease | Alternative to trypsin; milder activity [55] [4] |
| Collagenase/Hyaluronidase | Targets ECM components (e.g., collagen, hyaluronic acid) | Preserving stem cell markers (LGR5+/CD133+) in organoid generation [55] |
| Soybean Trypsin Inhibitor | Inactivates residual trypsin | Halting proteolysis after dissociation [56] [4] |
Best Practices for Experimental Design
- Recovery Period: Allow ≥20 hours post-trypsinization for surface protein studies [38].
- Controls: Include EDTA-detached or scraped cells as negative controls for protease effects [38].
- Enzyme Selection: For sensitive applications (e.g., stem cell isolation), use collagenase or hyaluronidase to maximize viability and stem cell yield [55].
- Validation: Always confirm protein recovery via flow cytometry or Western blot before functional assays.
By integrating these protocols and considerations, researchers can mitigate the confounding effects of trypsin-EDTA dissociation, ensuring reliable data in drug development and cellular research.
Strategies for Difficult-to-Dissociate Cells and Primary Tissues
The dissociation of complex primary tissues and difficult-to-detach cells into high-viability single-cell suspensions represents a critical bottleneck in biomedical research and therapeutic development [19]. While traditional enzymatic methods like trypsin-EDTA provide a foundation for cell dissociation, these approaches often prove inadequate for robust tissues such as tumors, connective-rich organs, and stem cell clusters, where they can compromise cell viability, alter surface markers, and reduce yield [19] [58] [59]. The limitations of conventional protocols have catalyzed the development of advanced enzymatic, non-enzymatic, and integrated strategies specifically designed to address these challenges. This application note details optimized, practical methodologies for dissociating challenging tissue types, providing researchers with validated protocols to enhance cell yield, viability, and functionality for downstream applications including single-cell analysis, cell therapy manufacturing, and drug discovery.
Core Challenges in Tissue Dissociation
Difficult-to-dissociate tissues typically present three interconnected challenges: dense extracellular matrix (ECM), robust cell-cell junctions, and the presence of delicate or rare cell populations. Enzymatic methods alone often require prolonged processing times, which can damage cell surface proteins and reduce viability [19] [4]. Mechanical methods, while faster, can induce significant mechanical stress and apoptosis [44] [59]. The optimal dissociation strategy effectively balances disruption efficiency with preservation of cellular integrity, often through combined-mechanism approaches.
Advanced Dissociation Strategies
Optimized Enzymatic and Mechanical Combination Protocol
For tissues with moderate ECM density, such as breast carcinomas and skin biopsies, an optimized chemical-mechanical workflow significantly outperforms enzymatic-only dissociation [19].
Protocol: Enzymatic-Mechanical Dissociation for Solid Tissues
- Tissue Preparation: Mince fresh tissue into 2-4 mm fragments using sterile scalpel or scissors in a petri dish cooled on ice. Wash fragments 2-3 times in cold DPBS without calcium and magnesium to remove blood and debris [4].
- Enzymatic Digestion: Immerse tissue fragments in pre-warmed enzyme solution (e.g., collagenase IV 1.5 mg/mL + dispase 2.0 U/mL + hyaluronidase 0.2 mg/mL in HBSS with calcium and magnesium). Use 5-10 mL enzyme solution per gram of tissue [19] [4].
- Incubation Conditions: Incubate at 37°C with continuous agitation for 30-45 minutes. For more robust tissues, consider a two-step digestion with collagenase followed by trypsin-EDTA (0.25%) for 15-20 minutes [4].
- Mechanical Disruption: Following enzymatic digestion, transfer the partially digested tissue to a gentle mechanical dissociation device or manually triturate using a 10 mL serological pipette. Continue until tissue fragments are no longer visible.
- Reaction Termination: Add complete media containing serum or specific enzyme inhibitors. Filter cell suspension through 70-100 μm cell strainer to remove aggregates and debris [4].
- Cell Collection: Centrifuge filtered suspension at 300-400 × g for 5 minutes. Resuspend pellet in appropriate buffer or culture medium and assess viability and cell count [4].
Enzyme-Free Electrochemical Dissociation
A novel electrochemical platform enables high-efficiency detachment of adherent cells and delicate primary tissues without enzymatic damage, ideal for cell therapy applications [58].
Protocol: Electrochemical Cell Detachment
- Surface Preparation: Utilize a conductive biocompatible polymer nanocomposite surface compatible with standard cell culture.
- Cell Culture: Culture adherent cells to 70-80% confluence on the electrochemical surface.
- Buffer Application: Replace culture medium with low-conductivity electrochemistry buffer (e.g., 10 mM HEPES in isotonic sucrose).
- Electrical Stimulation: Apply low-frequency alternating voltage (0.5-2 V, 0.1-1 Hz) for 3-5 minutes. Optimal parameters vary by cell type and should be determined empirically.
- Cell Harvesting: Gently tap vessel to dislodge detached cells. Transfer cell suspension to collection tube.
- Post-Processing: Centrifuge at 100-200 × g for 5 minutes. Resuspend in appropriate medium. This method typically achieves >90% viability and >95% detachment efficiency [58].
Hypersonic Levitation and Spinning (HLS) for Sensitive Tissues
The HLS method employs non-contact acoustic forces to dissociate tissues with minimal damage, particularly valuable for preserving rare cell populations [44].
Protocol: HLS Tissue Dissociation
- Apparatus Setup: Configure the automated tissue dissociation apparatus with triple-acoustic resonator probe and conical confinement structure.
- Tissue Loading: Place 1-5 mm tissue fragments into the dissociation chamber containing appropriate buffer or mild enzyme solution.
- Acoustic Activation: Apply GHz-frequency acoustic waves to generate hypersonic streaming, causing tissue levitation and rapid self-rotation ("press-and-rotate" operation).
- Processing Parameters: Process for 10-15 minutes at optimized frequency and amplitude. Monitor dissociation progress visually.
- Cell Collection: Automated system transfers dissociated cells through integrated filtration to collection chamber.
- Quality Assessment: Assess cell viability and yield. HLS typically achieves 92.3% viability and 90% tissue utilization in 15 minutes [44].
Quantitative Comparison of Dissociation Methods
Table 1: Performance Comparison of Advanced Dissociation Methods
| Method | Tissue Type | Viability (%) | Processing Time | Cell Yield | Key Advantages |
|---|---|---|---|---|---|
| Optimized Enzymatic-Mechanical [19] | Bovine Liver, Human Breast Cancer | >90% | 15-60 min | 37-92% (varies by tissue) | High yield for moderate tissues, readily available reagents |
| Enzyme-Free Electrochemical [58] | Osteosarcoma, Ovarian Cancer, CAR-T cells | >90% | 3-5 min | >95% detachment | No enzyme damage, ideal for sensitive applications, automated compatible |
| Hypersonic Levitation & Spinning (HLS) [44] | Human Renal Cancer, Delicate Primary Tissues | 92.3% | 15 min | 90% tissue utilization | Exceptional rare cell preservation, non-contact, integrated system |
| Traditional Enzymatic Only [19] | Various | 60-85% | 60 min to overnight | Variable, often lower | Established protocols, low equipment needs |
Table 2: Research Reagent Solutions for Tissue Dissociation
| Reagent/Category | Specific Examples | Function & Application |
|---|---|---|
| Proteolytic Enzymes | Trypsin, Collagenase, Dispase | Digest extracellular matrix proteins and cell adhesion molecules [4] |
| Chelating Agents | EDTA, EGTA | Bind calcium ions to disrupt calcium-dependent cell adhesions [4] [59] |
| Enzyme Blends | Collagenase/Dispase, Trypsin/EDTA | Comprehensive digestion for complex tissues through synergistic action [4] |
| Specialized Substrates | Conductive polymer nanocomposite surfaces | Enable electrochemical detachment without enzymatic damage [58] |
| Cell Culture Supplements | Soybean trypsin inhibitor, Serum-containing media | Terminate enzymatic activity post-digestion to preserve viability [4] |
| Separation Matrices | Akadeum microbubbles, Density gradient media | Isolate target cells post-dissociation with minimal stress [59] |
Workflow Visualization
Diagram 1: Decision workflow for tissue dissociation strategies. The pathway selection depends on tissue characteristics and research objectives.
Diagram 2: Mechanism-to-technology mapping for tissue dissociation. Different technologies target specific dissociation barriers.
Successful dissociation of difficult-to-dissociate tissues requires moving beyond one-size-fits-all enzymatic protocols toward mechanism-informed, integrated strategies. The methods detailed herein provide researchers with proven approaches for overcoming specific tissue challenges, whether dealing with dense ECM, sensitive cell types, or rare populations. As the field advances, the integration of these technologies with automated systems and standardized quality metrics will further enhance reproducibility and scalability for both research and clinical applications.
Serial Enzymatic Dissociation for Complex Tissues with Conflicting Requirements
The dissociation of complex tissues into viable single-cell suspensions is a critical foundational step in many areas of biological research and therapeutic development, including single-cell analysis, cell therapy manufacturing, and drug discovery [19]. However, researchers frequently encounter a fundamental challenge: conflicting requirements between preserving cell surface marker integrity, maintaining high viability, and achieving sufficient yield from tissues with heterogeneous cellular composition and extracellular matrix density [19] [38]. Traditional single-enzyme approaches often fail to address these competing needs adequately, potentially compromising downstream applications.
Serial enzymatic dissociation, which employs sequential application of different enzymes with specific substrate preferences, has emerged as a powerful strategy to overcome these limitations. This approach allows for the stepwise dismantling of complex tissue architecture while minimizing damage to sensitive cell populations. Within this framework, trypsin-EDTA remains a cornerstone reagent due to its well-characterized activity against protein-mediated cell adhesions [24]. This application note details optimized serial dissociation protocols that integrate trypsin-EDTA with other enzymatic and non-enzymatic reagents to address specific experimental challenges with complex tissues.
The Challenge of Conflicting Requirements in Tissue Dissociation
Complex tissues present multiple, often competing, challenges for effective dissociation. The table below summarizes the primary conflicting requirements and their experimental implications.
Table 1: Common Conflicting Requirements in Tissue Dissociation
| Conflicting Requirements | Experimental Implications | Affected Downstream Applications |
|---|---|---|
| Surface Protein Integrity vs. Dissociation Efficiency | Enzymes like trypsin efficiently dissociate tissues but cleave surface proteins [38], while gentler methods preserve epitopes but yield fewer cells [60]. | Flow cytometry, immunocytochemistry, receptor signaling studies [24] [38] |
| Viability vs. Yield | Prolonged or harsh enzymatic treatment increases cell yield but decreases viability; shorter, gentler treatments have the opposite effect [19] [48]. | Primary cell culture, cell therapy manufacturing, long-term functional assays [19] [46] |
| Stem Cell Population Recovery vs. Total Cell Yield | Harsh enzymes may compromise rare, sensitive stem cell populations, while gentler enzymes may not effectively release them from niche [46]. | Organoid generation, regenerative medicine, stem cell research [46] |
| Processing Time vs. Sample Quality | Rapid dissociation is often necessary to minimize ex vivo artifacts, but can compromise quality. Standardized automated systems can mitigate this [19] [61]. | Single-cell RNA sequencing, functional genomics, metabolic studies |
Quantitative Comparison of Dissociation Enzymes
Selecting the appropriate enzymatic toolkit requires a clear understanding of each reagent's performance characteristics. The following table synthesizes quantitative data from recent studies comparing common dissociation agents.
Table 2: Performance Metrics of Selected Tissue Dissociation Reagents
| Dissociation Reagent | Reported Cell Viability (%) | Key Performance Characteristics | Optimal Tissue Applications |
|---|---|---|---|
| Trypsin-EDTA | 93.2% (MSC) [60] | Fast-acting; can damage surface proteins [38]; requires inhibition [24]. | Continuous cell lines, strongly adherent early-passage lines [24] |
| TrypLE | >90% [46] | Gentler on surface proteins; animal-origin free; inhibited by dilution [24]. | Primary cells, cell surface protein-sensitive applications [24] [46] |
| Collagenase | High (Organoid yield) [46] | Effective on dense, collagen-rich matrices; higher stem cell population yield [24] [46]. | Colorectal cancer organoids, heart, liver, bone [24] [46] |
| Accutase | High (Maintained >90 min) [38] | Blended enzymatic activity; can cleave specific surface proteins (e.g., FasL) [38]. | Strongly adherent cells (e.g., macrophages); requires recovery time for surface markers [38] |
| Enzyme-Free Buffer | 68.7% (MSC) [60] | Preserves surface protein integrity; lower viability and reattachment rates [60] [38]. | Weakly adherent cells (e.g., epithelial cells), flow cytometry [24] |
Recommended Serial Dissociation Protocols
Protocol 1: For Tissues Requiring High Surface Protein Integrity
This protocol is designed for applications like flow cytometry or receptor studies where preserving cell surface markers is paramount, using TrypLE as a gentler alternative to trypsin.
A. Reagents and Materials
- Wash Buffer: HBSS or PBS without calcium and magnesium [24]
- Primary Dissociation Reagent: TrypLE Express Enzyme (1X) [24] [46]
- Secondary Dissociation Reagent: Cell Dissociation Buffer (PBS-based, enzyme-free) [24]
- Inactivation Medium: Culture medium containing serum or defined trypsin inhibitor [24]
- Equipment: Water bath (37°C), centrifuge, sterile pipettes
B. Step-by-Step Procedure
- Tissue Preparation: Mince fresh tissue into 1-2 mm³ fragments using sterile scalpels in a chilled petri dish containing wash buffer [61] [48].
- First Dissociation (TrypLE):
- Transfer tissue fragments to a tube containing pre-warmed TrypLE Express (5-10 mL per gram of tissue).
- Incubate at 37°C for 7-15 minutes with gentle agitation or pipetting every 5 minutes [48].
- Monitor dissociation visually. When the majority of fragments are dispersed, proceed.
- Add an equal volume of cold inactivation medium to stop enzymatic activity.
- Centrifuge at 300-400 × g for 5 minutes and collect supernatant containing released cells.
- Second Dissociation (Enzyme-Free Buffer):
- Post-Processing:
- Filter the combined cell suspension through a 70 μm cell strainer.
- Centrifuge and resuspend in appropriate buffer for counting and viability assessment [48].
Protocol 2: For Fibrous and Collagen-Rich Tissues
This protocol is optimized for tissues with dense extracellular matrix, such as tumors or certain organs, where collagenase is essential for effective dissociation.
A. Reagents and Materials
- Wash Buffer: HBSS without calcium and magnesium
- Primary Dissociation Reagent: Collagenase Type I, II, or IV (200 units/mL in balanced salt solution) [24] [46]
- Secondary Dissociation Reagent: Trypsin-EDTA (0.25%) or TrypLE Express [24] [46]
- DNase I (optional): To prevent cell clumping from released DNA [48]
- Inactivation Medium: Culture medium with serum
B. Step-by-Step Procedure
- Tissue Preparation: Mince tissue as in Protocol 1. For very dense tissues, initial mechanical disruption using a Medimachine system can be considered [61].
- First Dissociation (Collagenase):
- Incubate tissue fragments with pre-warmed collagenase solution (Type I for epithelial/adrenal tissue; Type II for heart/liver; Type IV for islets) [24] [48] for 20-35 minutes at 37°C with agitation [48].
- After initial incubation, gently pipette the mixture and allow large fragments to settle. Collect the supernatant containing released cells.
- Inactivate collagenase with cold complete medium.
- Second Dissociation (Trypsin-EDTA/TrypLE):
- Post-Processing:
- Combine all cell suspensions and filter through a 70-100 μm strainer.
- Perform debris removal and red blood cell lysis if necessary [48].
- Centrifuge, resuspend, and count cells.
Critical Factors for Success
- Tissue Specificity: Optimal dissociation time varies significantly by tissue type. Adrenal medullary tumors show best viability at 20 minutes, while pituitary neuroendocrine tumors (PitNETs) require only 7-15 minutes [48].
- Enzyme Selection: Collagenase has demonstrated superior performance in generating patient-derived organoids from colorectal cancer tissues, yielding higher counts of LGR5+ and CD133+ stem cells [46].
- Recovery Time: Cells dissociated with accutase require up to 20 hours for full recovery of surface proteins like FasL [38]. Plan downstream assays accordingly.
- Post-Dissociation Processing: Debris removal kits and red blood cell lysis steps can significantly improve sample quality and viability in certain tumor types [48].
The Scientist's Toolkit: Essential Reagents and Equipment
Table 3: Key Research Reagent Solutions for Serial Enzymatic Dissociation
| Reagent/Equipment | Function | Key Considerations |
|---|---|---|
| Trypsin-EDTA | Proteolytic enzyme that cleaves peptide bonds; EDTA chelates calcium to weaken cell adhesions [24]. | Concentration (0.05%-0.25%) and incubation time must be optimized; requires inhibition post-dissociation [24] [38]. |
| TrypLE Express | Recombinant fungal enzyme with trypsin-like activity [24]. | Gentler on cells; animal-origin free; stable at room temperature; inhibited by dilution [24] [46]. |
| Collagenase (Types I-IV) | Degrades native collagen in connective tissues [24]. | Type selection is tissue-dependent (e.g., Type I for epithelial, Type II for heart/liver) [24] [48]. |
| Cell Dissociation Buffer | Enzyme-free, chelating buffer for gentle dissociation [24] [60]. | Ideal for weakly adherent cells; preserves surface protein integrity but may yield lower viability [24] [60]. |
| DNase I | Degrades DNA released from lysed cells to prevent clumping [48]. | Particularly useful for tissues prone to high levels of cell death during dissociation [48]. |
| Medimachine System | Automated mechanical disaggregation system [61]. | Standardizes mechanical dissociation; minimizes operator-dependent variability [61]. |
Workflow and Decision Pathway
The following diagram illustrates the logical decision process for selecting an appropriate serial dissociation strategy based on tissue characteristics and experimental goals.
Serial enzymatic dissociation represents a sophisticated approach to addressing the inherent challenges of working with complex biological tissues. By strategically combining trypsin-EDTA with complementary enzymes like collagenase or gentler alternatives like TrypLE, researchers can effectively balance the frequently conflicting requirements of cell viability, surface marker integrity, and dissociation efficiency. The protocols outlined here provide a framework that can be further optimized for specific tissue types and applications, ultimately enhancing the reliability and quality of downstream analyses in drug development and basic research.
Trypsinization, the process of using the proteolytic enzyme trypsin to dissociate adherent cells from culture surfaces, is a foundational technique in cell biology and proteomics [62]. However, manual methods are prone to human error, leading to inconsistencies in cell viability, yield, and experimental results [62]. The automation of this process represents a significant advancement, enhancing reproducibility, increasing throughput, and minimizing contamination risks [62]. This application note details the critical protocols and quantitative benefits of automating trypsinization, providing researchers and drug development professionals with actionable methodologies to integrate into their workflows, framed within the broader context of trypsin-EDTA cell dissociation research.
Quantitative Benefits of Automated Trypsinization
Automation directly addresses the major limitations of manual trypsinization. The following table summarizes the key advantages supported by experimental data.
Table 1: Documented Benefits of Automating Trypsinization Workflows
| Benefit | Experimental Finding | Significance for Research & Drug Development |
|---|---|---|
| Enhanced Reproducibility | An automated workflow for proteomic sample prep reduced technical variation, achieving intra- and inter-day coefficient of variation (CV) below 20% for peptide quantification [63]. | Essential for reliable biomarker verification and high-quality, consistent data in large-scale studies. |
| Increased Throughput | Automated liquid handlers can process 96 samples in approximately 5 hours, including a 2-hour trypsin incubation [63]. | Enables rapid processing of large sample cohorts, accelerating discovery and validation pipelines. |
| Improved Cell Viability & Yield | Automated tissue dissociators (e.g., STEMprep) standardize mechanical and enzymatic dissociation, generating high-yield, high-viability single-cell suspensions from various tissues [64]. | Critical for downstream applications like single-cell sequencing and flow cytometry, where cell integrity is paramount. |
| Optimized Digestion Efficiency | For proteomics, reducing trypsin digestion from overnight to 1 hour at 37°C showed no negative impact on protein identification rates for Cryptococcus neoformans [65]. | Streamlines sample preparation without sacrificing proteomic coverage, improving workflow efficiency. |
Detailed Automated Protocols
Protocol 1: Automated Liquid Handling for Cell Culture Trypsinization
This protocol is designed for automated liquid handling systems, such as the Opentrons Flex, to standardize the passaging of adherent cell cultures [62].
Key Reagent Solutions:
- Trypsin-EDTA Solution: Typically 0.05% to 0.25% trypsin. The EDTA chelates calcium, disrupting cell-to-substrate attachments [62].
- Neutralization Buffer: Complete growth medium containing serum or specialized stop solutions (e.g., based on PBS for TrypLE) [4].
- DPBS without Calcium and Magnesium: Used for washing cells prior to trypsin addition.
Methodology:
- Aspiration and Wash: The automated system aspirates and discards the spent cell culture media. It then dispenses a pre-warmed, balanced salt solution (e.g., DPBS) to wash the cell monolayer and removes the wash solution [4].
- Trypsin Addition: The system adds a predefined volume of trypsin-EDTA or alternative dissociation enzyme (e.g., TrypLE Express) to ensure complete coverage of the cell layer [62] [4].
- Incubation and Monitoring: The culture vessel is incubated at 37°C for a predetermined time (typically 3-15 minutes). The system can monitor for cell detachment, often by integrating with microscope imaging or by detecting changes in fluid resistance [62].
- Neutralization: Once detachment is confirmed, the system adds a neutralization buffer to halt tryptic activity [62].
- Collection and Processing: The cell suspension is transferred to a collection tube or plate. Further steps, such as centrifugation, cell counting, and reseeding, can be incorporated into the automated workflow [62].
Protocol 2: Automated Proteomic Sample Preparation for Mass Spectrometry
This protocol, adapted from a high-throughput SRM-MS workflow, utilizes a Biomek NXP workstation for highly reproducible protein digestion [63].
Key Reagent Solutions:
- Digestion Buffer: Typically containing surfactants or chaotropic agents for denaturation.
- Reducing Reagent: Dithiothreitol (DTT) to break disulfide bonds.
- Alkylating Reagent: Iodoacetamide (IAA) to prevent reformation of disulfides.
- Sequencing-grade Trypsin: Enzyme for specific protein cleavage.
- Quenching Solution: 10% Formic Acid (FA) to stop the digestion reaction.
Methodology:
- Sample and Reagent Addition: Samples (e.g., 5 µL plasma) are loaded into a 96-well plate. The workstation then sequentially adds digestion buffer, denaturant, internal standards, and reducing reagent [63].
- Reduction and Alkylation: The plate is sealed and incubated with shaking (1000 RPM) at 60°C for 60 minutes for reduction. Methyl methanethiosulfonate (MMTS) is then added as an alkylating agent and shaken for 10 minutes [63].
- Automated Trypsin Digestion: Trypsin in 0.1% formic acid is added. The plate is incubated with shaking at 1000 RPM for 2 hours at 43°C [63].
- Reaction Quenching: The digestion is stopped by adding 10% formic acid. The plate is centrifuged, and the supernatant containing the peptides is prepared for LC-MS/MS analysis [63].
Protocol 3: Automated Tissue Dissociation for Primary Cells
This protocol employs systems like the STEMprep Tissue Dissociator to process solid tissues into single-cell suspensions [64].
Key Reagent Solutions:
- Optimized Enzymatic Cocktail: Kits containing a blend of collagenases, proteases, and other enzymes tailored for specific tissue types.
- Dissociation Buffer: A balanced salt solution to maintain osmolarity and cell health during the process.
Methodology:
- Tissue Preparation: The tissue sample is minced into 3-4 mm pieces and placed into a single-use STEMprep sample tube [64].
- Reagent Addition: A predefined volume of the enzymatic cocktail and buffer is added to the tube.
- Automated Dissociation: The tube is loaded into the instrument, which runs a validated protocol combining controlled mechanical agitation (via an integrated blade and rotor) and enzymatic digestion at a controlled temperature (4-37°C) [64].
- Collection: The resulting single-cell suspension is filtered and collected, ready for downstream applications like cell isolation or sequencing [64].
The Scientist's Toolkit: Essential Reagents for Trypsinization
Table 2: Key Research Reagent Solutions for Trypsinization Protocols
| Reagent | Function & Mechanism | Application Notes |
|---|---|---|
| Trypsin-EDTA | Trypsin cleaves peptide bonds; EDTA chelates calcium to disrupt cell adhesions [62]. | Standard for most adherent cell lines. Concentration (0.05-0.25%) and exposure time must be optimized to avoid cytotoxicity [29]. |
| TrypLE Express | A recombinant fungal-derived enzyme that mimics trypsin activity [4]. | Animal-origin free, gentler on cells, and does not require serum for inactivation. A direct substitute in most trypsin protocols [66] [4]. |
| Collagenase | Degrades native collagen, a key component of the extracellular matrix in tissues [4]. | Essential for dissociating primary tissues, often used in combination with other enzymes [4]. |
| Dispase | A neutral protease that cleaves fibronectin and collagen IV [4]. | Used for detaching epithelial cells as intact sheets; effective in tissue dissociation protocols [66] [4]. |
| Cell Dissociation Buffer | A non-enzymatic, salt-based solution that chelates cations to disrupt cell-surface protein interactions [4]. | Ideal for preserving sensitive cell surface markers (e.g., for flow cytometry); not recommended for strongly adherent cells [4]. |
Workflow Visualization
The following diagram illustrates the logical workflow for selecting and executing an automated trypsinization strategy, integrating the protocols and reagents described above.
Automated Trypsinization Workflow Selection
The automation of trypsinization is a critical enhancement for modern laboratories, directly addressing the needs for high reproducibility, scalability, and efficiency in both cell culture and proteomic applications. By implementing the detailed protocols and leveraging the specialized reagents outlined in this document, researchers can significantly improve the quality and reliability of their data. As the field advances, the integration of automated platforms with real-time monitoring and adaptive control will further solidify trypsinization as a robust, standardized, and indispensable component of biomedical research and drug development.
Preventing Contamination and Maintaining Sterility Throughout the Process
Within the broader context of research on trypsin-EDTA cell dissociation protocols, maintaining sterility is a foundational pillar for experimental reproducibility and data integrity. Cell culture contamination represents one of the most frequent setbacks in laboratory environments, potentially compromising years of research and leading to misleading scientific conclusions [67]. For researchers and drug development professionals working with cell dissociation procedures, the enzymatic nature of trypsin-EDTA treatment introduces specific vulnerabilities to contamination that require specialized handling protocols. These contaminants can include biological agents such as bacteria, molds, yeasts, viruses, and mycoplasma, as well as chemical impurities and cross-contamination by other cell lines [67]. This application note provides detailed methodologies for preventing, identifying, and addressing contamination risks specific to trypsin-EDTA cell dissociation workflows, ensuring the reliability of downstream applications in biomedical research and therapeutic development.
Background and Significance
The trypsin-EDTA cell dissociation process inherently creates contamination vulnerabilities through multiple mechanisms. Trypsin, a proteolytic enzyme, cleaves peptide bonds at the C-terminal side of lysine and arginine residues, breaking down vessel-adhering proteins that maintain cellular integrity [24]. EDTA enhances this process by chelating calcium and magnesium ions, weakening cell-cell adhesion and improving trypsin's access to peptide bonds [24]. This enzymatic disruption of cellular attachments simultaneously compromises natural barriers against microbial invasion, while the multiple vessel transfers and reagent additions required in standard protocols increase exposure opportunities for contaminants.
The consequences of contamination extend beyond mere cell loss. Biological contaminants can profoundly alter cellular behavior and experimental outcomes. Mycoplasma contamination, for instance, affects virtually every aspect of a cell's behavior, including inhibition of proliferation, chromosomal aberrations, changes to gene expression profiles, and interference with DNA and protein isolation workflows [68]. Similarly, cross-contamination with fast-growing cell lines such as HeLa has invalidated numerous studies, with estimates suggesting 16-35% of cell lines may be affected [69] [68]. The implementation of robust sterility protocols throughout the trypsin-EDTA dissociation process is therefore essential for maintaining the scientific validity of cell-based research.
Contamination Identification and Characterization
Routine monitoring and accurate identification of contamination are crucial components of sterile cell culture practice. Researchers should become familiar with both macroscopic and microscopic signs of contamination in their specific cell lines, as early detection can prevent spread to other cultures and equipment.
Table 1: Identification of Common Contaminants in Cell Culture
| Contaminant Type | Visual/Macroscopic Signs | Microscopic Characteristics | Additional Detection Methods |
|---|---|---|---|
| Bacteria | Cloudy (turbid) media; thin film on surface; sudden pH drop [67] | Tiny, moving granules between cells; rod-shaped or spherical forms [67] | Microbial testing kits [67] |
| Yeast | Turbid media; pH usually increases with heavy contamination [67] | Ovoid or spherical particles that may bud off smaller particles [67] | Microbial testing kits [67] |
| Mold | Turbid media; pH increases with heavy contamination [67] | Thin, wisp-like filaments (hyphae); denser clumps of spores [67] | Microbial testing kits [67] |
| Mycoplasma | No visible changes; no media turbidity [68] | Not detectable by standard light microscopy [68] | PCR, DNA staining, ELISA, histological staining [68] |
| Cross-Contamination | Changes in growth characteristics/morphology [68] | Altered morphology inconsistent with expected cell type [68] | STR profiling, karyotype analysis, isotype analysis [67] |
Differentiating contamination from normal cellular debris is an essential skill. Cell debris typically moves with the motion of the media when disturbed, while bacterial and fungal contaminants often display independent movement, sometimes with a vibrating or circular motion due to cilia or flagella [68]. Mycoplasma represents a particularly challenging contaminant as it can persist in culture without causing turbidity or pH changes, and its small size (0.2-0.3 μm) allows it to pass through standard filtration systems [68]. The estimated contamination rate in academic labs can be as high as 47% [68], making regular testing essential.
Sterile Technique and Workflow Management
Maintaining sterility throughout the trypsin-EDTA dissociation process requires strict adherence to aseptic technique and careful workflow planning. The following diagram illustrates a recommended workflow integrating critical sterility checkpoints:
Figure 1: Sterile Workflow for Trypsin-EDTA Cell Dissociation
Critical Aseptic Techniques
Several aseptic techniques are particularly relevant to trypsin-EDTA dissociation procedures:
- Single Cell Line Processing: Work with only one cell line at a time within the biosafety cabinet (BSC) to prevent cross-contamination [68]. Complete all handling steps for one cell line before introducing materials for another.
- Surface Decontamination: Disinfect all surfaces with 70% isopropanol or ethanol, especially within the BSC [70]. This is crucial after working with trypsin-EDTA, as drips or spills can create reservoirs for contamination.
- Water Bath Maintenance: Keep the water bath and incubator water clean, as these are common contamination sources [70]. Consider adding a decontamination agent to water baths used for warming trypsin-EDTA and other reagents.
- Personal Protective Practices: Bind long hair, avoid touching your face during cell handling, and refrain from talking, coughing, or sneezing toward open containers [70]. Humans represent the most frequent source of contamination.
Antibiotic Use Considerations
While antibiotics like penicillin-streptomycin solutions can be added to cell culture media at ~0.5-1.0% concentration to prevent bacterial contamination [68], their continuous use in trypsin-EDTA workflows requires careful consideration. Routine antibiotic use encourages the development of antibiotic-resistant strains and can allow low-level contamination to persist, potentially developing into full-scale contamination once antibiotics are removed [67]. More concerningly, antibiotics can mask mycoplasma infections and other cryptic contaminants [67]. For these reasons, many experts recommend using antibiotics only as a last resort and for short-term applications, with antibiotic-free cultures maintained in parallel as controls [67]. If antibiotics are used, cells should be cultured without them for 2-3 weeks periodically to test for underlying contaminations [70].
Reagent and Material Solutions
Selecting appropriate reagents and maintaining their sterility is essential for successful trypsin-EDTA dissociation protocols. The following table outlines key research reagent solutions for maintaining sterility:
Table 2: Essential Research Reagent Solutions for Sterile Trypsin-EDTA Cell Dissociation
| Reagent/Material | Function | Sterility Considerations |
|---|---|---|
| Trypsin-EDTA Solutions | Enzymatic dissociation of adherent cells [24] | Use pre-sterilized, irradiated solutions; aliquot to prevent repeated freeze-thaw cycles; verify concentration (typically 0.05%-0.25%) [24] |
| TrypLE Reagents | Animal origin-free alternative to trypsin [24] | Recombinant formulation eliminates viral contamination risk; stable at room temperature [24] [71] |
| Cell Dissociation Buffers | Non-enzymatic dissociation for sensitive applications [24] | Gentle on cells; maintains surface protein integrity; typically phenol red-free [24] |
| Trypsin Inhibitors | Neutralizes trypsin activity post-dissociation [24] | Soybean trypsin inhibitor or defined inhibitors available; prevents continued proteolytic damage [24] |
| Antibiotic/Antimycotic Solutions | Suppresses microbial growth [68] | Use judiciously; Penicillin/Streptomycin for bacteria; Amphotericin B for fungi [68] |
| Mycoplasma Detection/Eradication Kits | Identifies and eliminates mycoplasma contamination [68] | PCR-based detection most efficient; antibiotic cocktails available for decontamination [68] |
| Sterile PBS/BSA without Ca2+/Mg2+ | Washing buffer before trypsinization [24] [72] | Removes divalent cations that inhibit trypsin activity; must be sterile-filtered [24] |
Modern reagent formulations offer significant advantages for sterility maintenance. Recombinant trypsin products, for example, provide animal component-free alternatives that eliminate the risk of viruses and other potential adventitious agents carried in animal-derived components [71]. These defined solutions also avoid undesirable proteins such as carboxypeptidase A and chymotrypsin, which are common contaminants in porcine or bovine-derived trypsin solutions [71].
Quality Control and Authentication Protocols
Implementing rigorous quality control measures is essential for detecting contamination early and preventing its spread. The following protocols should be integrated into standard trypsin-EDTA dissociation workflows:
Mycoplasma Testing Protocol
Mycoplasma contamination represents one of the most challenging contamination issues in cell culture due to its difficulty to detect without specialized testing.
- Sample Collection: Collect supernatant from cultured cells 3-7 days after passage, when cells are approximately 80% confluent [68]. Avoid sampling immediately after antibiotic treatment, as this may yield false negatives.
- Testing Method Selection: Select an appropriate detection method based on laboratory capabilities and requirements. PCR-based methods offer the quickest and easiest detection, with results available in under two hours [68]. Alternative methods include histological staining, enzyme activity measurement, ELISA, fluorescence detection, or DNA staining with DAPI or Hoechst 33258 [68].
- PCR Detection Procedure:
- Use a commercial mycoplasma detection kit capable of detecting over 200 strains of mycoplasma [68].
- Extract DNA from cell culture supernatant according to kit instructions.
- Amplify mycoplasma DNA using provided primers and PCR conditions.
- Run amplification products on a 1% electrophoresis agarose gel for comparison against positive and negative controls [68].
- Interpret results: distinct bands in test samples indicate mycoplasma contamination.
For laboratories requiring broader coverage, select kits that detect over 300 strains of mycoplasma and acholeplasma with an internal control for added accuracy [68]. Testing should be performed every 1-3 months on all actively cultured cell lines, and always upon receipt of new cell lines.
Cell Line Authentication Protocol
Cell line cross-contamination represents a serious threat to research integrity, particularly when working with multiple cell lines in trypsin-EDTA dissociation protocols.
STR Profiling Procedure:
- Sample Preparation: Harvest cells during routine passage following trypsin-EDTA dissociation. Use early-passage cells whenever possible [68].
- DNA Extraction: Isolate genomic DNA using standardized methods, ensuring DNA quality and concentration appropriate for downstream analysis.
- PCR Amplification: Amplify short tandem repeat (STR) loci using commercially available kits specifically designed for cell line authentication.
- Fragment Analysis: Separate and detect amplified fragments using capillary electrophoresis.
- Data Interpretation: Compare resulting STR profile to reference databases for the purported cell line. Mismatches indicate cross-contamination [68].
Authentication Scheduling:
- Perform authentication when first working with a novel cell line [68].
- Conduct testing every few months during continuous culturing to ensure no contamination has occurred [68].
- Always authenticate before submitting publications or applying for grants [68].
- Test immediately if cells demonstrate unexpected morphological or behavioral changes [68].
Emergency Decontamination Protocols
Despite rigorous prevention efforts, contamination events occasionally occur. The following protocols provide systematic approaches for addressing contamination while minimizing impact on other cultures:
Microbial Contamination Response
When bacterial, fungal, or yeast contamination is detected:
- Immediate Isolation: Remove contaminated cultures from the BSC and incubator immediately to prevent spread to other cultures [67] [68].
- Equipment Decontamination: Clean incubators and laminar flow hoods with a laboratory disinfectant, and check HEPA filters [67].
- Toxicity Determination: For irreplaceable cultures, perform a dose-response test to determine antibiotic/antimycotic toxicity levels [67]:
- Dissociate, count, and dilute cells in antibiotic-free medium.
- Dispense cell suspension into a multi-well culture plate.
- Add the antibiotic of choice to each well in a range of concentrations.
- Observe cells daily for signs of toxicity (sloughing, vacuole appearance, decreased confluency, rounding).
- Culture cells for 2-3 passages using antibiotics at a concentration one- to two-fold lower than the toxic concentration.
- Culture cells in antibiotic-free media for one passage, then repeat antibiotic treatment.
- Maintain cells in antibiotic-free medium for 4-6 passages to verify contamination elimination [67].
Mycoplasma Decontamination Protocol
For mycoplasma-contaminated cultures that cannot be replaced:
- Antibiotic Cocktail Selection: Use a chemical treatment containing antibiotics effective against mycoplasma, such as tetracycline, macrolides, and quinolones [68]. Commercial formulations are available that combine multiple antibiotics for broader coverage.
- Concentration Optimization: Dilute antibiotic cocktail based on cell type sensitivity. Common dilutions are 1:500, 1:1000 and 1:10,000 depending on contamination intensity [68].
- Treatment Duration: Add antibiotics directly to culture and maintain for a minimum of four passages, typically requiring two weeks [68].
- Verification Testing: Confirm successful decontamination using PCR-based mycoplasma testing after treatment completion.
Note that mycoplasma decontamination often takes weeks to months to resolve completely, and success is not guaranteed [68]. For this reason, many laboratories choose to discard contaminated cultures rather than risk persistent, low-level contamination.
Maintaining sterility throughout the trypsin-EDTA cell dissociation process requires integrated approach combining rigorous technique, appropriate reagent selection, systematic quality control, and prompt response to contamination events. By implementing the protocols outlined in this application note, researchers can significantly reduce contamination frequency and impact, ensuring the reliability of their cellular models and the validity of their experimental results. In the context of trypsin-EDTA dissociation research, these practices form the foundation for generating reproducible, high-quality data that advances our understanding of cellular mechanisms and supports the development of novel therapeutic approaches.
Beyond Trypsin: Comparative Analysis and Method Validation in Research Models
Within cell culture laboratories, the dissociation of adherent cells is a fundamental, routine procedure. For decades, this process has been dominated by the use of animal-derived trypsin. While effective, trypsin presents several challenges, including the presence of contaminating enzymes, sensitivity to storage conditions, and the potential risk of introducing animal-sourced components into the cell culture system. TrypLE Express, a recombinant trypsin-like enzyme, has been developed as a direct, animal origin-free (AOF) substitute designed to overcome these limitations [73] [74]. Its broader specificity and high purity make it a superior choice for modern cell culture applications, particularly in sensitive research and drug development where consistency and source definition are critical. This application note details the use of TrypLE Express within the broader context of optimizing cell dissociation protocols, providing validated data and detailed methodologies to facilitate its adoption.
TrypLE Express vs. Traditional Trypsin: A Quantitative Comparison
The transition from trypsin to TrypLE Express is supported by several key advantages that enhance experimental reproducibility and cell health. The following table summarizes the critical differences between these two reagents.
Table 1: Comparative Analysis of TrypLE Express and Traditional Trypsin
| Characteristic | TrypLE Express | Traditional Trypsin |
|---|---|---|
| Origin | Recombinant (Fungus), Animal Origin-Free [24] [74] | Porcine Pancreas [24] |
| Enzyme Purity | High (single recombinant protease) [73] | Lower (may contain other proteases) [73] |
| Specificity | Cleaves C-terminal side of Lysine and Arginine [73] | Cleaves C-terminal side of Lysine and Arginine [24] |
| Storage Stability | 24 months at 15-30°C (Room Temperature) [73] [75] | -5 to -20°C (Freezing required) [24] |
| Inactivation Method | Dilution alone is sufficient [73] [74] | Requires trypsin inhibitors (e.g., FBS) [24] |
| Gentleness on Cells | Higher cell viability post-dissociation [24] | Can damage cells due to over-digestion [24] |
| Ideal Use Case | Sensitive cells (e.g., stem cells), serum-free systems, bioproduction [24] [74] | General purpose, robust cell lines |
The core advantage of TrypLE Express lies in its exceptional purity and gentle action. As a single, recombinant enzyme, it increases specificity and reduces the collateral damage to cell surface proteins that can be caused by the cocktail of enzymes present in some trypsin extracts [73]. Furthermore, its stability at room temperature for up to 24 months simplifies inventory management, reduces energy consumption, and ensures the reagent is always ready for use [73] [75].
Recommended Applications and Validated Cell Types
TrypLE Express is suitable for dissociating a wide range of adherent mammalian cells. Its gentle nature makes it particularly valuable for sensitive and therapeutically relevant cell types. The table below lists cell types for which its use has been specifically validated in the literature or by the manufacturer.
Table 2: Validated Cell Types for TrypLE Express Dissociation
| Cell Type Category | Specific Examples |
|---|---|
| Common Cell Lines | CHO, HEK 293, A549 [73] [75] |
| Stem Cells | Embryonic Stem Cells (e.g., H1, H9 lines) [76], Mesenchymal Stem Cells (MSCs) [74] |
| Primary Cells | Primary Human Keratinocytes [73] |
| Other Sensitive Cells | Early-passage cells, cells where surface protein integrity is important [24] |
Independent research has demonstrated the efficacy of TrypLE Express in challenging applications. A comparative study on human embryonic stem cells (hESC) concluded that TrypLE Express, alongside other dissociative solutions, enabled a fast switch from mechanical passaging to a single-cell split technique, supporting the large-scale expansion needed for tissue engineering purposes [76]. For mesenchymal stem cells (MSCs), the manufacturer explicitly recommends TrypLE Express over trypsin, as the latter results in lower cell viability upon passage [74].
Detailed Experimental Protocol for Cell Dissociation
The following workflow and detailed protocol provide a standard operating procedure for using TrypLE Express to dissociate adherent cells, which can be directly substituted for trypsin in existing protocols with minimal adjustment [77].
Materials Required
- TrypLE Express Enzyme (1X) [73]
- DPBS (Dulbecco's Phosphate Buffered Saline) without calcium and magnesium [77]
- Complete Growth Medium (pre-warmed)
- Culture Vessel (flask, plate)
- Centrifuge Tubes
- Pipettes and Aspiration System
Step-by-Step Procedure
- Preparation: Warm TrypLE Express enzyme and complete growth medium to 37°C prior to starting dissociation [77].
- Rinse: Aspirate the growth medium from the culture vessel and gently rinse the cell layer with DPBS (without calcium and magnesium) to remove any residual serum and divalent cations. Decant the DPBS [77].
- Enzyme Application: Add an appropriate volume of pre-warmed TrypLE Express to the vessel (e.g., 5 mL for a T-75 flask). Ensure the solution completely covers the cell monolayer by gently rocking the vessel [77].
- Incubation: Leave the vessel with TrypLE Express in a 37°C incubator for 5-10 minutes. Monitor the cells periodically under a microscope. The cells will typically round up and detach. For more resilient cells, incubation can be extended; studies indicate that incubations of up to 30-45 minutes at 37°C are generally well-tolerated [74].
- Harvesting: Once the majority of cells are detached, gently tap the side of the vessel to dislodge any remaining adherent cells. Add a volume of pre-warmed complete growth medium that is at least equal to the volume of TrypLE Express used (e.g., 5-10 mL for a T-75 flask) to inactivate the enzyme by dilution. Tilt the flask to rinse the surface thoroughly [77] [74].
- Centrifugation: Transfer the cell suspension to a centrifuge tube. Centrifuge at approximately 100 x g for 5-10 minutes to form a cell pellet [77].
- Resuspension: Carefully decant the supernatant. Resuspend the cell pellet in 2-5 mL of fresh, pre-warmed growth medium by pipetting to break up any cell clumps [77].
- Counting and Seeding: Count the cells using an automated cell counter or hemocytometer. The cells are now ready for downstream applications, such as subculturing, experimentation, or cryopreservation [77].
The Scientist's Toolkit: Essential Reagent Solutions
Successful cell dissociation relies on a suite of key reagents. The following table outlines essential solutions and their specific functions in the workflow.
Table 3: Key Research Reagent Solutions for Cell Dissociation
| Reagent Solution | Function & Role in the Protocol |
|---|---|
| TrypLE Express (1X) | The primary recombinant enzyme that digests cell-adhesion proteins, enabling gentle and effective cell detachment. It is the direct replacement for trypsin [73]. |
| DPBS (without Ca2+/Mg2+) | A balanced salt solution used to wash the cell monolayer before dissociation. The absence of calcium and magnesium is crucial as it weakens cell-cell adhesion and prevents inhibition of TrypLE activity [77]. |
| Complete Growth Medium | Used to inactivate TrypLE Express via dilution after dissociation. The serum or proteins in the medium act as a sink for any residual enzyme activity, eliminating the need for specific trypsin inhibitors [73] [74]. |
| Trypan Blue Stain (0.4%) | A vital dye used in cell counting to distinguish between live (unstained) and dead (blue-stained) cells, allowing for accurate assessment of viability post-dissociation [77]. |
Troubleshooting and Protocol Adaptation
Despite its robustness, users may occasionally encounter issues. The following diagram outlines a logical troubleshooting path for common problems.
Another common issue is poor cell adherence after passaging. If this occurs, it is recommended to trypsinize the cells for a shorter duration or use a gentler reagent like TrypLE Express. Furthermore, ensuring a proper centrifugation step (100 x g for 5-10 minutes) to remove the diluted enzyme and resuspending the pellet in fresh, pre-warmed medium before seeding can significantly improve adherence [74]. For applications requiring the passaging of cells as clumps rather than single cells, alternatives such as EDTA-based solutions should be considered instead of TrypLE Express [74].
Collagenase and Hyaluronidase for Tissue-Specific Dissociation
Within the broader context of trypsin EDTA cell dissociation protocol research, it is evident that while trypsin remains a common reagent for cell dissociation, its limitations in handling complex tissues have driven the development of more sophisticated enzymatic approaches. Trypsin primarily targets cell-cell adhesion molecules but is less effective against the dense extracellular matrix (ECM) components found in many tissues. For complex tissues and specialized applications like single-cell sequencing and organoid generation, researchers are increasingly turning to enzyme combinations that more comprehensively digest tissue-specific ECM components. Among these, collagenase and hyaluronidase have emerged as particularly effective agents, either used individually or in combination, for preserving cell viability and function while achieving high dissociation efficiency across diverse tissue types.
Chemical Mechanisms and Rationale
The efficacy of collagenase and hyaluronidase stems from their targeted action on specific extracellular matrix components:
Collagenase specifically hydrolyzes collagen, a major structural protein in the extracellular matrix that provides tensile strength to tissues. By breaking down collagen networks, collagenase disrupts the primary structural framework that maintains tissue integrity [78].
Hyaluronidase targets hyaluronic acid (hyaluronan), a key glycosaminoglycan component of the extracellular matrix that contributes to tissue volume, hydration, and cell migration pathways. Hyaluronidase principally cleaves this abundant polysaccharide, facilitating tissue permeability and access for other enzymes [78].
When used in combination, these enzymes exhibit synergistic effects for several reasons. The initial action of hyaluronidase increases tissue permeability, allowing collagenase better access to its collagen substrates. Furthermore, this combination simultaneously attacks both the fibrous protein scaffold (via collagenase) and the glycosaminoglycan ground substance (via hyaluronidase) of the ECM, resulting in more comprehensive tissue dissociation than either enzyme could achieve alone [33] [78].
Quantitative Comparison of Dissociation Efficiency
Performance Across Tissue Types
Table 1: Dissociation Efficiency and Cell Viability Across Different Tissues and Enzymatic Methods
| Tissue Type | Enzyme Used | Dissociation Efficiency | Cell Viability | Source/Study |
|---|---|---|---|---|
| Bovine Liver Tissue | 1% Collagenase + Pronase OR Hyaluronidase | 37%-42% (chemical only); 93 ± 8% (with mechanical force) | >90% | [33] |
| Colorectal Cancer Tissue | Collagenase type II | Superior dissociation efficiency; Highest organoid counts | Preserved viability | [46] [55] |
| Colorectal Cancer Tissue | Hyaluronidase type IV | Superior dissociation efficiency; Largest organoid expansion | Preserved viability | [46] [55] |
| Colorectal Cancer Tissue | TrypLE | Limited dissociation efficiency | Superior preservation | [46] [55] |
| Colorectal Cancer Tissue | Trypsin-EDTA (T/E) | Limited dissociation efficiency | Superior preservation | [46] [55] |
Cancer Stem Cell Preservation
Table 2: Impact of Enzymatic Dissociation on Stem Cell Marker Expression and Organoid Formation
| Parameter | Collagenase | Hyaluronidase | TrypLE | Trypsin-EDTA |
|---|---|---|---|---|
| LGR5+ Stem Cell Population | Highest proportion | High proportion | Lower proportion | Lower proportion |
| CD133+ Stem Cell Population | Highest proportion | High proportion | Lower proportion | Lower proportion |
| Organoid Formation | Highest organoid counts | Largest organoid expansion | Reduced formation | Reduced formation |
| Total Cell Yield/mg Tissue | Highest | High | Lower | Lower |
Experimental Protocols
Rapid Tissue Dissociation for Single-Cell Analysis
This protocol from 2021 research enables quick preparation of single-cell suspensions from biopsy cores for downstream applications like flow cytometry and single-cell sequencing [33].
Materials:
- Tissue biopsy cores (2.5 mm diameter, ~10 mg weight)
- 1% collagenase type I in HBSS
- Pronase OR hyaluronidase (100 U/µL in HBSS)
- Inactivating agent (EDTA or media with FBS)
- Flat-bottom 96-well plates
- Pipettes and DNA Lo-Bind Eppendorf tubes
Procedure:
- Obtain frozen biopsy cores and thaw at 4°C for 24 hours, then bring to room temperature for 1 hour.
- Weigh tissue samples precisely and transfer to appropriate vessels.
- Prepare fresh enzyme solutions immediately before use.
- Apply dissociation media in a 1 mg tissue per 10 µL reagent ratio.
- For chemical-mechanical dissociation, add controlled mechanical force during chemical incubation.
- Maintain at optimal enzyme activity temperatures (25°C or 37°C).
- At time intervals (0, 5, 10, 15 minutes), gently pipette 5 µL of solution from the center of the tube.
- Perform 1:10 dilution with inactivating agent to stop enzymatic activity.
- Analyze cell suspension using hemocytometry, flow cytometry, or other appropriate methods.
Notes: Total processing time is 15 minutes. Freshly prepared reagents are critical to prevent enzyme degradation variability. The combination with mechanical force dramatically improves dissociation efficiency from 37-42% to 93% [33].
Patient-Derived Colorectal Cancer Organoid Generation
This 2025 protocol systematically compares enzymatic methods for establishing patient-derived organoids from colorectal cancer tissues [46] [55].
Materials:
- Freshly resected colorectal cancer tissue
- Enzyme solutions: 1 mg/ml Hyaluronidase type IV-S, 1 mg/ml Collagenase type II, 1X TrypLE Express, 0.005% Trypsin-EDTA
- Transfer medium: Advanced DMEM/F12 with supplements
- Wash buffer: DPBS with antibiotics
- Shaking water bath
- Centrifuge
Procedure:
- Collect freshly resected CRC tissues and preserve in ice-cold transfer medium.
- Wash tissue >5 times with ice-cold DPBS containing antibiotics until supernatant is clear.
- Mechanically mince tissue into 0.5-1 mm pieces using sterile scalpel.
- Divide tissue fragments equally by weight into four portions.
- Digest each portion with one of the four enzyme solutions individually:
- 1 mg/ml Hyaluronidase type IV-S
- 1 mg/ml Collagenase type II
- 1X TrypLE Express
- 0.005% Trypsin-EDTA
- Incubate tubes on shaking water bath at 200 rpm, 37°C for 30 minutes.
- Halt digestion by adding 10% FBS-enriched DMEM.
- Centrifuge at 300 × g for 2 minutes at 4°C to pellet cells.
- Resuspend cell pellet in unsupplemented DMEM to wash away residual FBS.
- Assess cell viability using Trypan Blue exclusion assay and/or 7-AAD staining with flow cytometry.
Evaluation Parameters:
- Cell viability via Trypan Blue exclusion and 7-AAD flow cytometry
- Cell count per milligram of tissue
- Quality of isolation via brightfield microscopy
- Cancer stem cell populations (LGR5+, CD133+) via flow cytometry
- Organoid generation and growth over 11 days using confocal microscopy
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Tissue Dissociation Protocols
| Reagent | Concentration/Type | Primary Function | Application Notes |
|---|---|---|---|
| Collagenase Type I | 1% solution | Degrades collagen fibers in ECM | Effective for liver tissue dissociation; often used with pronase or hyaluronidase [33] |
| Collagenase Type II | 1 mg/mL | Targets collagen in complex tissues | Optimal for colorectal cancer tissue; preserves stem cell populations [55] |
| Hyaluronidase Type IV | 1 mg/mL or 100 U/µL | Cleaves hyaluronic acid in ECM | Increases tissue permeability; enhances access for other enzymes [33] [55] |
| Pronase | Concentration varies | Broad-spectrum protease activity | Complements collagenase action in liver tissue dissociation [33] |
| Trypsin-EDTA | 0.005%-0.25% | Disrupts cell-cell adhesions | Gentle on cells but limited dissociation efficiency in complex tissues [55] |
| TrypLE Express | 1X solution | Recombinant trypsin substitute | Preserves viability but lower yield for organoid culture [55] |
| DNase I | 1 mg/mL | Degrades DNA from lysed cells | Prevents cell clumping; often used in enzyme blends [78] |
| Dispase II | 5 U/mL | Neutral protease for basement membrane | Gentle on epithelial cells; prevents clumping in suspension cultures [78] [79] |
Workflow Visualization
Figure 1: Comprehensive workflow for tissue dissociation using collagenase and hyaluronidase, illustrating the integration of mechanical and enzymatic processing steps leading to various downstream applications.
Figure 2: Mechanism of action diagram illustrating how collagenase and hyaluronidase target different extracellular matrix components to facilitate tissue dissociation while preserving critical stem cell populations for organoid formation.
The strategic application of collagenase and hyaluronidase for tissue-specific dissociation represents a significant advancement beyond traditional trypsin EDTA protocols, particularly for complex tissues and stem cell-rich applications. The quantitative evidence demonstrates that these enzymes, especially when used in combination, achieve superior dissociation efficiency while critically preserving viability and stem cell populations essential for advanced research applications including single-cell analysis and patient-derived organoid generation. The provided protocols and quantitative benchmarks offer researchers a validated foundation for implementing these methods across various tissue types, with particular demonstrated efficacy in liver and colorectal cancer tissues. As the field moves toward more standardized yet flexible dissociation workflows, collagenase and hyaluronidase combinations present a powerful toolset for bridging tissue structure and single-cell analysis.
Within cell biology research and therapeutic development, the process of cell dissociation is a critical foundational step. While enzymatic methods, particularly those using trypsin-EDTA, are widely used for detaching adherent cells, they present significant limitations for certain applications. The cleavage of cell surface proteins by proteolytic enzymes can compromise experimental outcomes, particularly in studies focusing on membrane receptors, cell signaling, and immunophenotyping [38] [60]. Non-enzymatic dissociation methods, primarily employing ethylenediaminetetraacetic acid (EDTA)-based buffers and mechanical dislodgement techniques, offer a vital alternative. These methods enable the harvest of cells with intact surface architectures, preserving critical biological information that is essential for downstream analysis in drug development and basic research. This document details the application, protocols, and key considerations for utilizing these non-enzymatic approaches within a modern research framework.
Core Principles and Quantitative Comparisons
Mechanism of Action
Non-enzymatic cell dissociation operates on two primary principles:
- EDTA-Based Chelation: EDTA acts as a chelating agent that binds calcium (Ca²⁺) and magnesium (Mg²⁺) ions in the extracellular environment. These divalent cations are essential cofactors for cadherin proteins, which mediate cell-cell adhesion, and for integrins, which facilitate cell-matrix attachment. By sequestering these ions, EDTA disrupts these interactions, leading to a loosening of cell attachment [38] [4].
- Mechanical Dislodgement: This approach uses physical force to separate cells from their substrate and from each other. Methods range from gentle pipetting and scraping to more standardized automated systems. Mechanical force is often necessary to dissociate cells after EDTA pretreatment, especially for strongly adherent cell types [38] [61].
Impact on Cell Surface Proteins and Viability
The primary advantage of non-enzymatic methods is the preservation of cell surface molecules. Research has demonstrated that enzymatic treatments can significantly alter the cell surface proteome. For instance, accutase, often considered a mild enzyme, was found to cleave Fas ligands (FasL) and Fas receptors from the surface of macrophages, an effect not observed with EDTA-based detachment. The recovery of these surface proteins post-accutase treatment required up to 20 hours, highlighting the potential for enzymatic methods to introduce significant artifacts in time-sensitive experiments [38].
Table 1: Comparative Analysis of Cell Detachment Methods
| Parameter | Trypsin-EDTA (Enzymatic) | EDTA-Based Buffer (Non-Enzymatic) | Mechanical Scraping | Automated Mechanical (e.g., Medimachine) |
|---|---|---|---|---|
| Primary Mechanism | Proteolytic cleavage of adhesion proteins | Chelation of Ca²⁺ and Mg²⁺ ions | Physical force applied by a scraper | Tissue pressing and filtering through a mesh [61] |
| Impact on Surface Proteins | Degrades most surface proteins; can cleave specific targets like FasL/Fas [38] | Preserves integrity of surface proteins and receptors [38] [4] | Preserves surface proteins but may cause physical damage | Better preservation of lysosome and mitochondria labeling [61] |
| Typical Cell Viability | High (>90% for MSC) [60] | Lower (e.g., ~69% for MSC); highly cell-type dependent [60] | Variable; can be lower due to shear stress | Comparable to enzymatic methods in some tissues; improved in others [61] |
| Typical Detachment Time | Rapid (5-15 minutes) [4] | Slower (can take 15+ minutes) [60] [4] | Very rapid (minutes) | Standardized run times (e.g., 15 minutes) [61] |
| Best Suited For | Strongly adherent cells; routine sub-culturing | Lightly adherent cells; flow cytometry; surface protein studies [4] | Cells sensitive to all chemical/enzymatic treatments | Processing solid tissue samples for single-cell suspensions [61] |
| Key Limitations | Alters cell surface and extracellular matrix [38] | Less effective on strongly adherent cells; may require scraping [38] | Can reduce viability and clump cells; operator-dependent | May be less efficacious for certain tissues like frozen biopsies [80] |
Detailed Experimental Protocols
Protocol 1: Standard Cell Detachment Using EDTA-Based Buffer
This protocol is designed for dissociating lightly adherent cells from culture vessels for applications requiring intact cell surface proteins [4].
Research Reagent Solutions:
- EDTA-Based Cell Dissociation Buffer: A commercial, isotonic, enzyme-free solution of salts and chelating agents in Ca²⁺- and Mg²⁺-free PBS [60].
- Dulbecco's Phosphate Buffered Saline (DPBS), without calcium and magnesium: For rinsing cells to remove divalent cations.
- Complete Growth Medium: Contains serum, which helps neutralize the EDTA and provides nutrients for resuspended cells.
Methodology:
- Preparation: Warm the cell dissociation buffer and DPBS to 37°C prior to use. Pre-warm the complete growth medium.
- Rinsing: Aspirate and discard the spent cell culture media from the flask. Thoroughly rinse the cell monolayer twice with a generous volume (e.g., 5 mL for a T75 flask) of warm, Ca²⁺- and Mg²⁺-free DPBS. This step is critical to remove residual calcium and serum that would inhibit dissociation.
- Application of Dissociation Buffer: Add a sufficient volume of cell dissociation buffer (e.g., 5 mL for a T75 flask) to cover the cell sheet. Gently rock the vessel to ensure full coverage.
- Incubation: Incubate the vessel at room temperature for 1-2 minutes. Observe the cells under an inverted microscope. For many lightly adherent lines, this is sufficient. If cells remain attached, tap the flask firmly against the palm of your hand. If needed, allow the flask to sit for an additional 2-5 minutes at room temperature and tap again.
- Neutralization and Collection: Once the majority of cells are detached (appear rounded and refractive under the microscope), add at least 5 mL of complete growth medium to the flask. Gently pipette the medium across the surface to dislodge any remaining cells and create a homogeneous cell suspension.
- Centrifugation and Resuspension: Transfer the cell suspension to a conical tube and centrifuge at approximately 100–500 × g for 5–10 minutes. Discard the supernatant and resuspend the cell pellet in fresh, pre-warmed complete medium.
- Cell Counting: Determine viable cell density and percent viability using an automated cell counter or manual method (e.g., Trypan Blue exclusion) [4].
Protocol 2: Semi-Automated Mechanical Dissociation of Primary Tissue
This protocol, adapted from studies on establishing human intestinal organoids, describes the use of a semi-automated system (e.g., Cytiva Via Extractor) for dissociating primary tissue into single cells or crypts with minimal enzymatic exposure [80].
Research Reagent Solutions:
- DPBS, without calcium and magnesium: For washing tissue samples.
- EDTA Solution (e.g., 2.5 mM): To chelate ions and weaken cell junctions in the tissue.
- Basement Membrane Matrix (e.g., Matrigel): For embedding and culturing isolated cells or structures.
Methodology:
- Tissue Preparation: Obtain fresh or cryopreserved human tissue biopsies (e.g., mucosal biopsies). Wash the tissue three times with cold, sterile DPBS to remove any contaminants or microbiota.
- Loading: Place the tissue into a pre-chilled, sterile pouch or chamber of the dissociation instrument. For some systems, the pouch may be pre-coated with 0.1% BSA to reduce non-specific adhesion.
- Dissociation: Add 5 mL of cold 2.5 mM EDTA solution to the pouch. Seal the pouch in a sterile environment and load it into the instrument.
- Automated Processing: Run the instrument using optimized parameters (e.g., 150 rpm for 7 minutes for fresh tissue; 5 minutes for cryopreserved tissue) at 4°C. The system gently agitates the tissue in the EDTA solution, mechanically releasing the desired cellular structures.
- Collection: After the cycle is complete, collect the resulting suspension, which contains single cells or tissue structures like intestinal crypts.
- Pelletation and Culturing: Centrifuge the suspension at 800 × g for 5 minutes. Discard the supernatant and resuspend the pellet in an appropriate matrix for downstream culture or analysis [80].
Diagram 1: Non-enzymatic cell dissociation workflow.
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagent Solutions for Non-Enzymatic Dissociation
| Item | Function/Description | Example Application |
|---|---|---|
| EDTA-Based Cell Dissociation Buffer | Pre-formulated, isotonic, enzyme-free solution that chelates Ca²⁺/Mg²⁺ ions to disrupt cell adhesion without proteolytic activity [60] [4]. | Detaching lightly adherent cell lines for flow cytometry or receptor studies. |
| Versene Solution | A commercial PBS-based solution containing EDTA, acting as a non-enzymatic cell detachment agent [38]. | Used as a control in studies comparing the effects of enzymatic vs. non-enzymatic detachment on surface markers [38]. |
| Medimachine II System | An automated mechanical disaggregation system that processes minced tissue fragments through a sterile mesh to produce single-cell suspensions without enzymes [61]. | Generating single-cell suspensions from solid tissues like spleen, testis, and liver for functional assays. |
| Cytiva Via Extractor | A semi-automated platform that combines gentle mechanical agitation with reagent exposure (e.g., EDTA) to dissociate primary tissue [80]. | Establishing organoid cultures from fresh intestinal mucosal biopsies with improved consistency. |
| DPBS (without Ca²⁺ & Mg²⁺) | A balanced salt solution used to wash cells and tissues, removing divalent cations that are essential for cell adhesion. | Essential rinse step prior to application of EDTA-based dissociation buffers. |
| Cell Scraper | A manual tool with a flexible plastic blade used to physically dislodge cells from the culture surface by scraping. | Harvesting cells that are sensitive to or cannot be detached by enzymatic or chelating methods. |
Diagram 2: EDTA disrupts cell adhesion by chelating calcium ions.
Applications and Strategic Considerations
Non-enzymatic methods are indispensable in specific research contexts. Their use is critical in:
- Flow Cytometry and Immunophenotyping: Preserving the integrity of cell surface antigens (e.g., CD markers, FasL, Fas receptor) for accurate detection and quantification [38].
- Stem Cell Research: Maintaining the "stemness" and surface marker profile of stem cells during passaging, which can be altered by enzymatic digestion [38].
- Primary Tissue Digestion: As a standalone method or combined with gentle enzymes, for establishing primary cultures or organoids from sensitive tissues, minimizing cellular stress and damage [80].
- Receptor-Ligand Interaction Studies: Ensuring that surface receptors and ligands involved in signaling pathways (e.g., the FasL/Fas apoptosis pathway) are not cleaved or altered prior to experimentation [38].
A key strategic consideration is the trade-off between surface protein integrity and cell viability. While non-enzymatic methods excel at the former, viability post-dissociation can be lower than with trypsin, as observed in mesenchymal stem cells [60]. Furthermore, some cell types or tissues may require a combination of a brief EDTA pretreatment followed by very gentle mechanical dislodgement (e.g., vigorous pipetting or use of an automated system) to achieve effective dissociation without compromising cell health [38] [61]. The choice of method should therefore be empirically determined based on the cell type, the specific requirements of the downstream application, and a careful balance between yield, viability, and surface marker preservation.
Within the context of a broader thesis on trypsin-EDTA cell dissociation protocol research, this application note addresses the critical role of tissue dissociation in establishing physiologically relevant patient-derived organoids (PDOs). As three-dimensional (3D) in vitro models that recapitulate the histological and molecular characteristics of original patient tumors [81], PDOs have progressively revolutionized cancer research and drug development. The dissociation protocol employed—specifically the choice between enzymatic agents like trypsin-EDTA and non-enzymatic alternatives—profoundly impacts the success rate, cellular viability, and phenotypic fidelity of the resulting organoid cultures [80] [82]. This document provides a standardized framework for validating dissociation method performance in PDO generation, complete with quantitative metrics, detailed protocols, and analytical workflows tailored for researchers and drug development professionals.
Performance Metrics for Dissociation Methods in PDO Generation
Selecting an appropriate dissociation method requires careful consideration of multiple performance indicators. The table below summarizes key quantitative metrics for evaluating success in PDO establishment.
Table 1: Performance Metrics for Tissue Dissociation Methods in PDO Generation
| Performance Metric | Conventional EDTA | Semi-Automated EDTA | Trypsin-EDTA (0.05%) | Significance/Notes |
|---|---|---|---|---|
| Derivation Success Rate (Fresh Tissue) | Baseline | Improved (Specific data not provided) | Comparable to conventional EDTA | Semi-automated method demonstrates an improved rate of successful organoid derivation from fresh tissue [80]. |
| Crypt Isolation Efficacy (Fresh Tissue) | Baseline | Higher | Information Missing | Semi-automated dissociation provides greater cell yield for fresh biopsies [80]. |
| Organoid Growth Kinetics | Baseline | No Significant Difference | No Significant Difference (when optimized) | No significant differences in growth/recovery or cell shedding/viability observed between conventional and semi-automated methods [80]. |
| Cellular Composition & Phenotype | Baseline | No Significant Difference | No Significant Difference (when optimized) | Populations of key cell types (e.g., LGR5+ stem cells, MUC2+ Goblet cells) remain similar [80]. |
| Cell Viability Post-Dissociation | Information Missing | Information Missing | >90% (General standard for enzymatic dissociation) | Cell viability should be routinely monitored and exceed 90% for healthy cultures [82]. |
Experimental Protocols for Dissociation and Validation
Protocol 1: Semi-Automated Mechanical Dissociation for Intestinal Organoids
This protocol, adapted from a 2025 Scientific Reports paper, uses a semi-automated platform (e.g., Cytiva Via Extractor) for EDTA-based dissociation of human intestinal mucosal biopsies, demonstrating an expedited workflow and improved consistency [80].
- Step 1: Tissue Preparation: Obtain human intestinal mucosal biopsies. Wash three times with cold, sterile phosphate-buffered saline (PBS) to remove microbiota. For cryopreserved tissue, thaw appropriately.
- Step 2: Semi-Automated Dissociation: Place tissue in 0.1% BSA-coated pouches. Add 5 mL of 2.5 mM EDTA in PBS. Seal the pouch in a sterile environment.
- Step 3: Instrument Setup: Load the pouch into the semi-automated dissociator. Run at 4°C using optimized parameters: 150 rpm for 7 minutes for fresh tissue or 5 minutes for cryopreserved tissue.
- Step 4: Crypt Collection: Following the program, collect the crypt suspension in PBS. Centrifuge at 800 x g for 5 minutes. Proceed to organoid culture in Matrigel.
Protocol 2: Enzymatic Dissociation with Trypsin-EDTA for Established Organoids
This protocol, synthesized from manufacturer guidelines [83] [84] [82], is ideal for dissociating established intestinal organoid cultures for subsequent monolayer generation or subculturing.
- Step 1: Reagent Preparation: Pre-warm Trypsin-EDTA (0.05%), DMEM/F-12 with 15 mM HEPES, and complete growth medium to 37°C.
- Step 2: Organoid Harvesting: Aspirate medium from organoid cultures (e.g., 2-3 wells of a 24-well plate). Add 1 mL of Gentle Cell Dissociation Reagent per well and incubate at room temperature for 1 minute. Vigorously pipette to disrupt the Matrigel dome and pool the harvested wells.
- Step 3: Trypsin-EDTA Incubation: Centrifuge at 200 x g for 5 minutes at 2-8°C. Aspirate supernatant and resuspend the pellet in 1 mL of warm 0.05% Trypsin-EDTA. Incubate at 37°C for 5-10 minutes.
- Step 4: Neutralization and Seeding: Vortex or pipette vigorously to dissociate into single cells/small fragments. Add an equal volume of DMEM/F-12 to neutralize the trypsin. Centrifuge again, resuspend in culture medium, and seed for monolayer culture or further expansion.
Diagram 1: PDO Generation Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful PDO generation and culture rely on a suite of specialized reagents. The following table outlines key materials and their functions.
Table 2: Essential Reagents for PDO Generation and Culture
| Reagent/Catalog Item | Function in PDO Workflow |
|---|---|
| Trypsin-EDTA Solution (0.05%-0.25%) | Proteolytic enzyme (trypsin) breaks down cell-adhesion proteins, while EDTA chelates calcium ions, facilitating dissociation of adherent cells and tissues [10] [83]. |
| Gentle Cell Dissociation Reagent | A non-enzymatic, chelation-based solution used to gently dissociate cells, helping to preserve critical cell surface proteins [84] [82]. |
| Extracellular Matrix (e.g., Matrigel, BME) | A natural hydrogel providing a 3D scaffold that supports organoid growth, self-organization, and polarization [80] [81]. |
| Wnt Agonists (e.g., R-Spondin, Wnt3a) | Growth factors added to culture medium to activate the Wnt signaling pathway, which is essential for the proliferation and maintenance of LGR5+ stem cells in many organoid types [81]. |
| EGF (Epidermal Growth Factor) | A mitogen added to culture medium to promote epithelial cell proliferation via activation of the EGFR pathway [81]. |
| Rho-Kinase Inhibitor (Y-27632) | Significantly improves the viability and recovery of single cells and organoid fragments after dissociation and passage by inhibiting apoptosis [80] [84]. |
Analytical Validation and Downstream Application
Phenotypic and Functional Validation of PDOs
Following successful dissociation and organoid establishment, rigorous validation is required to confirm the model's fidelity.
- Histological and Immunophenotypic Analysis: Confirm that PDOs recapitulate the original tumor's histology and protein marker expression. For colorectal cancer PDOs, this includes verifying a CK20+/CK7- immunophenotype and expression of markers like CDX2 and β-catenin [85]. Immunofluorescence for stem cell markers (e.g., LGR5) and differentiation markers (e.g., MUC2 for goblet cells) is standard [80].
- Genetic Fidelity Assessment: Perform whole exome sequencing or panel sequencing to verify that PDOs retain the key driver mutations of the primary tumor, with studies showing up to 96% similarity [85].
- Functional Drug Response Testing: Treat PDOs with standard-of-care chemotherapeutics (e.g., FOLFOXIRI components) or targeted agents. Viability can be assessed using assays like CellTiter-Glo. The predictive accuracy of PDOs for patient response is reported to be around 76% on average, underscoring their clinical relevance [85].
Diagram 2: PDOs in Drug Development
The initial tissue dissociation step is a critical determinant of success in patient-derived organoid generation. While both conventional EDTA and trypsin-EDTA protocols are effective, the emergence of semi-automated dissociation platforms offers improved standardization and yield for specific tissue types. By adhering to the detailed protocols and validation metrics outlined in this document, researchers can robustly generate high-fidelity PDOs. These advanced 3D models are indispensable tools for bridging the gap between in vitro discovery and clinical application, ultimately accelerating the development of novel and personalized cancer therapeutics.
Within the broader scope of research on trypsin-EDTA cell dissociation protocols, the specific application of enzymatic digestion for the isolation of delicate structures like the retinal vasculature presents a unique set of challenges and considerations. While trypsin-EDTA is a cornerstone for dissociating adherent cell monolayers in standard cell culture, its utility in preserving complex, native microarchitectures in tissues is limited [24] [86]. This application note details a comparative case study evaluating trypsin and elastase digestion techniques for isolating the murine retinal vasculature. The integrity of this vascular network is crucial for studying microvascular diseases such as diabetic retinopathy [87] [88]. The findings demonstrate that elastase digestion emerges as a superior technique, effectively preserving key cellular associations and junctional complexes that are compromised by trypsin digestion [87].
Comparative Evaluation: Trypsin vs. Elastase
Key Differential Findings
A direct comparative study revealed significant differences in the performance of trypsin and elastase when used to isolate the retinal vascular network for pathological examination [87] [88].
Table 1: Comparative Analysis of Trypsin and Elastase Digestion on Retinal Vasculature
| Feature Evaluated | Trypsin Digestion | Elastase Digestion |
|---|---|---|
| Pericyte-Endothelial Cell Association | Disrupted association [87] | Effectively preserved [87] |
| Adherens Junctions (VE-Cadherin) | Disrupted localization and integrity [87] | Preserved integrity and membrane localization [87] |
| Tight Junctions (ZO-1) | Disrupted [87] | Preserved [87] |
| Ease of Tissue Cleaning | More difficult, not easy [87] | Easier, more effective [87] |
| Overall Vascular Architecture | Compromised | Maintained |
Underlying Mechanisms and Specificities
The differential impact of these two enzymes can be traced to their distinct biochemical specificities.
- Trypsin Specificity: Trypsin is a serine protease that cleaves peptide bonds at the carboxyl side of the basic amino acids lysine (K) and arginine (R), except when followed by a proline [89] [86]. This high specificity supports its effectiveness in standard cell dissociation, where it targets adhesion proteins. However, in native tissues, its activity is modulated by peptide size and isoelectric point, and its access to cleavage sites is governed by protein conformation [89].
- Elastase Specificity: Elastase is also a serine protease but cleaves at the carboxyl side of small, neutral amino acids like alanine, glycine, and valine [90] [91]. Its natural substrate is elastin, a key component of the extracellular matrix in connective tissues [91] [92]. This different cleavage specificity appears less detrimental to the integral membrane and junctional proteins that are critical for maintaining vascular integrity.
Detailed Experimental Protocols
Elastase Digestion Technique for Retinal Vasculature
The following protocol, adapted from the study, provides a reliable method for isolating an intact retinal vascular network [87].
Tissue Harvesting and Fixation:
- Euthanize the mouse and enucleate the eyes.
- Immediately fix the entire eyes in 4% Paraformaldehyde (PFA) for at least 1 week at 4°C.
Retina Isolation:
- Using fine scissors and forceps, cut off and discard the anterior half of the eye (cornea and lens).
- Carefully isolate the retina, ensuring the optic nerve head remains attached.
Washing and Incubation:
- Rinse the fixed retina overnight in distilled water to remove excess fixative.
- Incubate the retina in Elastase Solution [40 U/mL in 100 mM sodium phosphate buffer (pH 6.5), with 150 mM NaCl and 5 mM EDTA] at 37°C for 2 hours. Note: The enzyme is inactive at this pH but diffuses into the tissue.
Enzyme Activation and Digestion:
- Transfer the retina with a drop of elastase solution to 1 mL or more of Activating Solution (Tris buffer, pH 8.5).
- Incubate at room temperature for at least 12 hours. Elastase functions optimally at pH 8.5.
Tissue Cleaning and Slide Mounting:
- Transfer the retina to a shallow Petri dish filled with lint-free deionized water.
- Under a dissecting microscope, use single-hair brushes to gently remove the neuronal and glial tissue from the vascular tree.
- Transfer the cleaned vasculature to a fresh dish of clean water for final cleaning until the network is clear.
- Using an inverted Pasteur pipette, transfer the vasculature to a clean, lint-free microscope slide.
- Air-dry the slide overnight to allow the vasculature to adhere.
Trypsin Digestion Technique for Retinal Vasculature
The trypsin protocol used for comparison is outlined below [87].
Tissue Preparation:
- Fix retinas in 4% PFA and wash overnight under running water.
- Place retinas in deionized water with shaking for 4-6 hours to loosen neural tissue from the vascular bed.
Enzyme Digestion:
- Transfer retinas to a Trypsin Solution (concentrations ranging from 0.5% to 2.5% in a balanced salt solution without calcium and magnesium).
- Incubate at 37°C with gentle shaking overnight.
Tissue Cleaning:
- The following day, clean the retina under a dissecting microscope to remove non-vascular tissue. The study notes that this cleaning process was "not easy in this method like with elastase method" [87].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Retinal Vasculature Isolation and Analysis
| Reagent / Kit | Function / Application | Source / Example |
|---|---|---|
| Porcine Pancreatic Elastase | Digests non-vascular retinal components while preserving vascular integrity. | Sigma-Aldrich (Product #324682) [87] |
| Trypsin, Powder (1:250) | Standard protease for cell dissociation; used for comparative digestions. | ThermoFisher Scientific [87] [24] |
| Anti-VE-Cadherin Antibody | Immunofluorescence staining to visualize and assess adherens junction integrity. | Santa Cruz Biotechnology (SC-9989) [87] |
| Anti-ZO-1 Antibody | Immunofluorescence staining to visualize and assess tight junction integrity. | Cell Signaling Technologies (8193) [87] |
| Isolectin GS-IB4 | Labels endothelial cells to visualize the entire vascular network. | ThermoFisher Scientific (121413) [87] |
| Hematoxylin and Eosin (H&E) Stain Kit | General histological staining of the isolated vascular flat mounts. | Vector Laboratories (H-3502) [87] |
Workflow and Signaling Impact Visualization
The experimental workflow and the critical impact of enzyme selection on vascular junction integrity are summarized in the following diagrams.
Diagram 1: Experimental workflow for retinal vasculature isolation.
Diagram 2: Enzyme impact on vascular junction integrity.
This comparative case study underscores a critical principle in enzymatic tissue dissociation: the optimal enzyme is dictated by the specific research objective. For standard cell culture passaging of adherent monolayers, trypsin-EDTA remains a robust and efficient standard [24]. However, for specialized applications requiring the isolation of intact native microstructures—such as the retinal vasculature for the study of diabetic retinopathy—elastase digestion is the superior technique. Its ability to preserve pericyte-endothelial cell associations, adherens junctions, and tight junctions provides a more reliable and accurate model for investigating microvascular pathophysiology and for generating high-quality, reproducible data [87] [88]. Researchers should therefore carefully consider the structural endpoints of their experiments when selecting a dissociation or digestion enzyme.
Conclusion
Trypsin-EDTA remains a fundamental tool for cell dissociation, but its application requires careful consideration of specific research contexts. This guide demonstrates that while trypsin-EDTA offers efficiency for routine passaging, alternative enzymes like collagenase and hyaluronidase may better preserve stem cell populations in organoid generation, and elastase proves superior for maintaining specialized cellular junctions. Future directions point toward increased automation for reproducibility, development of gentler enzyme cocktails for sensitive cell types, and tissue-specific formulations that maintain native cell states. Researchers must validate dissociation methods against their specific experimental endpoints, particularly when working with precious primary samples or advanced disease models, to ensure biological fidelity in translational research.
References
- 1. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOookNJmfCjLHwozfkMYYCvQsbX54Hu5PhSbG4OG3ihgoeCTtLGZy]
- 2. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOorWAmUyx84br9rmeR5Ccvh66UR_M-15I5rGWU1ia2sdmJkrcf4N]
- 3. Tips for Successful Subculturing: Dissociation Method [https://www.atcc.org/resources/application-notes/tips-for-successful-sub-culturing-dissociation-method]
- 4. Enzymatic and Non-Enzymatic Cell Dissociation Protocols [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 5. Dissociation Protocols [https://www.cytion.com/us/Knowledge-Hub/Cell-Culture-Techniques/Dissociation-Protocols/]
- 6. Systematic assessment of tissue dissociation and storage ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-020-02048-6]
- 7. A method to quantitatively characterize the formation and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11705735/]
- 8. Trypsin-EDTA Solution for Cell Dissociation 2025-2033 ... [https://www.datainsightsmarket.com/reports/trypsin-edta-solution-for-cell-dissociation-990315]
- 9. North America Cell Culture Media Additives Market Size ... [https://www.linkedin.com/pulse/north-america-cell-culture-media-additives-wdhce]
- 10. How Trypsin-EDTA Solution For Cell Dissociation Works [https://www.linkedin.com/pulse/how-trypsin-edta-solution-cell-dissociation-works-one-worff]
- 11. Adherent Cell Culture Protocol [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/subculturing-adherent-cells.html]
- 12. PBS (Phosphate Buffered Saline) (1X, pH 7.4) Preparation ... [https://www.aatbio.com/resources/buffer-preparations-and-recipes/pbs-phosphate-buffered-saline]
- 13. Overview: Phosphate Buffered Saline (PBS) [https://www.bostonbioproducts.com/overview-phosphate-buffered-saline-pbs/]
- 14. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOopM8tlZtL_coqZYogszfM4DG6ivxK6GV40pCB1-NTesO7bq67We]
- 15. EDTA Solution 1X 0.25% Trypsin and 0.001% ... [https://www.diagnocine.com/Product/Trypsin-EDTA-Solution-1X-025-Trypsin-and-0001-EDTA-in-Dulbeccos-Phosphate-Buffered-Saline-Sterile-filtered/65716?srsltid=AfmBOoq8gz_OTRBfjiIhWYXOnU1xWoV2TvLJ8Yj_Kg3yEZ5lErqgXGvV]
- 16. Parameter Optimization and Capacitance-Based ... [https://www.mdpi.com/2227-9717/12/9/1887]
- 17. A beginners guide to Sf9 and Sf21 insect cell line culture ... [https://www.nature.com/articles/s41598-025-99812-0]
- 18. Adherent versus suspension based platforms: what is the ... [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/2244/Adherent-versus-suspension-based-platforms-what-is-the-near-future-of-viral-vector-manufacturing]
- 19. Recent advancements in tissue dissociation techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12622302/]
- 20. Guide to Bacterial Endotoxin / LAL Testing - Life in the Lab [https://www.thermofisher.com/blog/life-in-the-lab/endotoxins-101/]
- 21. Limulus Lysate Test - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/limulus-lysate-test]
- 22. Biochemical principle of Limulus test for detecting bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3756735/]
- 23. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOoqoONQi-FHr0UZYdj1QzLikqE6gYdfE1-mLwmaxNxalpw1ddwCC]
- 24. Cell Dissociation and Trypsin for Cell Culture [https://www.thermofisher.com/us/en/home/life-science/cell-culture/mammalian-cell-culture/reagents/cell-dissociation.html]
- 25. Seeding, Subculturing, and Maintaining Cells [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/maintaining-cultured-cells.html]
- 26. Trypsinization [https://grokipedia.com/page/Trypsinization]
- 27. Cell Culture Guide - Techniques and Protocols [https://www.assaygenie.com/cell-culture-guide-techniques-and-protocols/?srsltid=AfmBOopE1SzLyb4_uwZOE8anrV5kLG4rxkE2iGwv7gVaR6S_nH7JwzYY]
- 28. How to do a Proper Cell Culture Quick Check | Learn & Share [https://www.leica-microsystems.com/science-lab/life-science/how-to-do-a-proper-cell-culture-quick-check/]
- 29. Impact of trypsin on cell cytoplasm during detachment of ... [https://www.sciencedirect.com/science/article/pii/S0006349524004065]
- 30. A Deep Learning-Based Model Approach for Quantitative ... [https://www.mdpi.com/1424-8220/25/11/3515]
- 31. Trypsinization Automation Guide for Laboratory Sciences [https://opentrons.com/applications/trypsinization?srsltid=AfmBOooCwFgOADRJjSdomKWYF5B05gV5OWDUJBFybg19g6MeLy1wUNej]
- 32. Cell Culture Trends To Watch In 2025 [https://www.bioprocessonline.com/doc/cell-culture-trends-to-watch-in-0001]
- 33. Optimization of a Clinically Relevant Chemical-Mechanical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8175683/]
- 34. Optical sensor reveals the hidden influence of cell ... [https://www.nature.com/articles/s41598-024-61485-6]
- 35. Trypsin-induced proteome alteration during cell subculture in mammalian cells - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2873939/]
- 36. Development of a rapid Focus Reduction Neutralization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7864545/]
- 37. CoreTIA: a modular, statistically robust transduction ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1623848/full]
- 38. Different methods of detaching adherent cells and their ... [https://www.nature.com/articles/s41598-022-09605-y]
- 39. Protocol for monitoring of neutralizing antibodies to various ... [https://www.sciencedirect.com/science/article/pii/S2666166725003326]
- 40. Neutralizing antibody durability and SARS-CoV-2 infection ... [https://www.nature.com/articles/s41392-025-02437-y]
- 41. Nattokinase as a novel food-grade enzyme for long term ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996925001371]
- 42. Protocol to study juxtacrine signaling in human vascular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12056380/]
- 43. Specific cell-permeable inhibitor of proteasome trypsin-like ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3134264/]
- 44. Hypersonic levitation and spinning: paving the way for ... [https://www.nature.com/articles/s44172-025-00497-0]
- 45. Quality controls after tissue dissociation for single cell RNA ... [https://singleron.bio/quality-controls-after-tissue-dissociation-for-single-cell-rna-sequencing/]
- 46. Impact of enzymatic isolation on the propagation efficiency ... [https://pubmed.ncbi.nlm.nih.gov/40251201/]
- 47. Factors Affecting Cell Viability during the Enzymatic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11429318/]
- 48. Factors Affecting Cell Viability during the Enzymatic ... [https://www.mdpi.com/2079-7737/13/9/665]
- 49. How Recombinant Trypsin Solution For EDTA ... Cell Dissociation [https://www.linkedin.com/pulse/how-recombinant-trypsin-edta-solution-cell-dissociation-29kde]
- 50. The Use of Mild Trypsinization Conditions in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2025691/]
- 51. cell detachment by Trypsin-EDTA loses N-cadherin and ... [https://www.sciencedirect.com/science/article/abs/pii/S0006291X19312033]
- 52. Effects of Different Cell-Detaching Methods on the Viability ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5657749/]
- 53. Optimizing Cell Dissociation Using 0.25% Trypsin and ... [https://caissonlabs.com/optimizing-cell-dissociation-using-0-25-trypsin-and-1mm-edta-in-hanks-balanced-salt-solution/?srsltid=AfmBOopWl9rapDeoekfLbTIEOFvYkQaapeGMA6iUYw28IZphs4fKJlaa]
- 54. Evaluation of enzymatic protocols to optimize efficiency ... [https://www.nature.com/articles/s41538-024-00313-7]
- 55. Impact of enzymatic isolation on the propagation efficiency ... [https://www.nature.com/articles/s41598-025-97650-8]
- 56. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOopugBuegLtPdc5DPA3m5hJFCltA7-_cIHaU3YDRptJbt-sv1QXf]
- 57. Efficient and consistent cell dissociation using TrypsiNNex [https://novonordiskpharmatech.com/trypsinnex-application/efficient-and-consistent-cell-dissociation-using-trypsinnex/]
- 58. An improved way to detach cells from culture surfaces [https://news.mit.edu/2025/improved-way-detach-cells-culture-surfaces-1118]
- 59. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOor6IF4ZHX_vgs9LSJ6Hq5xKEzo1GAy-aaCkNM4BRvVaBRlxxmfz]
- 60. Comparison of Enzymatic and Non- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3055293/]
- 61. Automated—Mechanical Procedure Compared to Gentle ... [https://www.mdpi.com/2218-273X/12/5/701]
- 62. Trypsinization Automation Guide for Laboratory Sciences [https://opentrons.com/applications/trypsinization?srsltid=AfmBOorUI0f_CTI3ffKpq1ZHkdluvw9ou6MLkexZqCIEjekFn4eUi141]
- 63. A highly-reproducible automated proteomics sample ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6845026/]
- 64. Automated Tissue Dissociation for High-Quality Single-Cell ... [https://www.stemcell.com/products/brands/stemprep.html]
- 65. Temporal and thermal optimization of trypsin digestion for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12243500/]
- 66. Modified human skin cell isolation protocol and its ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11214264/]
- 67. Cell Culture Contamination | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/biological-contamination.html]
- 68. Basics of Cell Culture Quality Control: Contamination [https://info.abmgood.com/cell-culture-quality-control-contamination]
- 69. A Beginner's Guide to Cell Culture: Practical Advice for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10000895/]
- 70. Prevention of Contaminations in Cell Culture [https://ibidi.com/content/436-prevention-of-contaminations]
- 71. Recombinant Trypsin EDTA Solution | Cell Dissociation & ... [https://shop.sartorius.com/us/p/recombinant-trypsin-edta-solution/Recombinant_Trypsin_EDTA_Solution]
- 72. Cell Culture Protocols - Cell Biologics Inc. [https://www.cellbiologics.net/index.php?route=information/information&information_id=18]
- 73. Gibco™ TrypLE™ Express Enzyme (1X), phenol red [https://www.fishersci.ca/shop/products/gibco-tryple-express-enzyme-1x-phenol-red-4/12605010]
- 74. TrypLE™ Express Enzyme (1X), phenol red, 5 L - FAQs [https://www.thermofisher.com/store/v3/products/faqs/12605093]
- 75. TrypLE Express Enzyme (1X), No Phenol Red [https://sahoury.com/product/12604013]
- 76. Expansion of human embryonic stem cells: a comparative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6496537/]
- 77. TrypLE Protocol & Counting with an Automated Cell Counter [https://www.thermofisher.com/uk/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-counting-tryple-countess.html]
- 78. Collagenase Blends for Gentle Tissue Dissociation [https://www.merckmillipore.com/CR/es/technical-documents/technical-article/research-and-disease-areas/cell-signaling/collagenase-blends-for-gentle-tissue-dissociation]
- 79. Tissue Dissociation and Isolation - Prostate Epithelial Cell ... [https://www.stemcell.com/product-portfolios/prostate-epithelial-cell-research/tissue-dissociation-and-isolation.html]
- 80. Automated tissue dissociation for the establishment of ... [https://www.nature.com/articles/s41598-025-03905-9]
- 81. Patient-derived tumor organoids: a new avenue ... - Nature [https://www.nature.com/articles/s12276-024-01272-5]
- 82. Enzymatic and Non-Enzymatic Cell Dissociation Protocols [https://www.thermofisher.com/ng/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 83. Trypsin Cell Dissociation Protocol [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cell-dissociation-with-trypsin?srsltid=AfmBOopT5sVCoRpnCp8DpsfJ9-96ei1rXYQKw1TRN5iB8PyailxmESEq]
- 84. Generate Human Intestinal Organoid-Derived Monolayers [https://www.stemcell.com/how-to-generate-human-intestinal-organoid-derived-monolayers-using-intesticult.html]
- 85. Challenges in validation of combination treatment ... [https://jeccr.biomedcentral.com/articles/10.1186/s13046-024-03173-x]
- 86. Dissociating Enzymes: Trypsin | Worthington Biochemical [https://www.worthington-biochem.com/tissuedissociation/trypsin.html]
- 87. Comparative evaluation of trypsin and elastase digestion techniques for isolation of murine retinal vasculature - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11180566/]
- 88. Comparative evaluation of trypsin and elastase digestion ... [https://www.sciencedirect.com/science/article/abs/pii/S0026286224000311]
- 89. Beyond the known cuts: trypsin specificity in native proteins [https://pubs.rsc.org/en/content/articlehtml/2025/cc/d5cc02378e]
- 90. Elastase - Worthington Enzyme Manual [https://www.worthington-biochem.com/products/elastase/manual]
- 91. Elastase - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/elastase]
- 92. Elastase - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/neuroscience/elastase]